Advanced Synthesis of Tert-Butyl 2-Fluoro-4-Bromobenzoate for Commercial Scale-Up
Introduction to Advanced Intermediate Synthesis
The development of efficient synthetic routes for halogenated aromatic esters is a cornerstone of modern medicinal chemistry, particularly for constructing complex biaryl scaffolds found in kinase inhibitors and GPCR modulators. Patent CN113264832A, published in August 2021, introduces a significant methodological breakthrough in the preparation of tert-butyl 2-fluoro-4-bromobenzoate, a critical building block for various pharmaceutical candidates. This technology addresses long-standing inefficiencies in traditional esterification protocols by leveraging the unique reactivity profile of lithium alkoxides over their potassium counterparts. For R&D directors and process chemists, this represents a viable pathway to enhance throughput while minimizing impurity profiles that often plague scale-up campaigns. By shifting the paradigm from solid-phase potassium bases to soluble lithium species generated in situ, the process achieves a marked improvement in both conversion rates and operational simplicity, positioning it as a robust candidate for reliable pharmaceutical intermediate supplier networks seeking to optimize their raw material sourcing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sterically hindered tert-butyl esters containing sensitive halogen substituents has relied heavily on potassium tert-butoxide (tBuOK) as the nucleophilic driver. However, this conventional approach suffers from inherent physicochemical limitations that severely impact process economics and yield. The poor solubility of potassium tert-butoxide in common organic solvents often necessitates the use of heterogeneous reaction conditions, which drastically reduces mass transfer efficiency and overall reaction kinetics. To compensate for this sluggish reactivity, prior art methods frequently demand the use of excessive reagent quantities, typically exceeding 2.0 equivalents, which not only inflates raw material costs but also exacerbates the formation of undesirable by-products. Furthermore, the aggressive nature of excess base combined with prolonged reaction times often leads to conversion rates stagnating below 50%, creating a massive burden on downstream purification units. This low efficiency translates directly into increased waste generation and extended production cycles, making the traditional potassium-based route suboptimal for cost reduction in pharma manufacturing where margin compression is a constant concern.
The Novel Approach
In stark contrast, the methodology disclosed in the referenced patent utilizes an in situ generation of lithium tert-butoxide (tBuOLi) via the reaction of n-butyllithium with tert-butanol in tetrahydrofuran (THF). This strategic substitution capitalizes on the smaller ionic radius and higher charge density of the lithium cation, which significantly enhances the nucleophilicity and solubility of the alkoxide species within the reaction matrix. As illustrated in the reaction scheme below, the process begins with the activation of 2-fluoro-4-bromobenzoic acid to its corresponding acid chloride, followed by a highly controlled coupling with the lithium alkoxide.
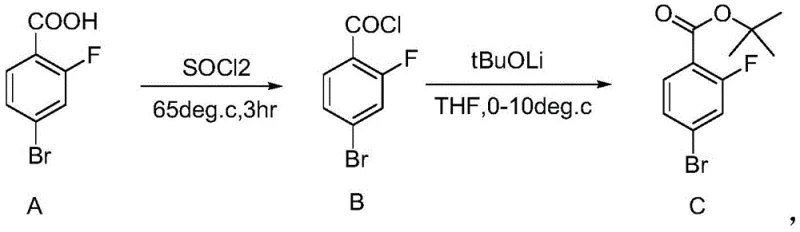
This novel approach allows for a drastic reduction in reagent stoichiometry, requiring only 1.1 to 1.5 equivalents to drive the reaction to completion, thereby minimizing chemical waste. The superior solubility of the lithium species ensures a homogeneous reaction environment, facilitating rapid heat dissipation and consistent mixing even at larger scales. Consequently, this method achieves a two-step yield of approximately 64% with a gas chromatography purity of 96%, demonstrating a clear superiority over legacy techniques. For supply chain managers, this translates to a more predictable and streamlined manufacturing process that reduces the risk of batch failures and ensures a steady flow of high-purity pharmaceutical intermediates to downstream API synthesis facilities.
Mechanistic Insights into Lithium-Mediated Esterification
The core mechanistic advantage of this process lies in the nucleophilic acyl substitution mechanism facilitated by the lithium alkoxide. Unlike the potassium analog, which often exists as aggregated clusters with limited accessibility to the electrophilic carbonyl carbon of the acid chloride, the lithium tert-butoxide species exhibits a higher degree of dissociation in THF. This results in a more "naked" and reactive alkoxide anion that can efficiently attack the carbonyl center of the 2-fluoro-4-bromobenzoyl chloride intermediate. The reaction is conducted at a controlled temperature range of 0-10°C, which is critical for suppressing potential side reactions such as elimination or nucleophilic aromatic substitution on the fluorine or bromine positions. The use of n-butyllithium to generate the alkoxide allows for precise stoichiometric control, ensuring that the basicity of the system is tuned exactly to the requirements of the esterification without introducing excess strong base that could degrade the sensitive halogenated substrate.
Furthermore, the impurity control mechanism is inherently built into the solubility characteristics of the by-products. The lithium chloride salt generated during the reaction possesses different solubility properties compared to potassium salts, often allowing for easier separation during the aqueous workup phase. The protocol specifies a rigorous workup involving methyl tert-butyl ether (MTBE) extraction and brine washing, which effectively removes residual lithium salts and unreacted starting materials. The final purification via vacuum distillation at 120°C oil bath temperature and 3.8mmHg pressure ensures the removal of any high-boiling oligomers or trace solvent residues. This level of mechanistic understanding allows process engineers to design robust control strategies for the commercial scale-up of complex pharmaceutical intermediates, ensuring that the critical quality attributes (CQAs) of the final ester meet the stringent specifications required for GMP manufacturing environments.
How to Synthesize Tert-Butyl 2-Fluoro-4-Bromobenzoate Efficiently
The synthesis protocol outlined in the patent provides a detailed, step-by-step guide for executing this transformation with high reproducibility. It begins with the activation of the carboxylic acid using thionyl chloride, followed by the careful generation of the lithium alkoxide reagent under inert atmosphere. The subsequent coupling reaction requires precise thermal management to maintain the integrity of the halogenated aromatic ring. While the general workflow is straightforward, adherence to the specific temperature gradients and addition rates described in the intellectual property is essential for maximizing yield. For laboratory and pilot plant teams looking to implement this route, the following standardized steps provide the foundational framework for operation, though detailed engineering parameters should be validated against specific equipment configurations.
- Convert 2-fluoro-4-bromobenzoic acid to 2-fluoro-4-bromobenzoyl chloride using thionyl chloride at 60-65°C.
- Generate lithium tert-butoxide by reacting tert-butanol with n-butyllithium in THF at 0-10°C.
- React the acid chloride with the lithium tert-butoxide solution, followed by vacuum distillation to isolate the high-purity ester.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this lithium-mediated synthesis route offers substantial strategic benefits for procurement and supply chain stakeholders. The primary value driver is the significant simplification of the reaction stoichiometry, which directly correlates to reduced raw material consumption and lower waste disposal costs. By eliminating the need for large excesses of expensive alkoxide bases, manufacturers can achieve a leaner cost structure without compromising on output quality. Additionally, the improved solubility of the reaction intermediates allows for the use of smaller reactor volumes relative to the output mass, effectively increasing the volumetric productivity of existing manufacturing assets. This efficiency gain is crucial for reducing lead time for high-purity pharmaceutical intermediates, as it enables faster batch turnover and more responsive fulfillment of customer orders in a volatile market environment.
- Cost Reduction in Manufacturing: The shift to a lithium-based system eliminates the economic drag associated with low-conversion potassium reactions. By reducing the equivalent ratio of the base from >2.0 to roughly 1.1-1.5, the direct material cost per kilogram of product is significantly lowered. Moreover, the higher yield (64% over two steps compared to typically lower yields in older methods) means less starting material is wasted, further driving down the effective cost of goods sold (COGS). The simplified workup procedure also reduces the consumption of solvents and filtration media, contributing to overall operational expenditure savings.
- Enhanced Supply Chain Reliability: The operational robustness of this method enhances supply continuity. The ability to perform the alkoxide generation at slightly higher temperatures (0-10°C) compared to cryogenic conditions required for some other organolithium reactions simplifies the cooling infrastructure requirements. This makes the process more adaptable to standard multipurpose reactors found in most fine chemical plants, reducing the dependency on specialized equipment that might create bottlenecks. Consequently, suppliers can offer more reliable delivery schedules and maintain safety stock levels with greater confidence, mitigating the risk of supply disruptions for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable due to the homogeneous nature of the reaction mixture, which facilitates efficient heat transfer and mixing in large vessels. The reduced generation of solid waste (inorganic salts) aligns with increasingly stringent environmental regulations regarding waste disposal in the chemical sector. By minimizing the volume of hazardous waste and optimizing solvent recovery through distillation, this route supports sustainable manufacturing practices. This environmental compliance is a key factor for multinational corporations aiming to reduce their carbon footprint and meet corporate social responsibility (CSR) goals while sourcing critical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of this specialized intermediate. These insights are derived directly from the patented technology and practical experience in scaling similar halogenated esterification processes. Understanding these nuances is vital for technical buyers evaluating the feasibility of integrating this building block into their synthesis pipelines. The answers provided reflect the current state-of-the-art capabilities in producing this molecule with the requisite purity and consistency.
Q: Why is lithium tert-butoxide preferred over potassium tert-butoxide for this synthesis?
A: Lithium tert-butoxide offers superior solubility in THF and higher reactivity due to the smaller ionic radius of lithium, allowing for lower stoichiometric usage (1.1-1.5 eq) compared to potassium salts which often require excess amounts and result in lower conversion rates.
Q: What are the critical temperature controls for the esterification step?
A: The reaction requires strict temperature maintenance between 0-10°C during the addition of n-butyllithium and the subsequent coupling with the acid chloride to prevent side reactions and ensure high selectivity.
Q: How is the final product purified to achieve high purity?
A: The crude oily liquid is purified via reduced pressure distillation, collecting fractions at an overhead temperature of 106-108°C under a pressure difference of 3.8mmHg, yielding a product with approximately 96% GC purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tert-Butyl 2-Fluoro-4-Bromobenzoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our technical team has extensively analyzed the pathway described in CN113264832A and possesses the expertise to execute this lithium-mediated esterification with precision. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met whether you are in the early discovery phase or preparing for full-scale commercialization. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of tert-butyl 2-fluoro-4-bromobenzoate meets the exacting standards required for pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain. By leveraging our process optimization capabilities, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that balance cost, quality, and delivery timelines effectively.
