Advanced Manufacturing of Cenicriviroc Intermediates via Optimized Catalytic Coupling and Purification
Advanced Manufacturing of Cenicriviroc Intermediates via Optimized Catalytic Coupling and Purification
The pharmaceutical industry continuously demands higher purity standards for complex antiviral intermediates, particularly for CCR5/CCR2 antagonists like Cenicriviroc. Patent CN112266392A introduces a groundbreaking synthetic pathway that addresses long-standing challenges in impurity control and process scalability. This disclosure outlines a method for preparing Cenicriviroc and its mesylate salt with exceptional purity profiles, specifically targeting the reduction of genotoxic impurities and difficult-to-remove dimers. By re-engineering the core carbon-carbon bond formation and amide coupling steps, the process achieves a level of chemical integrity essential for modern regulatory compliance. The following analysis details how this technology transforms the manufacturing landscape for reliable API intermediate supplier operations.
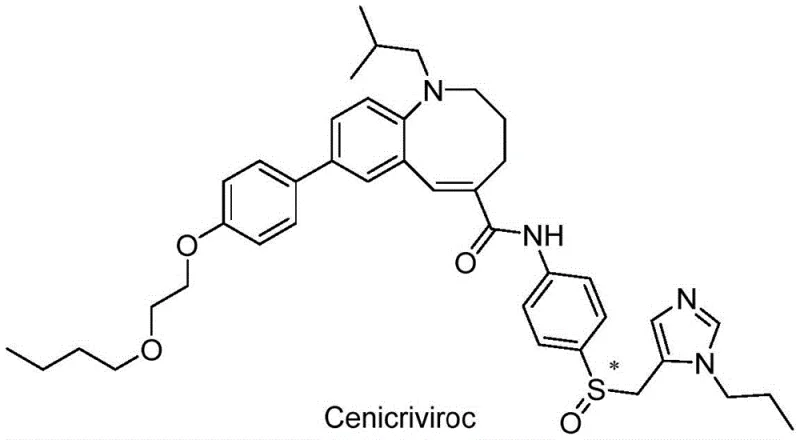
Cenicriviroc represents a structurally complex molecule featuring a tetrahydrobenzo[b]azocine core linked to a functionalized phenyl ring and a chiral sulfoxide side chain. The synthesis of such intricate architectures typically suffers from low yields and heterogeneous impurity profiles when using conventional methodologies. Previous routes often struggled with the efficient construction of the biaryl linkage and the subsequent amidation without generating significant amounts of hydrolysis byproducts. The disclosed invention provides a comprehensive solution by optimizing every stage from the initial Grignard reagent preparation to the final crystallization of the mesylate salt. This holistic approach ensures that the final product meets stringent specifications required for clinical and commercial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the key biaryl intermediate involved Suzuki coupling reactions that were plagued by inefficiencies and impurity generation. Conventional protocols often relied on palladium catalyst systems like Pd(PPh3)4 which required high loading levels to achieve acceptable conversion, yet still resulted in moderate yields ranging from 55% to 64%. A major drawback was the formation of homocoupling byproducts, specifically the biphenyl dimer impurities, which are notoriously difficult to remove during downstream purification. Furthermore, the initiation of the Grignard reagent necessary for boronic acid synthesis was inconsistent, often requiring dilute conditions and extended reflux times that hampered throughput. These inefficiencies created bottlenecks in cost reduction in pharmaceutical manufacturing and compromised the overall supply reliability.
The Novel Approach
The novel approach detailed in the patent fundamentally restructures the synthetic sequence to maximize efficiency and purity. Instead of relying on legacy catalyst systems, the process employs a highly active Pd(OAc)2/P(o-tol)3 catalyst system that operates effectively at significantly lower loadings, sometimes as low as 0.25 mol%. This shift not only improves reaction kinetics but also drastically reduces the formation of critical dimer impurities. Additionally, the Grignard formation step is optimized through thermal activation of magnesium turnings and the use of concentrated bromide solutions, eliminating the need for difficult initiators like iPrMgCl. The result is a streamlined workflow that delivers the key carboxylic acid intermediate with purity exceeding 99%, setting a new benchmark for high-purity OLED material and pharma intermediate production standards.
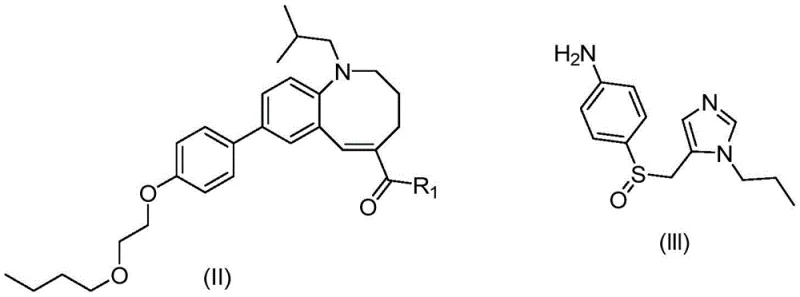
Mechanistic Insights into Pd-Catalyzed Suzuki-Miyaura Coupling
The core of this technological advancement lies in the optimized Suzuki-Miyaura coupling between the bromo-azocine acid and the boronic ester derivative. Mechanistically, the use of palladium acetate combined with tri(o-tolyl)phosphine creates a catalytic cycle that is far more robust against deactivation compared to traditional phosphine ligands. The electron-rich nature of the o-tolyl groups stabilizes the palladium center, facilitating the oxidative addition into the aryl bromide bond even under mild heating conditions below 65°C. This temperature control is critical, as higher temperatures were found to deactivate the catalyst and promote side reactions. The biphasic reaction environment, created by the careful addition of aqueous base prior to catalyst introduction, further enhances the transmetallation step while suppressing the homocoupling of the boronic species.
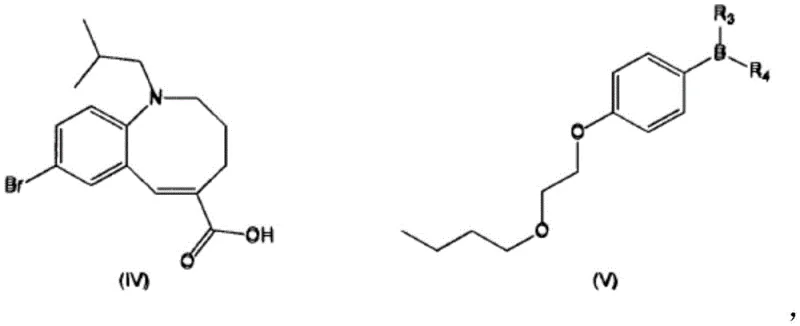
Impurity control is another pillar of this mechanistic strategy. The patent identifies several specific degradation products, such as Compound VIII (a bis-azocine dimer) and Compound IX (a butyl analog), which can arise from incomplete reactions or reagent impurities. By strictly controlling the stoichiometry of the boronic ester and maintaining an inert atmosphere through nitrogen sparging, the process minimizes these pathways. The purification protocol leverages these mechanistic insights by employing a combination of charcoal treatment and antisolvent recrystallization. This dual approach effectively scavenges residual palladium and removes lipophilic impurities, ensuring that the final intermediate possesses the requisite chemical stability for subsequent amide bond formation steps.
How to Synthesize Cenicriviroc Mesylate Efficiently
The synthesis of Cenicriviroc mesylate requires precise execution of three main stages: Grignard-mediated boronic ester formation, palladium-catalyzed cross-coupling, and final amide condensation followed by salt formation. The patent provides a detailed roadmap for navigating these steps, emphasizing the importance of moisture control and temperature regulation. Operators must pay close attention to the activation of magnesium and the rate of trimethoxyborane addition to prevent exotherms that could degrade the sensitive boronic species. Following the coupling reaction, the work-up involves careful pH adjustment and extraction to isolate the acid intermediate before converting it to the reactive acid chloride. Detailed standardized synthetic steps for this high-efficiency route are provided in the guide below.
- Initiate Grignard formation using activated magnesium and pure 1-bromo-4-(2-butoxyethoxy)benzene in THF, followed by reaction with trimethoxyborane.
- Perform Suzuki coupling between the boronic ester and 8-bromo-1-isobutyl-azocine carboxylic acid using a Pd(OAc)2/P(o-tol)3 catalyst system.
- Convert the resulting acid to an acid chloride, react with the sulfoxide aniline derivative, and crystallize the final mesylate salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this optimized process translates into tangible operational benefits beyond mere chemical yield. The reduction in catalyst loading from molar percentages to sub-molar levels directly impacts the bill of materials, offering substantial cost savings in raw material procurement. Furthermore, the robustness of the Grignard initiation step eliminates the variability often associated with large-scale organometallic reactions, thereby enhancing supply chain reliability and reducing the risk of batch failures. The simplified purification train, which avoids multiple thermal recrystallizations in favor of a streamlined charcoal and antisolvent protocol, reduces processing time and solvent consumption. These factors collectively contribute to a more resilient manufacturing infrastructure capable of meeting tight delivery schedules.
- Cost Reduction in Manufacturing: The transition to a low-loading palladium catalyst system significantly lowers the cost per kilogram of the intermediate by reducing the consumption of expensive noble metals and ligands. Additionally, the elimination of difficult-to-remove impurities reduces the burden on downstream purification, leading to higher overall recovery rates and less waste generation. This efficiency gain allows for competitive pricing strategies without compromising on quality margins.
- Enhanced Supply Chain Reliability: By solving the Grignard initiation bottleneck, the process ensures consistent batch-to-batch performance, which is critical for maintaining continuous supply lines. The use of stable reagents and mild reaction conditions minimizes the dependency on specialized equipment or extreme operating parameters, making the technology easily transferable across different manufacturing sites. This flexibility strengthens the supply network against potential disruptions.
- Scalability and Environmental Compliance: The process is designed with commercial scale-up of complex pharmaceutical intermediates in mind, utilizing common solvents like THF and ethyl acetate that are readily available in bulk. The reduced generation of heavy metal waste due to lower catalyst usage aligns with stricter environmental regulations, simplifying waste disposal protocols and lowering compliance costs. This green chemistry approach future-proofs the manufacturing asset.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis route. They are derived from the specific experimental data and embodiments disclosed in the patent documentation. Understanding these nuances is vital for R&D teams evaluating the feasibility of technology transfer. The answers reflect the optimized conditions that lead to the highest purity profiles and yields.
Q: How does the new catalyst system improve Cenicriviroc intermediate purity?
A: The patent discloses switching from Pd(PPh3)4 to a Pd(OAc)2/P(o-tol)3 system, which significantly reduces catalyst loading while minimizing critical dimer impurities like Compound VIII.
Q: What are the critical control points for preventing hydrolysis during salt formation?
A: Strict moisture control is essential. The process utilizes powdered molecular sieves to dry the organic layer before adding methanesulfonic acid, preventing hydrolysis back to the carboxylic acid.
Q: Why is the Grignard initiation step modified in this process?
A: Traditional dilute methods caused slow initiation. The optimized process uses thermal activation of magnesium and concentrated addition of the bromide to ensure rapid and complete Grignard formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cenicriviroc Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of antiviral therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex molecules like Cenicriviroc intermediates are manufactured with precision. We adhere to stringent purity specifications and operate rigorous QC labs to verify that every batch meets the highest international standards. Our commitment to quality assurance means that clients can rely on us for consistent supply of high-value pharmaceutical building blocks.
We invite you to discuss how our manufacturing capabilities can support your project goals. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency. Let us partner with you to accelerate your drug development timeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
