Revolutionizing Indole Derivative Production: High-Yield Synthesis for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for bioactive scaffolds, particularly those serving as critical building blocks for enzyme inhibitors. Patent CN1130343C discloses a groundbreaking methodology for the synthesis of indolyl-3-acetamides, a class of compounds identified as potent non-substrate analog inhibitors of phospholipase A2 (PLA2). These inhibitors hold profound significance for the treatment of rheumatoid arthritis, addressing a critical therapeutic need where traditional substrate analogues often fail due to metabolic instability caused by conflicting hydrophobic and hydrophilic groups within the molecule. The disclosed technology represents a paradigm shift from previous multi-step, low-yield processes to a streamlined, high-efficiency protocol that leverages the abundant availability of indomethacin as a starting feedstock. By optimizing reaction conditions and selecting superior activation strategies, this innovation delivers a total yield that drastically outperforms historical benchmarks, positioning it as a vital asset for manufacturers aiming to secure a reliable pharmaceutical intermediate supplier status in the competitive anti-inflammatory market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, specifically the synthetic route disclosed by Eli Lilly and Company in the Journal of Medicinal Chemistry, relied on a cumbersome four-step sequence to construct the indole core and install the necessary functional groups. This legacy approach suffered from severe inefficiencies, particularly in the initial cyclization and final hydrogenation steps, which exhibited yields as low as 36% and 25% respectively. Furthermore, the lack of regioselectivity in multiple stages resulted in a complex mixture of byproducts, necessitating rigorous and costly purification via column chromatography. Such reliance on chromatographic separation is a major bottleneck in industrial chemistry, rendering the process economically unviable for large-scale manufacturing due to high solvent consumption, extended processing times, and significant material loss. The cumulative effect of these inefficiencies resulted in a dismal overall yield of merely 5.88%, creating substantial supply chain vulnerabilities and inflating the cost of goods for downstream API production.
The Novel Approach
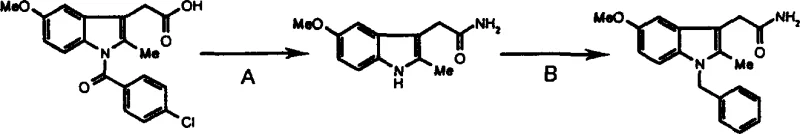
In stark contrast, the novel methodology presented in the patent data utilizes a strategic retrosynthetic disconnection that capitalizes on the pre-existing indole scaffold of indomethacin. This approach condenses the synthesis into just two high-yielding steps, bypassing the need for de novo indole ring construction. The first step employs a mixed anhydride activation strategy to convert the carboxylic acid moiety directly into the primary amide with exceptional efficiency. The subsequent N-alkylation step introduces the benzyl group under controlled basic conditions, ensuring high selectivity for the nitrogen atom without compromising the integrity of the sensitive amide linkage. This streamlined workflow not only simplifies the operational complexity but also facilitates easier isolation of the product through simple recrystallization techniques, effectively eliminating the need for column chromatography and making the process ideally suited for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Mixed Anhydride Activation and N-Alkylation
The cornerstone of this synthetic success lies in the precise application of the mixed anhydride method during the initial amidation phase. By reacting indomethacin with ethyl chloroformate in the presence of N-methylmorpholine at cryogenic temperatures (-10 to -15°C), a highly reactive mixed anhydride intermediate is generated in situ. This activated species is significantly more electrophilic than the parent carboxylic acid, allowing for rapid nucleophilic attack by ammonia upon warming to room temperature. The kinetic control afforded by the low-temperature activation minimizes racemization or degradation of the chiral centers if present, although indomethacin itself is achiral at the reaction site, ensuring a clean conversion to the amide. Following ammonolysis, a hydrolysis step with sodium hydroxide ensures the removal of any unreacted anhydride or ester byproducts, driving the equilibrium towards the desired primary amide with a remarkable isolated yield of 95.6%.
Impurity control is further enhanced in the second stage through the careful selection of base and solvent for the N-alkylation. The use of sodium hydride in dimethylformamide (DMF) generates the indole anion quantitatively, which then undergoes an SN2 reaction with benzyl bromide. The choice of DMF as a polar aprotic solvent stabilizes the transition state and solubilizes the ionic intermediates, facilitating a smooth reaction progression over 8 to 15 hours. Crucially, the reaction conditions are tuned to prevent O-alkylation or over-alkylation, which are common side reactions in indole chemistry. The result is a crude product of such high purity that it requires only a simple water wash and recrystallization from ethyl acetate to meet stringent quality specifications, demonstrating a level of process robustness that is essential for the commercial scale-up of complex heterocycles.
How to Synthesize Indolyl-3-Acetamide Efficiently
The synthesis of this high-value intermediate is achieved through a concise two-step protocol that begins with the activation of indomethacin using ethyl chloroformate and N-methylmorpholine in tetrahydrofuran, followed by ammonolysis to generate the free amine precursor. The subsequent alkylation with benzyl bromide in the presence of sodium hydride completes the transformation, offering a practical guide for laboratory and pilot plant operations. For detailed standardized operating procedures, safety protocols, and specific stoichiometric ratios required for GMP compliance, please refer to the technical guidelines below.
- Activate indomethacin with ethyl chloroformate and N-methylmorpholine at low temperature, followed by ammonolysis and hydrolysis to form the primary amide.
- Perform N-alkylation of the resulting indole amide using sodium hydride and benzyl bromide in DMF to yield the final target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented synthesis route offers transformative benefits that directly impact the bottom line and operational resilience. The dramatic improvement in overall yield, increasing from less than 6% in legacy methods to over 62% in this new process, fundamentally alters the cost structure of the final intermediate. This efficiency gain means that significantly less raw material is required to produce the same amount of product, leading to substantial cost savings in feedstock procurement. Furthermore, the elimination of column chromatography removes a major variable from the production schedule, reducing solvent waste disposal costs and freeing up valuable reactor capacity for additional batches. These factors combine to create a more predictable and economical manufacturing environment, which is critical for maintaining competitiveness in the global pharmaceutical market.
- Cost Reduction in Manufacturing: The implementation of this high-yield route eliminates the economic drag associated with multi-step syntheses that suffer from cumulative yield losses. By removing the need for expensive chromatographic purification media and reducing solvent consumption for separation, the variable cost per kilogram of product is drastically lowered. Additionally, the use of indomethacin, a widely available and inexpensive commodity chemical, as the starting material ensures that raw material costs remain stable and low, avoiding the price volatility often seen with specialized custom building blocks. This economic efficiency allows manufacturers to offer more competitive pricing to downstream API producers while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Relying on indomethacin as the primary feedstock leverages an established and robust global supply chain, mitigating the risk of shortages that can plague niche starting materials. The simplified two-step process reduces the number of unit operations and intermediate handling steps, which inherently lowers the probability of batch failures or delays due to equipment bottlenecks. With faster reaction times and simpler workup procedures involving filtration and crystallization rather than slow column loading and elution, the throughput of the manufacturing facility is significantly increased. This agility enables suppliers to respond more rapidly to fluctuating market demands and reduce lead times for high-purity intermediates, ensuring continuity of supply for critical drug development programs.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard unit operations such as stirred tank reactors and filtration units that are easily adaptable from pilot to commercial scale. The avoidance of column chromatography not only speeds up production but also aligns with green chemistry principles by significantly reducing the volume of organic solvents required for purification. This reduction in solvent usage lowers the environmental footprint of the manufacturing process, simplifying waste management and ensuring compliance with increasingly stringent environmental regulations. The mild reaction conditions, particularly the ambient temperature alkylation, further contribute to energy efficiency, making this a sustainable choice for long-term industrial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of indolyl-3-acetamide derivatives, based on the specific advantages outlined in the patent literature. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains. The answers provided reflect the optimized parameters and yield data confirmed through experimental examples in the source documentation.
Q: What is the primary advantage of this new synthesis route over conventional methods?
A: The new route utilizes indomethacin as a starting material and achieves a total yield of 62.5%, which is approximately 10.6 times higher than the conventional Eli Lilly method (5.88%), while eliminating the need for complex column chromatography.
Q: Why is the mixed anhydride method preferred for this transformation?
A: The mixed anhydride method allows for mild reaction conditions and high selectivity during the amide formation step, resulting in a 95.6% yield for the first step and minimizing the formation of side products that complicate purification.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly suitable for mass production because it uses readily available raw materials, involves only two steps with fast reaction rates, and relies on simple workup procedures like recrystallization rather than labor-intensive chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolyl-3-Acetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic routes play in the successful commercialization of new therapeutic agents. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1130343C are fully realized in practical manufacturing settings. Our state-of-the-art facilities are equipped to handle the specific requirements of indole chemistry, including the safe handling of reagents like sodium hydride and the precise temperature control needed for mixed anhydride formation. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for pharmaceutical applications, providing our partners with the confidence they need to advance their drug candidates.
We invite potential partners to engage with our technical team to explore how this optimized synthesis can benefit your specific project requirements. By leveraging our expertise, you can access a Customized Cost-Saving Analysis that quantifies the potential economic impact of switching to this high-yield route for your supply chain. We encourage you to contact our technical procurement team today to request specific COA data for our indole derivatives and to discuss route feasibility assessments tailored to your development timeline. Together, we can accelerate the delivery of life-saving medications to patients worldwide through superior chemical manufacturing.
