Advanced Chemical Synthesis of NMN for Commercial Scale-up and Purity Control
The global demand for Nicotinamide Mononucleotide (NMN) has surged due to its recognized potential in anti-aging and metabolic health applications, driving an urgent need for robust manufacturing technologies. Patent CN112961197A discloses a groundbreaking chemical synthesis method that addresses the critical bottlenecks of traditional production routes. This technical insight report analyzes the proprietary process which involves reacting ribofuranose derivatives with nicotinamide amide, followed by metaphosphate coupling and rigorous purification. Unlike biological methods, this chemical pathway offers a deterministic approach to molecular construction, ensuring that every batch meets stringent quality specifications required by top-tier pharmaceutical partners. For R&D directors and procurement leaders, understanding this shift from enzymatic to chemical synthesis is vital for securing a reliable NMN supplier capable of delivering consistent, high-purity intermediates at a sustainable cost structure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the large-scale synthesis of nicotinamide mononucleotide has been dominated by enzymatic reactions and fermentation technologies, both of which present significant industrial challenges. Enzymatic methods often suffer from high operational costs due to the expense of biocatalysts and require harsh reaction conditions that can destabilize the sensitive nucleotide structure. Furthermore, biological processes are inherently variable, leading to large differences in product indexes between batches, which complicates quality control and regulatory compliance. Fermentation methods introduce additional risks, such as the potential presence of excessive endotoxins and the complexities associated with transgenic technology, creating hidden dangers for the final application of the product in human health supplements or pharmaceuticals. These limitations severely restrict the ability to achieve cost reduction in pharmaceutical intermediate manufacturing and hinder the reliable supply of high-purity NMN.
The Novel Approach
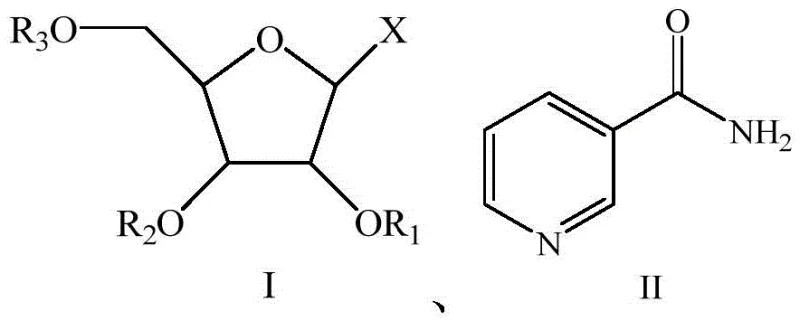
The chemical synthesis method outlined in the patent represents a paradigm shift by utilizing small molecule organic synthesis to construct the NMN backbone with precision. This novel approach leverages the reaction between a protected ribofuranose derivative (Formula I) and nicotinamide (Formula II) under controlled acidic conditions. By replacing biological catalysts with chemical reagents like hydrobromic acid and sodium alkoxide, the process achieves mild, stable, and easily-controlled reaction conditions that are scalable from laboratory to industrial volumes. The use of defined chemical starting materials ensures approximate product indexes across different batches, significantly enhancing reaction productivity and supply chain reliability. This method effectively bypasses the biological variability of fermentation, offering a pathway for the commercial scale-up of complex pharmaceutical intermediates with superior safety profiles.
Mechanistic Insights into Chemical Glycosylation and Phosphorylation
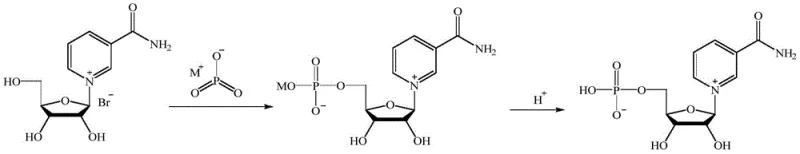
The core of this synthesis lies in the precise activation of the sugar moiety and its subsequent coupling with the nicotinamide base. The process initiates with the treatment of the ribofuranose derivative with hydrobromic acid, which activates the anomeric center for nucleophilic attack. This step is critical for establishing the beta-glycosidic bond, which defines the biological activity of the resulting NMN. The reaction conditions are meticulously optimized, with temperatures maintained between 45-60°C and reaction times of 4-8 hours, to ensure complete conversion while minimizing side reactions. The use of an acetic acid solution of hydrobromic acid provides a buffered acidic environment that facilitates the formation of the oxocarbenium ion intermediate, which is then trapped by the nitrogen of the nicotinamide ring. This mechanistic control is essential for R&D directors focusing on the purity and impurity profile of the final API intermediate.
Following the glycosylation, the synthesis proceeds to the phosphorylation step, which installs the crucial phosphate group at the 5'-position of the ribose sugar. This transformation is achieved by reacting the deprotected nucleoside intermediate with metaphosphate under alkaline conditions (pH 8-10) at 30-50°C. The control of pH is paramount here, as it dictates the nucleophilicity of the hydroxyl group and the stability of the phosphate ester bond. The subsequent acidification to pH 3-5 quenches the reaction and prepares the mixture for purification. The patent details a sophisticated purification sequence involving protein membrane filtration and anion exchange resin separation, which effectively removes unreacted metaphosphate and inorganic salts. This rigorous downstream processing ensures that the final product meets the stringent purity specifications required for high-purity pharmaceutical intermediates, eliminating the need for expensive chromatographic separations often required in less optimized routes.
How to Synthesize Nicotinamide Mononucleotide Efficiently
The synthesis of NMN via this chemical route requires strict adherence to the reaction parameters defined in the patent to ensure high yield and purity. The process involves a sequence of activation, coupling, deprotection, and phosphorylation steps, each requiring precise control of temperature, pH, and stoichiometry. For technical teams looking to implement this route, it is essential to focus on the quality of the starting ribofuranose derivatives and the efficiency of the membrane filtration steps. The detailed standardized synthesis steps provided below outline the specific operational conditions, including solvent choices like chloroform or dichloromethane and specific reagent concentrations, which are critical for reproducing the 47-48% overall yield reported in the examples. This guide serves as a foundational reference for scaling this technology from 100 kgs to 100 MT annual commercial production.
- Activate the ribofuranose derivative by reacting it with 55-65% hydrobromic acid aqueous solution at room temperature for 2-5 hours to form the first mixture.
- Couple the activated sugar with nicotinamide (Formula II) using an acetic acid solution of hydrobromic acid at 45-60°C for 4-8 hours to form the nucleoside intermediate.
- Deprotect the intermediate using sodium alkoxide, followed by phosphorylation with metaphosphate at pH 8-10 and 30-50°C, then purify via membrane filtration and resin separation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this chemical synthesis method offers substantial strategic benefits beyond mere technical feasibility. The elimination of expensive enzymes and the reliance on commodity chemical reagents significantly reduces the raw material cost base, allowing for more competitive pricing in the global market. The stability of the chemical process means that production schedules are not subject to the biological variabilities that often plague fermentation facilities, thereby reducing lead time for high-purity pharmaceutical intermediates. Furthermore, the simplified purification workflow, which avoids complex biological separations, streamlines the manufacturing timeline and reduces the overall operational expenditure. These factors combine to create a supply chain that is more resilient, cost-effective, and capable of meeting the surging global demand for NMN without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The chemical route eliminates the need for costly biocatalysts and the specialized infrastructure required for sterile fermentation, leading to a drastic simplification of the production process. By utilizing standard chemical reactors and reagents, manufacturers can achieve substantial cost savings in capital expenditure and operational maintenance. The removal of transition metal catalysts or expensive enzymes also simplifies the downstream purification, reducing the consumption of chromatography resins and solvents. This economic efficiency translates directly into a more attractive cost structure for buyers seeking long-term supply agreements for bulk NMN ingredients.
- Enhanced Supply Chain Reliability: Chemical synthesis offers a level of predictability that biological methods cannot match, ensuring consistent output regardless of seasonal or biological fluctuations. The raw materials, such as nicotinamide and protected ribose derivatives, are widely available commodity chemicals, mitigating the risk of supply chain disruptions associated with specialized biological feedstocks. This reliability allows supply chain planners to forecast production volumes with greater accuracy, ensuring continuous availability of the product for downstream formulation partners. The robust nature of the process also means that technology transfer between manufacturing sites is more straightforward, further securing the supply continuity for global clients.
- Scalability and Environmental Compliance: The process is designed for easy scale-up, moving from laboratory benchtop to multi-ton production without the complex re-optimization often required for bioprocesses. The use of membrane filtration and resin separation aligns with modern green chemistry principles by reducing solvent waste and energy consumption compared to traditional extraction methods. The absence of genetically modified organisms (GMOs) simplifies regulatory compliance in markets with strict biosafety laws, expanding the potential market reach for the product. This environmental and regulatory advantage positions the chemical synthesis route as a sustainable long-term solution for the industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the chemical synthesis of NMN, based on the specific advantages and mechanisms detailed in the patent data. These insights are curated to assist decision-makers in evaluating the feasibility and benefits of adopting this manufacturing technology. Understanding these nuances is crucial for aligning procurement strategies with the technical capabilities of potential suppliers.
Q: What are the primary advantages of this chemical synthesis method over enzymatic methods?
A: The chemical synthesis method described in patent CN112961197A offers significantly lower raw material costs and more stable, easily-controlled process conditions compared to enzymatic reactions. It ensures approximate product indexes across different batches, thereby improving reaction productivity and consistency for large-scale manufacturing.
Q: How does this process address safety concerns associated with fermentation methods?
A: Unlike fermentation methods which may involve sensitive transgenic technology and risks of excessive endotoxins, this chemical route eliminates biological contaminants. The resulting product is high in safety, removing hidden dangers associated with biological expression systems and improving the overall application prospect of the NMN product.
Q: What purification techniques are employed to ensure high purity NMN?
A: The process utilizes a multi-stage purification strategy including protein membrane filtration, high-pressure desalting membrane concentration, and anion exchange resin separation. This rigorous approach, followed by crystallization, ensures the removal of impurities and salts to achieve high-purity NMN suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nicotinamide Mononucleotide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the complexities of nucleotide synthesis and is equipped to implement the advanced chemical routes described in patent CN112961197A with precision. We maintain stringent purity specifications through our rigorous QC labs, ensuring that every batch of NMN meets the highest standards for pharmaceutical and nutraceutical applications. Our commitment to quality and scalability makes us the ideal partner for companies seeking to secure a stable supply of this critical anti-aging ingredient.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for NMN and related intermediates. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies can translate into value for your organization. We encourage potential partners to contact us for specific COA data and route feasibility assessments to verify our capability to meet your unique project requirements. Let us collaborate to drive innovation and efficiency in your product development pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
