Advanced Synthetic Route for Sodium Ibandronate: Enhancing Purity and Scalability for Global Pharma
Introduction to Advanced Sodium Ibandronate Manufacturing
The pharmaceutical landscape for osteoporosis and oncology support continues to demand high-purity bisphosphonates, with Sodium Ibandronate standing out as a critical third-generation therapeutic agent. As detailed in patent CN102093416B, a transformative synthetic methodology has been developed that addresses the longstanding inefficiencies and safety concerns associated with traditional production. This novel approach leverages a streamlined three-step sequence that bypasses the need for hazardous phosphorus chlorinating agents, thereby offering a robust pathway for reliable sodium ibandronate supplier operations. The structural integrity and purity of the final active pharmaceutical ingredient are paramount, and this new route ensures that the complex gem-bisphosphonate core is constructed with precision.
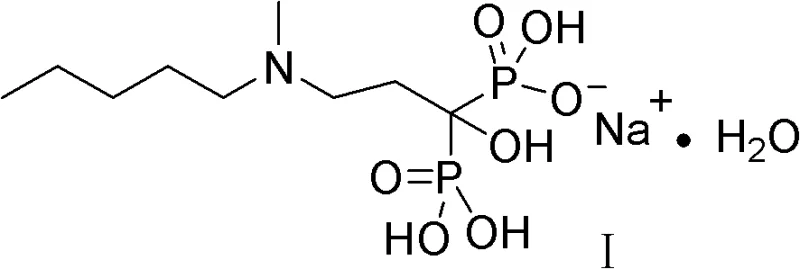
By shifting away from legacy chemistries that rely on aggressive reagents, this innovation not only enhances the safety profile of the manufacturing environment but also simplifies the downstream purification processes. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential for securing a stable supply of high-purity sodium ibandronate. The method capitalizes on the reactivity of dialkyl phosphites and N-methyl-beta-alanine derivatives, creating a convergent synthesis that minimizes waste and maximizes atom economy. This represents a significant leap forward in cost reduction in bisphosphonate manufacturing, aligning perfectly with modern green chemistry principles while maintaining the rigorous quality standards required for global regulatory approval.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Sodium Ibandronate has been plagued by convoluted reaction sequences and the utilization of extremely hazardous reagents. Traditional routes, such as the phenyl aldehyde method or the valeric acid pathway, often necessitate the use of phosphorus trichloride (PCl3), phosphorus pentachloride (PCl5), or phosphoryl chloride (POCl3) to introduce the phosphonic acid moieties. These reagents are not only highly toxic and corrosive but also generate substantial amounts of acidic waste that require complex neutralization and disposal protocols. Furthermore, these legacy methods frequently employ chlorobenzene as a reaction solvent, a substance classified with significant toxicity concerns and strict exposure limits, complicating worker safety and environmental compliance.
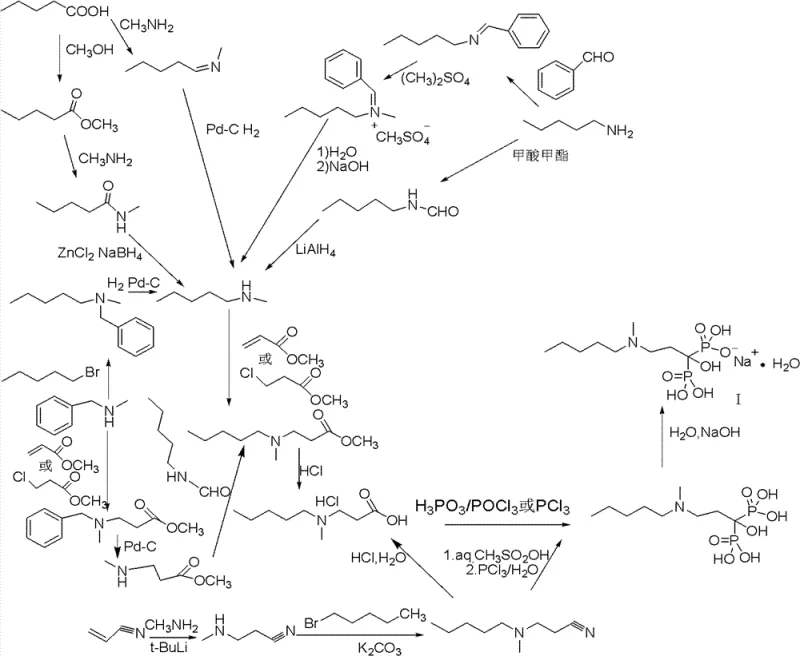
Beyond the safety hazards, the chemical efficiency of these conventional methods leaves much to be desired. The multi-step nature of the older pathways, often involving protection and deprotection strategies for amine groups, results in cumulative yield losses that are economically unsustainable. Literature data indicates that the overall yield for these traditional processes often hovers between a mere 9% and 15%, rendering them inefficient for large-scale commercial scale-up of complex bisphosphonates. The combination of low throughput, high hazard potential, and difficult waste management creates a fragile supply chain that is vulnerable to regulatory scrutiny and production delays, urging the industry to adopt safer and more efficient alternatives.
The Novel Approach
In stark contrast to the cumbersome legacy techniques, the methodology disclosed in CN102093416B introduces a streamlined and inherently safer synthetic strategy. This innovative route identifies a key intermediate, the tetraalkyl ester of [1-hydroxy-3-methylaminopropylidene]bisphosphonic acid, which serves as a versatile pivot point for the synthesis. By reacting this intermediate directly with halo-n-pentane, the method efficiently installs the requisite lipophilic side chain in a single alkylation step. This eliminates the need for the lengthy amine protection sequences seen in prior art, drastically shortening the production timeline and reducing the number of unit operations required to reach the final API.
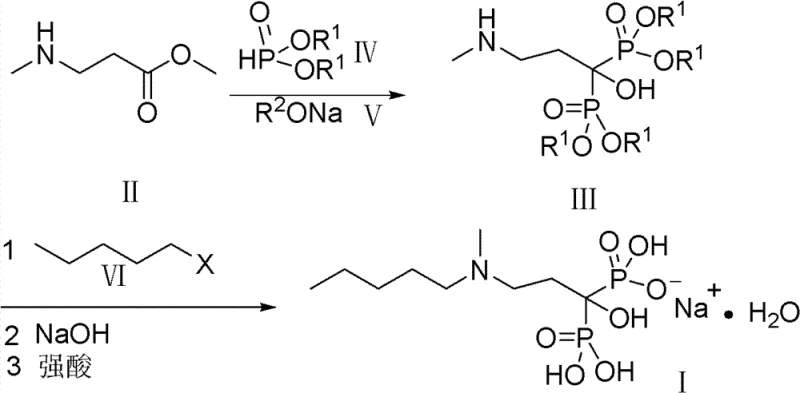
Furthermore, this novel approach replaces the toxic chlorobenzene solvent with benign, low-boiling alcohols such as methanol and ethanol. These solvents are not only safer for operators but are also easily removed during the drying process, ensuring minimal residual solvent levels in the final product. The avoidance of phosphorus chlorides in favor of dialkyl phosphites fundamentally changes the risk profile of the reaction, removing the generation of corrosive hydrogen chloride gas and simplifying the reactor material requirements. This shift facilitates a more robust and scalable process, enabling manufacturers to achieve overall yields exceeding 30%, which represents a substantial improvement in resource utilization and cost effectiveness for high-volume production.
Mechanistic Insights into Alkoxide-Catalyzed Bisphosphonate Formation
The core chemical transformation in this patented route revolves around the base-catalyzed condensation of N-methyl-beta-alanine methyl ester with dialkyl phosphite. In the presence of a sodium alkoxide catalyst, the ester undergoes a nucleophilic attack or condensation sequence that constructs the geminal bisphosphonate framework directly. This step is critical as it establishes the two carbon-phosphorus bonds that define the pharmacological activity of the molecule. The use of an alkoxide catalyst, typically present in molar ratios of 5% to 25% relative to the substrate, ensures that the reaction proceeds under mild thermal conditions, typically at the reflux temperature of the alcoholic solvent (60-108°C). This controlled environment prevents the degradation of sensitive functional groups and minimizes the formation of side products that could complicate purification.
Following the formation of the bisphosphonate intermediate, the subsequent N-alkylation with halo-n-pentane proceeds via a standard nucleophilic substitution mechanism. The secondary amine of the intermediate attacks the primary carbon of the haloalkane, displacing the halide ion to form the tertiary amine structure characteristic of Ibandronate. The choice of solvent in this step remains consistent with the green chemistry theme, utilizing alcohols that solvate both the organic intermediate and the inorganic byproducts effectively. Finally, the hydrolysis step employs sodium hydroxide to cleave the ester protecting groups, converting the tetraalkyl ester into the water-soluble tetrasodium salt. The precise control of pH during the final acidification step, targeting a range of 3.8 to 4.5, is crucial for inducing the crystallization of the monohydrate form, ensuring the correct polymorph and high purity specifications are met.
How to Synthesize Sodium Ibandronate Efficiently
Implementing this synthesis requires careful attention to reaction stoichiometry and temperature control to maximize the yield of the key intermediates. The process begins with the preparation of the bisphosphonate core, followed by the alkylation and final salt formation. Each step has been optimized to balance reaction rate with product stability, ensuring that the process is reproducible on a kilogram to tonne scale. The following guide outlines the critical operational phases derived from the patent examples, providing a roadmap for technical teams to evaluate feasibility.
- React N-methyl-beta-alanine methyl ester with dialkyl phosphite using an alkoxide catalyst to form the bisphosphonate tetraalkyl ester intermediate.
- Perform N-alkylation of the intermediate using halo-n-pentane in an alcoholic solvent under reflux conditions.
- Hydrolyze the resulting ester with sodium hydroxide, followed by acidification to pH 4.0 to crystallize pure Sodium Ibandronate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers compelling economic and logistical benefits. The primary advantage lies in the drastic simplification of the raw material portfolio; by eliminating the need for specialized and hazardous phosphorus chlorides, companies can source reagents from a broader, more competitive market. This diversification reduces dependency on single-source suppliers of dangerous chemicals and mitigates the risk of supply disruptions caused by stringent transportation regulations for hazardous goods. Additionally, the use of common alcoholic solvents simplifies solvent recovery and recycling infrastructure, further driving down operational expenditures.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic reagents like PCl5 and POCl3 directly lowers the bill of materials, while the higher overall yield (>30% vs <15%) means less raw material is wasted per kilogram of finished API. The simplified workup procedures, which avoid complex extraction steps required to remove chlorobenzene and phosphorus acid byproducts, reduce labor hours and energy consumption significantly. Consequently, the total cost of goods sold (COGS) is optimized, allowing for more competitive pricing in the generic pharmaceutical market without sacrificing margin.
- Enhanced Supply Chain Reliability: By utilizing reagents with lower hazard classifications, the logistical burden of transporting chemicals is substantially reduced. Alcohols and simple haloalkanes are widely available commodities with stable supply chains, unlike the tightly regulated phosphorus chlorides. This stability ensures consistent production schedules and reduces the lead time for high-purity sodium ibandronate batches. Furthermore, the robustness of the reaction conditions minimizes the risk of batch failures due to reagent sensitivity, guaranteeing a steady flow of material to downstream formulation partners.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing solvents that are easy to handle in large reactors and easy to remove via distillation. The absence of chlorinated solvents and toxic phosphorus waste streams simplifies wastewater treatment and aligns with increasingly strict environmental regulations globally. This 'green' profile not only reduces disposal costs but also enhances the corporate sustainability metrics of the manufacturer, making the supply chain more resilient to future regulatory changes and environmental audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on safety, yield, and operational parameters. Understanding these details is vital for technical teams assessing the transferability of this technology to their own manufacturing sites.
Q: How does this new synthesis route improve safety compared to traditional methods?
A: This method eliminates the use of highly toxic phosphorus chlorides (PCl3, PCl5, POCl3) and chlorobenzene solvents found in conventional routes, significantly reducing operational hazards and environmental impact.
Q: What is the expected yield improvement with this patented process?
A: The patented route achieves an overall synthesis yield exceeding 30%, which is substantially higher than the 9%-15% yield typically observed in existing literature methods.
Q: Can this process be scaled for commercial API production?
A: Yes, the process utilizes common alcoholic solvents like methanol and ethanol, avoids hazardous reagents, and involves straightforward crystallization steps, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sodium Ibandronate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity intermediates and APIs that adhere to stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in bisphosphonate chemistry allows us to navigate the complexities of this synthesis with precision, guaranteeing a supply that is both reliable and compliant with international standards.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and safe manufacturing strategy available.
