Advanced Manufacturing of High-Purity Entecavir Intermediates via Optimized Chiral Synthesis
Advanced Manufacturing of High-Purity Entecavir Intermediates via Optimized Chiral Synthesis
The global demand for effective antiviral therapies continues to drive innovation in the synthesis of nucleoside analogues, with Entecavir standing out as a potent inhibitor of Hepatitis B Virus (HBV) replication. As detailed in patent CN109293508B, a groundbreaking preparation method has been developed for synthesizing a key intermediate essential for Entecavir production. This technology addresses critical bottlenecks in traditional manufacturing by leveraging a unique chiral starting material, bis-benzyl (+)-Coreylactone diol, to achieve superior stereochemical control. The structural complexity of Entecavir, characterized by its cyclopentyl ring and purine base, necessitates precise synthetic strategies to ensure therapeutic efficacy and safety.
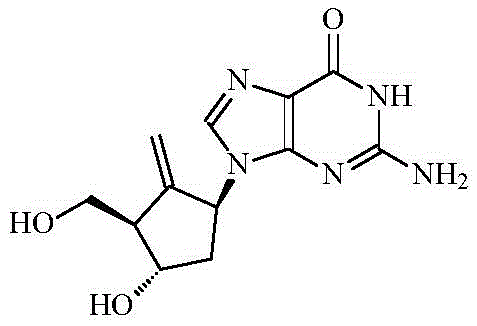
This novel approach not only streamlines the synthetic pathway but also significantly enhances the economic viability of producing high-purity antiviral intermediates. By bypassing the cumbersome gradual introduction of chiral centers found in legacy processes, this method offers a robust solution for pharmaceutical manufacturers seeking to optimize their supply chains. The integration of efficient acylation, protection, Grignard addition, and oxidation steps creates a seamless workflow that minimizes waste and maximizes yield, positioning this technology as a cornerstone for next-generation Entecavir manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Entecavir and its precursors has relied on routes originating from cyclopentadiene, as illustrated in earlier patents such as U.S. Pat. No. 4,364,44. These conventional pathways suffer from inherent inefficiencies, primarily due to the necessity of gradually introducing multiple chiral centers throughout the synthesis sequence. This stepwise construction of stereochemistry often results in the formation of difficult-to-separate optical isomers, necessitating rigorous and costly purification protocols such as repeated column chromatography. Furthermore, the cumulative yield losses across these multi-step sequences significantly inflate production costs and extend lead times, creating substantial supply chain vulnerabilities for API manufacturers.
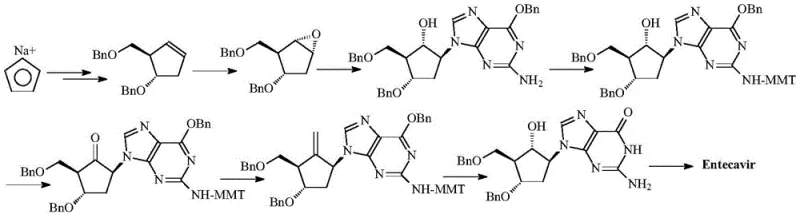
The reliance on complex chiral resolution techniques in traditional methods also introduces variability in product quality, posing risks to regulatory compliance and batch consistency. The environmental footprint of these older processes is another concern, as the generation of significant chemical waste from purification steps contradicts modern green chemistry mandates. Consequently, there is an urgent industry need for a streamlined alternative that can deliver high optical purity without the operational burdens of legacy synthesis.
The Novel Approach
The methodology disclosed in patent CN109293508B represents a paradigm shift by utilizing (+)-Coreylactone diol as a chiral pool starting material. This strategic choice eliminates the need for gradual chiral induction, as the requisite stereocenters are pre-installed in the starting material with high optical purity. The process involves a concise four-step sequence: acylation with tetrahydropyrrole, hydroxyl protection, Grignard addition, and a final Baeyer-Villiger oxidation. This streamlined workflow drastically reduces the number of unit operations and purification stages required, directly translating to improved process efficiency and lower manufacturing costs.
Moreover, the utilization of (+)-Coreylactone diol, which is often a byproduct in the synthesis of prostaglandin precursors, adds a layer of economic and environmental sustainability to the process. By valorizing a material that might otherwise be discarded or underutilized, this route achieves significant cost reduction in pharmaceutical intermediates manufacturing. The robustness of the reaction conditions, which avoid extreme temperatures and hazardous reagents where possible, further enhances the safety profile and scalability of the process, making it an ideal candidate for reliable entecavir intermediate supplier networks aiming for long-term stability.
Mechanistic Insights into the Four-Step Synthetic Sequence
The core of this innovative synthesis lies in the precise execution of four distinct chemical transformations that convert the bicyclic lactone starting material into the functionalized cyclopentyl intermediate. The initial step involves the reaction of bis-benzyl (+)-Coreylactone diol (Formula I) with tetrahydropyrrole. This acylation reaction, conducted either under solvent-free conditions or in an aprotic solvent at temperatures ranging from 40°C to reflux, effectively opens the lactone ring while installing the necessary nitrogen-containing side chain. The absence of solvent in preferred embodiments not only simplifies the workup but also intensifies the reaction rate, showcasing a commitment to process intensification principles.
Following acylation, the intermediate (Formula II) undergoes selective hydroxyl protection using silyl chlorides such as TBSCl or TESCl in the presence of imidazole. This step is critical for masking the reactive hydroxyl group to prevent unwanted side reactions during subsequent nucleophilic additions. The choice of polar aprotic solvents like DMF facilitates the dissolution of reagents and ensures homogeneous reaction conditions. The subsequent Grignard reaction involves the addition of methyl magnesium chloride to the protected intermediate (Formula III). Conducted in THF at controlled temperatures of 60-65°C, this step introduces the methyl ketone functionality essential for the final oxidative rearrangement.
The culmination of the synthesis is the Baeyer-Villiger oxidation of the methyl ketone (Formula IV) using peroxytrifluoroacetic acid. This transformation is pivotal as it inserts an oxygen atom adjacent to the carbonyl group, generating the acetate ester functionality found in the final intermediate (Formula V). The reaction is meticulously controlled at temperatures between 20-30°C in dichloromethane to ensure selectivity and prevent over-oxidation or degradation of the sensitive cyclopentyl scaffold.
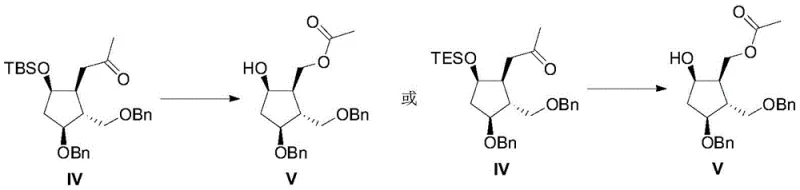
Impurity control is inherently managed through the high fidelity of these reactions. The use of a chiral starting material with defined stereochemistry minimizes the formation of diastereomeric impurities that typically plague racemic syntheses. Furthermore, the specific reaction conditions, such as the stoichiometric control of the Grignard reagent (1:1.2 ratio) and the oxidant, are optimized to suppress side products. This mechanistic precision ensures that the resulting high-purity entecavir intermediate meets the stringent quality specifications required for downstream coupling with the purine base, ultimately safeguarding the safety and efficacy of the final antiviral drug.
How to Synthesize Entecavir Intermediate Efficiently
The synthesis of the key Entecavir intermediate described in Formula V requires strict adherence to the optimized reaction parameters outlined in the patent to ensure maximum yield and purity. The process leverages standard organic synthesis techniques but applies them in a novel sequence that capitalizes on the chirality of the starting material. Operators must pay close attention to temperature control during the Grignard and oxidation steps, as these are exothermic and sensitive to thermal variance. The following guide summarizes the critical operational phases derived from the patented methodology.
- React bis-benzyl (+)-Coreylactone diol (Formula I) with tetrahydropyrrole under solvent-free or aprotic conditions at 40°C to reflux to obtain Formula II.
- Protect the hydroxyl group of Formula II using TBSCl or TESCl in the presence of imidazole and a polar aprotic solvent to yield Formula III.
- Perform a Grignard reaction on Formula III using methyl magnesium chloride in THF at 60-65°C to introduce the methyl ketone moiety, forming Formula IV.
- Subject Formula IV to Baeyer-Villiger oxidation using peroxytrifluoroacetic acid in dichloromethane at 20-30°C to generate the final intermediate Formula V.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers compelling strategic advantages that extend beyond mere technical feasibility. The primary value proposition lies in the substantial cost savings achieved through raw material optimization and process simplification. By utilizing (+)-Coreylactone diol, a material often available as a byproduct from other high-volume pharmaceutical syntheses, manufacturers can secure a cost-effective feedstock that decouples production costs from volatile specialty chemical markets. This sourcing strategy enhances supply chain resilience and provides a buffer against price fluctuations common in the fine chemical sector.
- Cost Reduction in Manufacturing: The elimination of multiple column chromatography steps, which are resource-intensive and time-consuming, results in a drastic reduction in operational expenditures. Traditional methods often require silica gel and large volumes of solvents for purification, generating significant hazardous waste. By contrast, this novel route relies on crystallization or simpler extraction methods facilitated by the high selectivity of the reactions. The removal of expensive transition metal catalysts or complex chiral resolving agents further contributes to a leaner cost structure, allowing for more competitive pricing in the global API market.
- Enhanced Supply Chain Reliability: The robustness of the four-step sequence ensures consistent batch-to-batch quality, which is critical for maintaining regulatory approval and avoiding production delays. The use of commodity reagents such as Grignard reagents, silyl chlorides, and peroxides means that supply disruptions are unlikely, as these chemicals are widely available from multiple global vendors. This diversification of the supply base reduces the risk of single-source dependency, ensuring that production schedules for high-purity antiviral intermediates can be met reliably even during periods of market instability.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex chiral intermediates, with reaction conditions that are easily transferable from pilot plant to multi-ton production scales. The reduction in solvent usage and waste generation aligns with increasingly stringent environmental regulations, minimizing the liability and cost associated with waste disposal. The ability to produce large quantities of the intermediate with a smaller environmental footprint supports corporate sustainability goals and facilitates smoother regulatory audits, thereby reducing lead time for high-purity antiviral intermediates to reach the market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities.
Q: How does this new synthesis route improve optical purity compared to traditional methods?
A: Traditional routes starting from cyclopentadiene require the gradual introduction of chiral centers, which often leads to difficult-to-control optical impurities and requires extensive column chromatography. This novel method utilizes (+)-Coreylactone diol, a starting material that already possesses the required chiral centers. This 'chiral pool' approach inherently ensures high optical purity from the outset, significantly reducing the burden of chiral separation and impurity control during downstream processing.
Q: What are the cost advantages of using (+)-Coreylactone diol as a starting material?
A: The (+)-Coreylactone diol used in this process is typically a byproduct or enantiomer generated during the synthesis of prostaglandin starting materials, where the (-)-enantiomer is the primary target. By utilizing this otherwise underutilized enantiomer, the process transforms a waste stream or low-value byproduct into a high-value pharmaceutical intermediate. This strategy drastically reduces raw material costs and minimizes waste treatment expenses, aligning with green chemistry principles.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the route is specifically designed for industrial scalability. It eliminates the need for multiple, labor-intensive column chromatography steps that plague conventional methods. The reactions utilize standard reagents like Grignard reagents and peroxy acids under manageable temperature conditions (0-65°C). The simplified purification requirements and the use of robust chemical transformations make this method highly amenable to commercial scale-up, ensuring consistent supply and reduced lead times.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Entecavir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of key pharmaceutical building blocks. Our team of expert chemists has thoroughly analyzed the technological potential of the route described in CN109293508B and is fully prepared to implement this advanced synthesis at an industrial scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Entecavir intermediate delivered meets the highest international standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your antiviral drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this optimized route can enhance your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to drive innovation and efficiency in the global fight against Hepatitis B.
