Advanced Synthesis of Amphiphilic Aza-BODIPY Dyes for Biomedical Imaging
Advanced Synthesis of Amphiphilic Aza-BODIPY Dyes for Biomedical Imaging
The rapid evolution of optical imaging technologies has necessitated the development of fluorescent probes capable of deep tissue penetration with minimal background interference. Patent CN111171595A discloses a groundbreaking class of aza-fluoroborondipyrrole (aza-BODIPY) near-infrared fluorescent dyes that address critical limitations in current biomedical imaging agents. These novel compounds are engineered with a unique amphiphilic architecture, combining hydrophobic alkyl chains with hydrophilic polyethylene glycol segments to achieve exceptional water solubility while maintaining strong absorption in the near-infrared region. This technical breakthrough enables single-wavelength excitation for multimodal imaging, including photoacoustic and fluorescence imaging, which is pivotal for guiding photodynamic and photothermal synergistic tumor therapies. As a leading manufacturer, we recognize the immense potential of this chemistry to redefine the standards for high-purity pharmaceutical intermediates used in next-generation diagnostic and therapeutic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional near-infrared fluorescent dyes, such as squarylium, porphyrin, and phthalocyanine derivatives, have long served as the backbone for optical sensing and bio-imaging. However, these conventional molecules frequently suffer from inherent physicochemical drawbacks that limit their clinical utility. A primary deficiency is their poor water solubility, which often necessitates the use of organic co-solvents or complex encapsulation strategies that can introduce toxicity or alter biodistribution profiles in vivo. Furthermore, many existing aza-BODIPY dyes exhibit absorption and emission wavelengths below 750nm, falling short of the optimal biological window of 650-1000nm where tissue autofluorescence and light absorption by hemoglobin and water are minimized. This spectral limitation results in reduced penetration depth and lower signal-to-noise ratios, thereby compromising the sensitivity and selectivity required for precise tumor detection and monitoring.
The Novel Approach
The synthetic strategy outlined in the patent data introduces a sophisticated molecular design that overcomes these historical barriers through structural modulation and functional group engineering. By incorporating nitrogen atoms directly into the azapyrrolidine skeleton, the electron-donating effect is significantly enhanced, driving a bathochromic shift that pushes the maximum absorption wavelength to approximately 808nm. This places the dye firmly within the optimal near-infrared window for deep tissue imaging. Moreover, the innovation lies in the post-synthetic modification via click chemistry, where hydrophilic oxygen chains are grafted onto the alkyne-functionalized core. This creates an amphiphilic balance that ensures excellent biocompatibility and aqueous dispersibility without sacrificing the photothermal conversion efficiency. 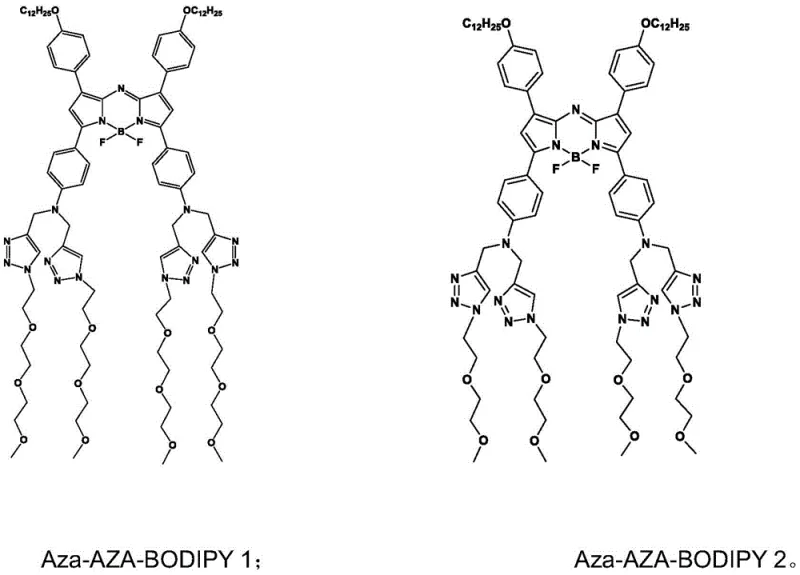
Mechanistic Insights into Multi-Step Cascade Synthesis
The construction of these advanced fluorophores relies on a robust five-step cascade that builds complexity from simple aromatic precursors with high atom economy. The process initiates with a base-catalyzed Claisen-Schmidt condensation between 4-(dodecyloxy)benzaldehyde and a propargyl-functionalized acetophenone derivative to establish the conjugated chalcone backbone. This is followed by a Michael addition with nitromethane, introducing the necessary nitrogen functionality for the subsequent heterocyclic formation. The critical ring-closure step utilizes a large excess of ammonium acetate at elevated temperatures to cyclize the intermediate into the aza-dipyrromethene core, a transformation that dictates the overall yield and purity of the final scaffold. Following this, coordination with boron trifluoride diethyl etherate rigidifies the structure, enhancing fluorescence quantum yield and photostability. 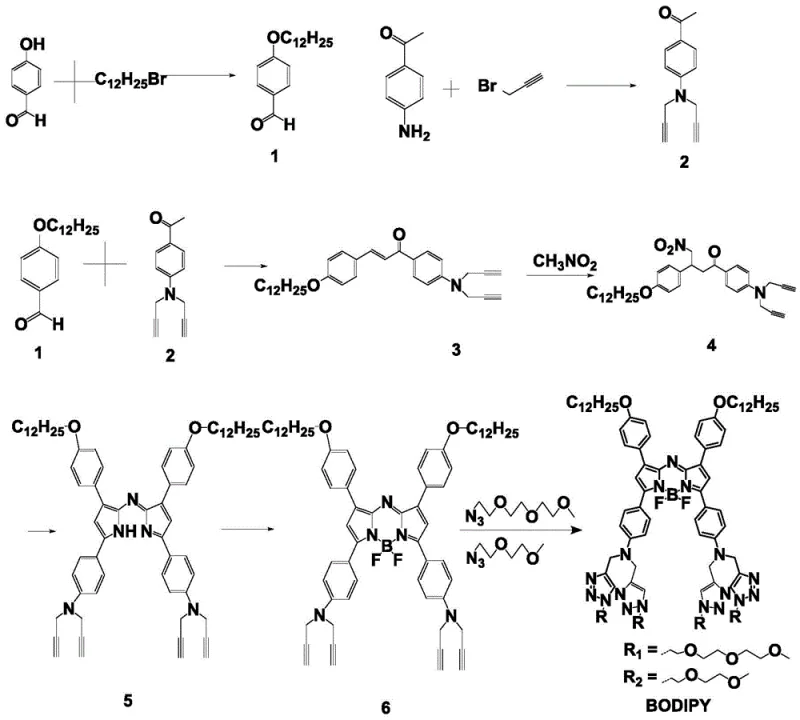
Impurity control is meticulously managed throughout the synthetic sequence, particularly during the final click chemistry stage where copper(I) iodide catalyzes the coupling of the azide-functionalized hydrophilic chains. The use of column chromatography at multiple stages, specifically after the coordination and click reaction steps, ensures the removal of metal residues and unreacted starting materials. This rigorous purification protocol is essential for pharmaceutical-grade intermediates, as trace metal contaminants can interfere with biological assays or induce cytotoxicity. The modular nature of the click reaction also allows for the tuning of the hydrophilic chain length, providing a mechanism to adjust solubility profiles dynamically to meet specific formulation requirements for different biological environments or cell types.
How to Synthesize Aza-BODIPY Efficiently
The synthesis of these high-performance near-infrared dyes requires precise control over reaction stoichiometry and temperature profiles to maximize yield and minimize byproduct formation. The patented route offers a reproducible framework that transitions smoothly from gram-scale laboratory synthesis to potential industrial production. Key parameters include maintaining a molar ratio of 1:100 for the ammonium acetate cyclization step and utilizing dry dichloromethane for the boron coordination to prevent hydrolysis of the sensitive BF2 complex. The detailed standardized synthetic steps, including specific solvent volumes, reaction times, and purification methods, are provided in the technical guide below to assist process chemists in replicating this high-value pathway.
- Perform aldol condensation between 4-(dodecyloxy)benzaldehyde and propargyl-functionalized acetophenone under alkaline conditions to form the chalcone intermediate.
- Execute a Michael addition reaction with nitromethane followed by ring-closure using ammonium acetate to construct the aza-dipyrromethene core.
- Complete the synthesis via boron coordination with BF3·Et2O and final functionalization using copper-catalyzed azide-alkyne cycloaddition (Click Chemistry) to attach hydrophilic chains.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits regarding raw material availability and process scalability. The starting materials, such as p-hydroxybenzaldehyde and bromododecane, are commodity chemicals available from multiple global suppliers, reducing the risk of supply chain bottlenecks associated with exotic or proprietary reagents. Furthermore, the reaction conditions are relatively mild, with most steps proceeding at temperatures between 25°C and 130°C, which lowers energy consumption and simplifies the engineering controls required for reactor systems. This operational simplicity translates directly into cost reduction in pharmaceutical intermediate manufacturing, as it eliminates the need for specialized high-pressure or cryogenic equipment often required for alternative dye synthesis methods.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts in the early stages of the synthesis, relying instead on abundant bases like potassium carbonate and sodium ethoxide, significantly lowers the cost of goods sold. Additionally, the high yields reported in the patent examples, particularly in the condensation and coordination steps, reduce the volume of waste solvent generated per kilogram of product. This efficiency minimizes the burden on waste treatment facilities and lowers the overall environmental compliance costs associated with large-scale production runs.
- Enhanced Supply Chain Reliability: The modular design of the molecule allows for the decoupling of the hydrophobic core synthesis from the hydrophilic tail attachment. This means that manufacturers can stockpile the stable boron-coordinated intermediate and perform the final click chemistry on demand, drastically reducing lead time for high-purity fluorescent dyes. Such flexibility ensures a continuous supply of finished goods even if there are temporary disruptions in the supply of specific azide reagents, thereby safeguarding production schedules for downstream diagnostic kit manufacturers.
- Scalability and Environmental Compliance: The process avoids the use of highly toxic heavy metals or hazardous oxidizing agents, aligning with green chemistry principles and simplifying regulatory approval for clinical use. The solvents employed, primarily ethanol and dichloromethane, are well-understood in industrial settings with established recovery and recycling protocols. This facilitates the commercial scale-up of complex pharmaceutical intermediates while maintaining a low environmental footprint, a critical factor for multinational corporations adhering to strict sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability, application, and handling of these novel aza-BODIPY derivatives. Understanding these nuances is essential for R&D teams integrating these dyes into new imaging platforms or therapeutic formulations. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers evaluating this technology for adoption.
Q: What is the primary advantage of the new Aza-BODIPY synthesis method?
A: The primary advantage is the significant improvement in water solubility and the red-shift of absorption wavelengths beyond 800nm, achieved by introducing nitrogen atoms into the pyrrole skeleton and attaching hydrophilic polyethylene glycol chains via click chemistry.
Q: What are the key reaction conditions for the cyclization step?
A: The cyclization step requires reacting the nitro-intermediate with a large excess of ammonium acetate at elevated temperatures between 80°C and 130°C for approximately 24 hours to ensure complete ring closure.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process utilizes readily available starting materials and standard organic transformations such as condensation and coordination reactions, making it highly scalable for commercial production of biomedical imaging agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aza-BODIPY Supplier
As the demand for advanced biomedical imaging agents continues to surge, partnering with an experienced CDMO is crucial for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including residual solvent analysis and heavy metal testing, which are vital for materials intended for in vivo applications. We understand the critical nature of timeline and quality in the pharmaceutical sector and are committed to delivering intermediates that meet the highest international standards.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for this new class of dyes, our experts are ready to provide tailored solutions. By leveraging our manufacturing expertise and the robust synthetic methodology described in patent CN111171595A, we can help you accelerate the development of next-generation photothermal therapies and diagnostic tools.
