Advanced Phosphorylation Technology for Commercial Vidarabine Monophosphate Production
Advanced Phosphorylation Technology for Commercial Vidarabine Monophosphate Production
The pharmaceutical landscape for antiviral therapeutics continues to evolve, driven by the critical need for high-purity nucleotide analogs such as Vidarabine Monophosphate (Ara-AMP). As detailed in patent CN103204890A, a novel phosphorylation methodology has emerged that addresses longstanding challenges in the synthesis of this vital active pharmaceutical ingredient (API) intermediate. This technology leverages a refined catalytic system to transform Vidarabine into its monophosphate derivative with exceptional efficiency and reduced environmental footprint. The structural integrity of the final product, as depicted below, is paramount for its efficacy against herpes viruses and chronic viral hepatitis, making the purity profile a non-negotiable metric for downstream drug formulation.
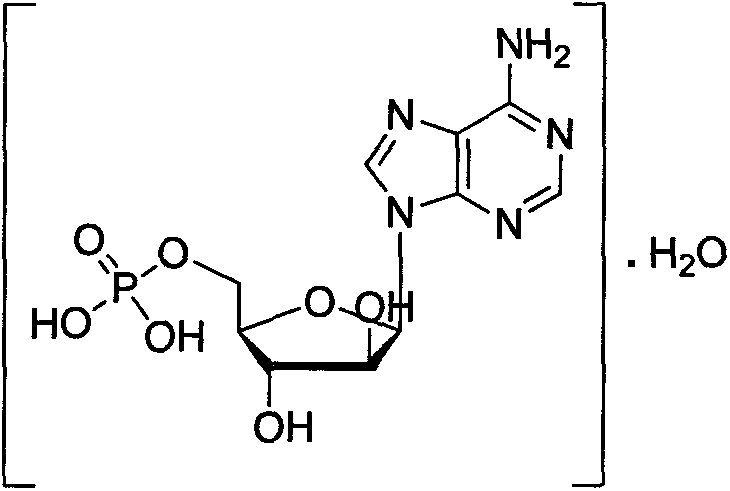
For procurement specialists and supply chain directors, understanding the underlying chemistry of patent CN103204890A is essential for securing a reliable vidarabine monophosphate supplier. The disclosed method not only optimizes yield but also streamlines the purification workflow, directly impacting the cost of goods sold (COGS) and lead times. By shifting away from hazardous solvents and cumbersome separation techniques, this process represents a significant leap forward in cost reduction in antiviral intermediate manufacturing. The ability to produce high-purity vidarabine monophosphate consistently is a key differentiator in a competitive market where regulatory compliance and patient safety are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the phosphorylation of nucleosides like vidarabine has been plagued by significant operational and environmental hurdles. Traditional protocols often relied heavily on pyridine as both a solvent and an acid scavenger, a practice that introduces severe toxicity concerns and complicates waste management. The removal of residual pyridine from the final product is notoriously difficult, requiring extensive washing and purification steps that drive up processing time and solvent consumption. Furthermore, alternative methods utilizing trialkyl phosphites or glacial acetic acid often suffer from inconsistent yields or require complex multi-step workups involving ion-exchange resins. These legacy processes are not only economically inefficient due to high solvent recovery costs but also pose substantial risks regarding operator safety and environmental compliance, making them increasingly untenable for modern commercial scale-up of complex nucleotide analogs.
The Novel Approach
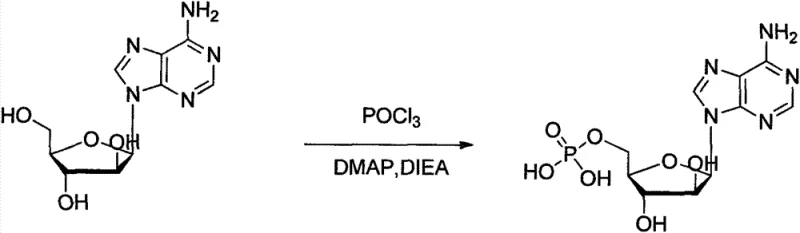
The innovative strategy outlined in the patent data circumvents these issues by employing a dichloromethane-based system catalyzed by 4-Dimethylaminopyridine (DMAP) and N,N-Diisopropylethylamine (DIEA). This approach fundamentally alters the reaction kinetics, allowing for precise control over the phosphorylation event without the need for excessive amounts of toxic pyridine. A standout feature of this novel route is the direct isolation of the intermediate phosphate salt via filtration, a process intensification step that drastically simplifies the downstream purification. By eliminating the need for liquid-liquid extractions and resin treatments, the new method enhances throughput and reduces the physical footprint required for production. This streamlined workflow is instrumental in reducing lead time for high-purity antiviral intermediates, ensuring that supply chains remain robust and responsive to market demands.
Mechanistic Insights into DMAP-Catalyzed Phosphorylation
The core of this synthetic breakthrough lies in the synergistic interaction between phosphorus oxychloride (POCl3), DMAP, and DIEA within a dichloromethane medium. DMAP acts as a potent nucleophilic catalyst, activating the phosphorylating agent to facilitate the attack on the 5'-hydroxyl group of the vidarabine sugar moiety. This activation lowers the energy barrier for the reaction, enabling it to proceed efficiently at cryogenic temperatures ranging from -10°C to 5°C. Maintaining this strict thermal window is critical; it suppresses the formation of unwanted byproducts, particularly the bis-phosphate derivative, which can arise if the reaction becomes too vigorous. The presence of DIEA serves as a non-nucleophilic base to scavenge the hydrochloric acid generated during the reaction, preventing the degradation of the acid-sensitive glycosidic bond and ensuring the structural stability of the nucleoside backbone throughout the transformation.
Furthermore, the mechanism includes a unique precipitation phenomenon that aids in impurity control. As the reaction progresses, the formed phosphate salts exhibit reduced solubility in the reaction matrix, prompting them to precipitate directly from the solution. This "salting out" effect effectively removes the product from the reactive environment before over-phosphorylation can occur, acting as a self-limiting mechanism for purity enhancement. Following the reaction, the solid is dissolved in heated water (60-90°C) and treated with activated carbon, which adsorbs colored impurities and residual organic contaminants. The subsequent recrystallization from water at 0-5°C ensures that the final high-purity vidarabine monophosphate meets stringent pharmacopeial standards, free from the heavy metal residues or solvent traces often associated with less refined synthetic routes.
How to Synthesize Vidarabine Monophosphate Efficiently
The execution of this phosphorylation protocol requires meticulous attention to temperature control and reagent addition rates to maximize yield and safety. The process begins with the dissolution of pharmaceutical-grade vidarabine in anhydrous dichloromethane under an inert nitrogen atmosphere to prevent moisture interference. The addition of the catalytic system (DMAP and DIEA) prior to cooling ensures a homogeneous mixture before the exothermic phosphorylation begins. Operators must adhere strictly to the specified temperature range of -10°C to 5°C during the dropwise addition of phosphorus oxychloride, as deviations can compromise the selectivity of the mono-phosphorylation. The detailed standardized synthetic steps, including specific molar ratios and stirring times, are provided in the technical guide below to ensure reproducibility across different manufacturing scales.
- Dissolve vidarabine in dichloromethane under nitrogen protection, add DIEA and DMAP catalyst, and cool the mixture to between -10°C and 5°C.
- Slowly add a solution of phosphorus oxychloride in dichloromethane dropwise while maintaining low temperature, then stir for 2 to 4 hours.
- Filter the resulting solid, dissolve in hot water (60-90°C), decolorize with activated carbon, and crystallize by cooling to 0-5°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented methodology offers profound benefits that extend beyond simple chemical yield. By replacing pyridine with a DIEA/DMAP system, manufacturers eliminate the substantial costs associated with the recovery and disposal of large volumes of toxic solvents. This shift not only aligns with green chemistry principles but also results in significant operational savings, as the energy-intensive distillation processes required for pyridine recycling are rendered unnecessary. Additionally, the simplified work-up procedure, which relies on direct filtration rather than complex extraction sequences, reduces the man-hours and equipment utilization time per batch. These efficiencies translate directly into a more competitive pricing structure for the final API intermediate, providing a clear advantage in cost reduction in antiviral intermediate manufacturing.
- Cost Reduction in Manufacturing: The elimination of pyridine removes a major cost driver related to solvent purchase, recovery, and hazardous waste treatment. The use of dichloromethane, which is easier to recover and recycle in standard pharmaceutical setups, further optimizes the variable costs of production. Moreover, the high crude yield (approximately 89-92%) and total recovery (around 75-81%) minimize raw material wastage, ensuring that the expensive starting material, vidarabine, is utilized with maximum efficiency. This economic model supports a sustainable pricing strategy that can withstand market fluctuations in raw material costs.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply lines to downstream API manufacturers. The simplicity of the purification steps reduces the risk of batch failures due to operational errors or equipment malfunctions during complex separations. By shortening the overall cycle time from reaction to dried product, suppliers can respond more rapidly to urgent procurement requests, thereby enhancing the reliability of the supply chain. This agility is particularly valuable in the antiviral sector, where demand can spike unexpectedly during outbreak scenarios.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind. The direct filtration step avoids the emulsion issues often encountered during large-scale liquid-liquid extractions, facilitating a smooth transition from pilot plant to multi-ton commercial production. Furthermore, the reduced toxicity profile of the reagents simplifies compliance with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions and worker exposure limits. This forward-looking approach future-proofs the manufacturing asset against regulatory tightening, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of vidarabine monophosphate using this advanced phosphorylation technique. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions regarding vendor qualification and process validation.
Q: What are the primary advantages of the DMAP/DIEA catalytic system over traditional pyridine methods?
A: The DMAP/DIEA system significantly reduces toxicity and environmental impact compared to pyridine-based methods. It eliminates the need for difficult pyridine recovery processes and lowers production costs while maintaining high yields.
Q: How does the new process control the formation of bis-phosphate impurities?
A: The process utilizes strict temperature control between -10°C and 5°C during the addition of phosphorus oxychloride. This kinetic control, combined with the specific catalytic action of DMAP, minimizes over-phosphorylation.
Q: Is this synthesis method suitable for large-scale industrial manufacturing?
A: Yes, the method is designed for industrial suitability. It features a simplified work-up procedure involving direct filtration of the intermediate phosphate salt, which avoids complex extraction steps and facilitates scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vidarabine Monophosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antiviral medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in patent CN103204890A is executed with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and potency of every batch. Our commitment to excellence guarantees that the vidarabine monophosphate we supply meets the exacting standards required for global pharmaceutical registration and clinical use.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages inherent in this low-toxicity process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless integration of our high-performance intermediates into your manufacturing pipeline.
