Advanced Synthesis of Lysosome-Targeted Fluorescent Probes for Commercial Scale-Up and High-Purity Applications
The rapid advancement of modern cell micro-optical imaging technology has created an urgent demand for fluorescent reagents that combine high efficiency, sensitivity, and specific organelle targeting capabilities. Patent CN101962536A addresses this critical gap by disclosing a novel lysosome-targeted fluorescent substance and its robust synthesis method, representing a significant breakthrough in the field of biological imaging intermediates. This technology enables the visual dynamic labeling of organelle fine functions, providing intuitive scientific basis for research into human diseases and cell aging. Unlike traditional methods that rely on unstable antibodies or phototoxic dyes, this invention utilizes a unique axisymmetric compound structure capable of visible light fluorescence emission under both ultraviolet and near-infrared two-photon excitation. For R&D directors and procurement specialists in the life sciences sector, understanding the synthetic pathway detailed in this patent is essential for securing a reliable lysosome imaging agent supplier that can deliver high-purity materials at a commercially viable scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the microscopic imaging of cells and organelles has been plagued by significant technical hurdles, particularly regarding the availability of effective targeting agents. Conventional approaches often rely on dyes that suffer from severe photobleaching and phototoxicity, losing fluorescence activity rapidly under ultraviolet irradiation or generating harmful singlet oxygen that damages living cells. Alternatively, antibody-based methods, while offering low toxicity, are constrained by their exorbitant costs, susceptibility to inactivation, and limited shelf-life, which severely restricts their application in long-term cell imaging studies. Furthermore, the range of available options for both dyes and antibodies is relatively narrow, failing to meet the diverse needs of scientific research regarding fluorescence excitation ranges and specific organelle targeting precision. These limitations create a bottleneck in the development of new drugs and the study of cellular mechanisms, necessitating a shift towards more stable and cost-effective small molecule alternatives.
The Novel Approach
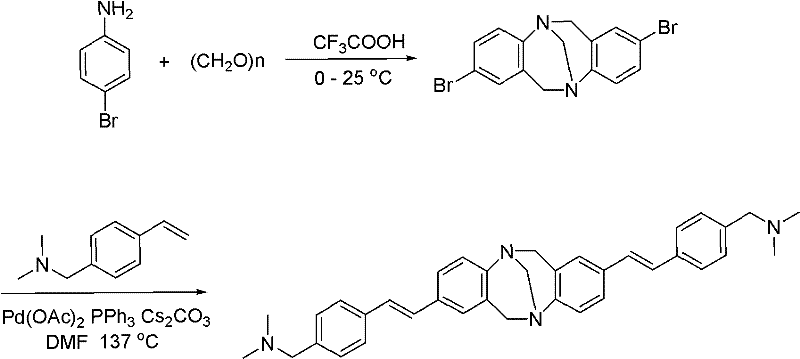
The methodology presented in patent CN101962536A overcomes these historical barriers through a streamlined two-step synthesis that effectively applies the Heck reaction to construct a complex, axisymmetric fluorophore. By utilizing 4-bromoaniline and paraformaldehyde as inexpensive starting materials, the process generates a bromine-substituted intermediate which is subsequently functionalized via palladium-catalyzed coupling. This novel approach not only simplifies the synthesis path but also significantly enhances the yield and stability of the final product compared to traditional biological staining agents. The resulting compound features a specific N,N-dimethylbenzylamine bioactive group that ensures precise targeting of cell lysosomes without the associated toxicity of conventional dyes. For supply chain managers, this translates to a more resilient sourcing strategy for high-purity cell imaging reagents, as the reliance on fragile biological components is eliminated in favor of robust chemical synthesis.
Mechanistic Insights into Acid-Catalyzed Cyclization and Heck Coupling
The core of this synthesis lies in the initial formation of the bridged heterocyclic scaffold, achieved through a trifluoroacetic acid (TFA) catalyzed condensation reaction. In this step, 4-bromoaniline reacts with paraformaldehyde in a molar ratio ranging from 1:2 to 1:2.5, with TFA serving dual roles as both the solvent and the reaction catalyst. The reaction is conducted at a controlled temperature between 0°C and 25°C for an extended period of 70 to 160 hours, allowing for the gradual formation of the 5,11-dibromo-[1,5]diazocine intermediate. This mild acidic environment is crucial for promoting the electrophilic aromatic substitution required to build the rigid central core while minimizing decomposition of the sensitive amine functionalities. Following the reaction, the mixture is neutralized to a pH of 7-8 and extracted, yielding the intermediate as a crystalline solid, which serves as the foundational block for the subsequent fluorescence-enhancing modifications.
The second critical phase involves the palladium-catalyzed Heck coupling, which extends the conjugation system essential for the compound's fluorescent properties. Using the dibromo-intermediate as the substrate, the reaction introduces 4-vinyl-N,N-dimethylbenzylamine groups at the 5 and 11 positions using a catalyst system comprising palladium acetate (5-10%), triphenylphosphine (10-20%), and cesium carbonate (4-6 equivalents) in anhydrous DMF. Heating the system to 137°C under nitrogen protection for 8 to 10 hours facilitates the cross-coupling, effectively replacing the bromine atoms with the vinyl-amine moieties. This structural modification creates a donor-acceptor system across the carbon-carbon double bonds, expanding the electron transition delocalization range. Consequently, the molecule gains the ability to emit visible fluorescence upon excitation, while the dimethylbenzylamine groups provide the specific lysosome-targeting capability required for high-contrast biological imaging applications.
How to Synthesize Lysosome Targeted Fluorescent Substance Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing this advanced imaging agent, emphasizing strict control over reaction parameters to maximize yield and purity. The process begins with the careful mixing of 4-bromoaniline and paraformaldehyde, followed by the dropwise addition of excess trifluoroacetic acid to initiate the cyclization. After the prolonged reaction period, the workup involves neutralization and crystallization to isolate the key intermediate, ensuring that impurities are removed before the final coupling step. The subsequent Heck reaction requires rigorous exclusion of moisture and oxygen, utilizing dry solvents and nitrogen purging to maintain catalyst activity. For detailed operational specifics regarding stoichiometry, temperature ramps, and purification techniques, please refer to the standardized guide below.
- Condense 4-bromoaniline with paraformaldehyde in trifluoroacetic acid at 0-25°C for 70-160 hours to form the dibromo-bridged intermediate.
- Neutralize the reaction mixture to pH 7-8, extract with organic solvent, and crystallize the 5,11-dibromo intermediate.
- Perform Heck coupling using Pd(OAc)2, PPh3, and Cs2CO3 in DMF at 137°C with 4-vinyl-N,N-dimethylbenzylamine to yield the final fluorescent product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the synthesis route described in CN101962536A offers substantial advantages for procurement managers seeking cost reduction in fluorescent probe manufacturing. The reliance on commodity chemicals such as 4-bromoaniline and paraformaldehyde as starting materials ensures a stable and inexpensive supply chain, mitigating the risks associated with sourcing specialized biological reagents. Furthermore, the elimination of complex biological fermentation or antibody production processes drastically simplifies the manufacturing workflow, leading to significant operational efficiencies. The use of standard organic synthesis techniques, including crystallization and column chromatography, allows for straightforward scale-up from laboratory to industrial production without the need for exotic equipment or hazardous bioprocessing facilities.
- Cost Reduction in Manufacturing: The synthetic pathway leverages widely available industrial raw materials and avoids the high costs associated with biological sourcing, resulting in a fundamentally lower cost of goods sold. By utilizing a robust Heck coupling reaction with recoverable palladium catalysts, the process minimizes waste and maximizes atom economy, further driving down production expenses. Additionally, the ability to crystallize the intermediate directly from the reaction mixture reduces the need for extensive purification steps, saving both time and solvent costs. This economic efficiency makes the technology highly attractive for large-scale production of high-purity OLED material precursors or biological stains.
- Enhanced Supply Chain Reliability: Unlike antibody-based reagents which are susceptible to batch-to-batch variability and short shelf-lives, this chemically synthesized fluorescent substance offers superior stability and consistency. The synthetic route is not dependent on biological systems that can fail or fluctuate, ensuring a continuous and predictable supply of the final product. This reliability is critical for pharmaceutical and research institutions that require uninterrupted access to high-quality imaging agents for long-term studies. The robust nature of the chemical intermediates also simplifies logistics and storage, reducing the risk of spoilage during transit.
- Scalability and Environmental Compliance: The process is designed for scalability, utilizing standard reactor types and conditions that are easily replicated in commercial manufacturing plants. The use of trifluoroacetic acid and DMF, while requiring proper handling, is well-established in the fine chemical industry, allowing for effective waste management and recycling protocols. The high yields reported in the patent examples demonstrate the efficiency of the reaction, minimizing the generation of byproduct waste. This aligns with modern environmental compliance standards, making it easier for manufacturers to obtain necessary permits and maintain sustainable operations while scaling up complex polymer additives or fine chemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this lysosome-targeted fluorescent substance. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this material into existing research workflows or commercial product lines.
Q: What are the critical reaction conditions for the Heck coupling step in this synthesis?
A: The Heck coupling requires anhydrous DMF as a solvent, cesium carbonate as a base, and a temperature of 137°C under nitrogen protection for 8-10 hours to ensure high conversion and minimize side reactions.
Q: How does this small molecule compare to antibody-based lysosome stains?
A: Unlike antibodies which are expensive and prone to inactivation, this small molecule offers superior stability, lower cost, and specific lysosome targeting via the N,N-dimethylbenzylamine group without phototoxicity issues common in traditional dyes.
Q: Is this fluorescent substance suitable for two-photon microscopy?
A: Yes, the compound features a conjugated donor-acceptor system that allows for excitation in the near-infrared region (e.g., 790nm), making it highly effective for deep-tissue two-photon fluorescence imaging.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lysosome Targeted Fluorescent Substance Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis technology disclosed in CN101962536A for the field of cell imaging and diagnostics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of fluorescent intermediate delivered meets the highest standards required for sensitive biological applications. We are committed to supporting your R&D initiatives with materials that enable clear, high-contrast lysosome imaging.
We invite you to collaborate with our technical team to explore how this advanced synthesis route can be optimized for your specific requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that identifies opportunities to further streamline your supply chain and reduce overall manufacturing expenses. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments tailored to your project goals. Let us help you secure a competitive advantage with high-quality, cost-effective fluorescent reagents.
