Advanced Synthesis of Methylmercapto-Substituted Spiroisoxazole-Pyrrolizine Derivatives for Oncology
The pharmaceutical landscape is continuously evolving with the demand for novel heterocyclic scaffolds that exhibit potent biological activity, particularly in the realm of oncology. Patent CN111943998A introduces a groundbreaking class of methylmercapto-substituted arabinose triazole structure spiroisoxazole-pyrrolizine derivatives, representing a significant leap forward in the design of antitumor agents. This technology leverages a sophisticated multi-step synthetic strategy to fuse three distinct pharmacophores: the pyrrolizine alkaloid core, the bioactive isoxazole ring, and a glycosylated triazole moiety. The resulting compounds, specifically exemplified by Compound 6, demonstrate remarkable inhibitory effects against various human tumor cell lines, with particularly strong activity against lung adenocarcinoma (A549) cells. For research and development teams seeking reliable pharmaceutical intermediates, this patent offers a robust pathway to accessing high-value chemical space that was previously difficult to navigate efficiently.
The strategic integration of these structural elements addresses critical challenges in modern drug discovery, such as improving metabolic stability and enhancing target binding affinity. By utilizing a 1,3-dipolar cycloaddition reaction, the synthesis achieves high stereocontrol over the spiro-center, which is often a bottleneck in traditional methods. This level of precision is essential for developing candidates that can proceed through rigorous preclinical testing. Furthermore, the inclusion of the arabinose sugar unit is not merely structural decoration; it serves a functional purpose in modulating the physicochemical properties of the molecule, thereby improving its drug-likeness profile. As a leading specialty chemical manufacturer, understanding these nuanced synthetic routes allows us to support clients in accelerating their pipeline from benchtop discovery to clinical evaluation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of spiroisoxazole frameworks has been fraught with synthetic challenges, often relying on harsh reaction conditions that compromise sensitive functional groups or result in poor regioselectivity. Traditional approaches to forming the isoxazole ring frequently involve the use of unstable nitrile oxides generated in situ under vigorous conditions, which can lead to dimerization side reactions or decomposition of the substrate. Moreover, introducing complex sugar moieties onto such crowded heterocyclic systems typically requires extensive protection and deprotection sequences that drastically reduce overall yield and increase waste generation. These inefficiencies translate directly into higher costs and longer lead times for API manufacturing, making it difficult for pharmaceutical companies to secure a consistent supply of high-purity intermediates. Additionally, conventional methods often struggle to control the stereochemistry at the spiro-junction, leading to mixtures of diastereomers that are difficult and expensive to separate, ultimately hindering the biological evaluation of the pure active species.
The Novel Approach
The methodology disclosed in CN111943998A circumvents these historical bottlenecks by employing a mild and highly efficient 1,3-dipolar cycloaddition strategy mediated by Chloramine T. This innovative approach allows for the gentle generation of the nitrile oxide dipole directly from the corresponding oxime precursor in the presence of the dipolarophile, ensuring immediate capture and cyclization. The reaction proceeds smoothly in ethanol under reflux, eliminating the need for toxic organic solvents or cryogenic conditions typically associated with nitrile oxide chemistry. This streamlined process not only improves the overall atom economy but also simplifies the purification workflow, as the desired spiro-compound can often be isolated via simple recrystallization. 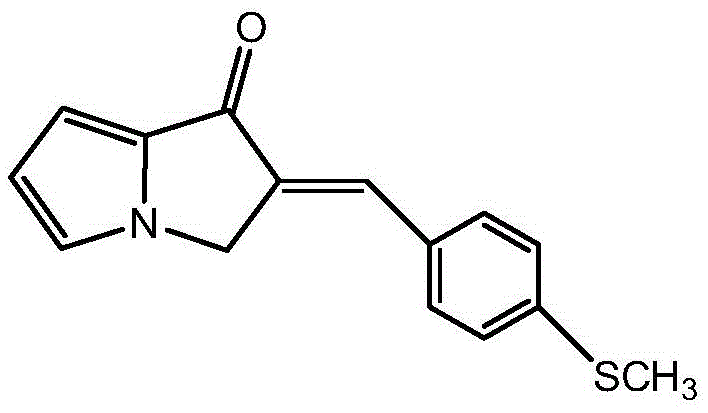 Furthermore, the sequential introduction of the arabinose-triazole unit prior to the final deprotection step ensures that the sensitive glycosidic linkage remains intact throughout the rigorous cyclization process. This novel route represents a paradigm shift in cost reduction in pharmaceutical intermediates manufacturing, offering a scalable solution that maintains high fidelity to the desired molecular architecture while minimizing operational complexity.
Furthermore, the sequential introduction of the arabinose-triazole unit prior to the final deprotection step ensures that the sensitive glycosidic linkage remains intact throughout the rigorous cyclization process. This novel route represents a paradigm shift in cost reduction in pharmaceutical intermediates manufacturing, offering a scalable solution that maintains high fidelity to the desired molecular architecture while minimizing operational complexity.
Mechanistic Insights into Chloramine T-Mediated 1,3-Dipolar Cycloaddition
The core of this synthetic innovation lies in the mechanistic elegance of the Chloramine T-mediated oxidation of the aldoxime to a nitrile oxide, which subsequently undergoes a concerted [3+2] cycloaddition with the exocyclic double bond of the pyrrolizine ketone. In this transformation, Chloramine T acts as a mild oxidant, facilitating the dehydration of the hydroxylamine derivative to generate the reactive 1,3-dipole without over-oxidizing other sensitive functionalities such as the thioether or the triazole ring. The electron-deficient nature of the exocyclic alkene in the pyrrolizine scaffold makes it an ideal dipolarophile, driving the reaction towards the formation of the five-membered isoxazole ring with excellent regioselectivity. 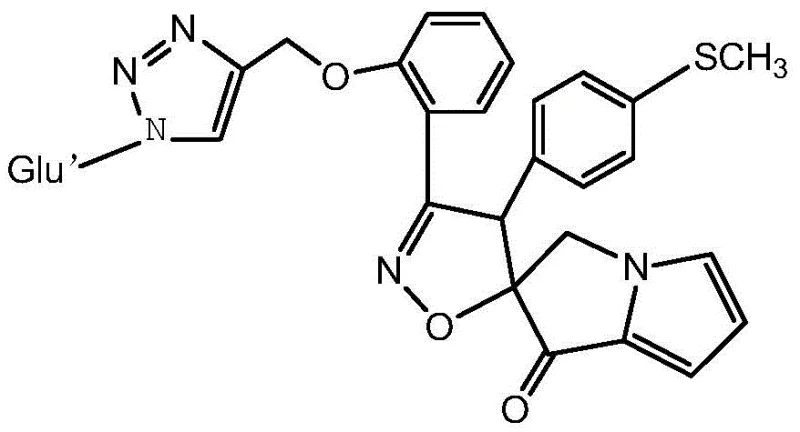 The transition state of this cycloaddition is stabilized by the adjacent carbonyl group, which lowers the LUMO energy of the dipolarophile, thereby accelerating the reaction kinetics under thermal conditions. This mechanistic understanding is crucial for R&D directors aiming to optimize reaction parameters for commercial scale-up of complex pharmaceutical intermediates, as it highlights the robustness of the chemistry against minor fluctuations in temperature or stoichiometry. Furthermore, the stereoelectronic effects imposed by the bulky arabinose-triazole substituent guide the approach of the dipole, ensuring the formation of the specific spiro-diastereomer required for optimal biological activity.
The transition state of this cycloaddition is stabilized by the adjacent carbonyl group, which lowers the LUMO energy of the dipolarophile, thereby accelerating the reaction kinetics under thermal conditions. This mechanistic understanding is crucial for R&D directors aiming to optimize reaction parameters for commercial scale-up of complex pharmaceutical intermediates, as it highlights the robustness of the chemistry against minor fluctuations in temperature or stoichiometry. Furthermore, the stereoelectronic effects imposed by the bulky arabinose-triazole substituent guide the approach of the dipole, ensuring the formation of the specific spiro-diastereomer required for optimal biological activity.
Beyond the cyclization event, the strategic incorporation of the arabinose moiety plays a pivotal role in the final pharmacological profile of the derivative. The sugar unit, as depicted in the structural analysis, introduces multiple hydroxyl groups capable of forming extensive hydrogen bond networks with aqueous solvent molecules and biological targets. 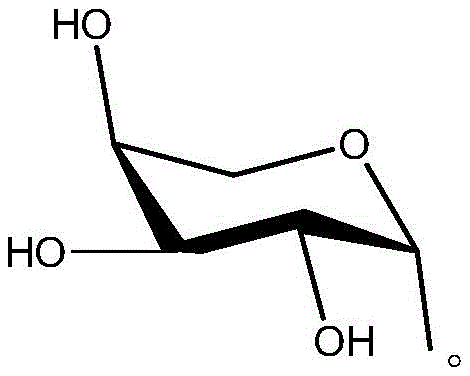 This modification effectively mitigates the inherent lipophilicity of the polycyclic aromatic core, thereby enhancing the aqueous solubility which is often a limiting factor in the oral bioavailability of kinase inhibitors and similar anticancer agents. From an impurity control perspective, the final deprotection step using sodium methoxide is highly selective for the ester groups on the sugar, leaving the robust spiro-isoxazole and triazole linkages untouched. This orthogonality in reactivity ensures that the final product is free from partially deprotected intermediates, simplifying the purification process and ensuring that the high-purity pharmaceutical intermediates delivered to downstream users meet stringent regulatory specifications for residual solvents and related substances.
This modification effectively mitigates the inherent lipophilicity of the polycyclic aromatic core, thereby enhancing the aqueous solubility which is often a limiting factor in the oral bioavailability of kinase inhibitors and similar anticancer agents. From an impurity control perspective, the final deprotection step using sodium methoxide is highly selective for the ester groups on the sugar, leaving the robust spiro-isoxazole and triazole linkages untouched. This orthogonality in reactivity ensures that the final product is free from partially deprotected intermediates, simplifying the purification process and ensuring that the high-purity pharmaceutical intermediates delivered to downstream users meet stringent regulatory specifications for residual solvents and related substances.
How to Synthesize Methylmercapto-Substituted Spiroisoxazole-Pyrrolizine Derivatives Efficiently
The synthesis of these potent antitumor agents follows a logical four-step sequence that balances chemical complexity with operational simplicity, making it amenable to both laboratory scale optimization and industrial production. The process begins with the condensation of readily available starting materials to form the key pyrrolizine intermediate, followed by the preparation of the glycosylated oxime dipole precursor. The convergence of these two fragments via the 1,3-dipolar cycloaddition constructs the complex spiro-core in a single operation, demonstrating high step economy. Finally, a mild basic hydrolysis reveals the free hydroxyl groups on the sugar, yielding the biologically active final product. For detailed operational parameters, stoichiometry, and safety considerations, please refer to the standardized protocol below which encapsulates the critical process controls identified in the patent literature.
- Synthesize (E)-2-(4-methylmercaptobenzylidene)-2,3-dihydropyrrolizin-1-one via condensation of 2,3-dihydropyrrolizin-1-one and 4-methylmercaptobenzaldehyde in ethanol with NaOH.
- Prepare acetylarabinose triazole salicylaldoxime by reacting acetylarabinose triazole salicylaldehyde with hydroxylamine hydrochloride.
- Perform 1,3-dipolar cycloaddition using Chloramine T in refluxing ethanol to form the spiroisoxazole core structure.
- Execute deprotection using sodium methoxide in methanol at low temperature to remove acetyl groups and yield the final hydroxy-substituted product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical novelty, directly impacting the bottom line through improved process efficiency and resource utilization. The reliance on commodity chemicals such as ethanol, methanol, and sodium hydroxide reduces the dependency on exotic or controlled reagents that often suffer from supply volatility and price spikes. Moreover, the elimination of transition metal catalysts in the key cyclization step removes the need for expensive and time-consuming metal scavenging processes, which are a significant cost driver in the production of fine chemical intermediates. This simplification of the downstream processing train allows for faster batch turnover times and reduced equipment occupancy, effectively increasing the throughput of existing manufacturing facilities without the need for capital-intensive expansion. The robustness of the chemistry also translates to higher first-pass yields, minimizing the generation of off-spec material and reducing the environmental footprint associated with waste disposal and solvent recovery.
- Cost Reduction in Manufacturing: The process utilizes a telescoped approach where possible and avoids the use of precious metal catalysts, which significantly lowers the raw material cost per kilogram of the final active ingredient. By employing Chloramine T as a stoichiometric oxidant rather than a catalytic system requiring ligands and inert atmospheres, the operational expenditure is drastically simplified. The workup procedures involve standard filtration and crystallization techniques that do not require specialized chromatography equipment at large scales, further driving down the cost of goods sold (COGS). This economic efficiency makes the derivative a viable candidate for cost-sensitive therapeutic areas while maintaining high margins for the supplier.
- Enhanced Supply Chain Reliability: The starting materials, including 4-methylmercaptobenzaldehyde and 2,3-dihydropyrrolizin-1-one, are commercially available from multiple global vendors, mitigating the risk of single-source supply disruptions. The synthetic route is insensitive to moisture and oxygen during the key cyclization step, allowing for flexible scheduling and reduced requirement for strictly anhydrous conditions which can be a bottleneck in large-scale reactors. This resilience ensures reducing lead time for high-purity pharmaceutical intermediates, enabling just-in-time delivery models that align with the dynamic needs of pharmaceutical clients. The stability of the intermediates also allows for strategic stockpiling, providing a buffer against market fluctuations.
- Scalability and Environmental Compliance: The use of green solvents like ethanol and methanol aligns with modern sustainability goals and regulatory pressures to reduce the use of chlorinated hydrocarbons. The reaction generates minimal hazardous waste, and the byproducts are largely benign salts that are easily treated in standard wastewater facilities. The scalability of the 1,3-dipolar cycloaddition has been proven to maintain selectivity even upon increasing batch sizes, ensuring that the quality profile established at the gram scale is preserved at the metric ton level. This consistency is vital for maintaining regulatory compliance and ensuring uninterrupted supply for clinical and commercial programs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these spiroisoxazole-pyrrolizine derivatives. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, providing a transparent view of the technology's capabilities and limitations. Understanding these aspects helps stakeholders make informed decisions regarding the integration of this chemistry into their broader drug discovery or manufacturing strategies.
Q: What is the key advantage of the 1,3-dipolar cycloaddition method used in this synthesis?
A: The 1,3-dipolar cycloaddition utilizing Chloramine T allows for the mild and highly regioselective formation of the complex spiroisoxazole ring system without requiring harsh acidic conditions or expensive transition metal catalysts, ensuring high purity and simplified downstream processing.
Q: How does the introduction of the arabinose moiety affect the pharmacological properties?
A: Incorporating the arabinose sugar unit significantly enhances the water solubility and bioavailability of the lipophilic spiroisoxazole-pyrrolizine scaffold, facilitating better interaction with biological targets such as enzymes and receptors through hydrogen bonding.
Q: What represents the rate-limiting step in the commercial scale-up of this derivative?
A: The deprotection step involving sodium methoxide requires precise temperature control (-5°C to 0°C) to prevent side reactions; however, the use of common solvents like methanol and simple ion-exchange resin workups makes this step highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methylmercapto-Substituted Spiroisoxazole-Pyrrolizine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative academic research into reliable commercial reality. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising antitumor activity observed in the laboratory can be harnessed for patient benefit. We are committed to delivering stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the structural integrity and stereochemical purity of every batch. Our state-of-the-art facilities are equipped to handle the specific requirements of heterocyclic chemistry, including the safe handling of energetic intermediates and the precise temperature control needed for the deprotection steps described in this patent.
We invite pharmaceutical partners to collaborate with us to unlock the full potential of this chemical series. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can accelerate your project timelines and reduce overall development risks. Together, we can advance the next generation of oncology therapeutics from concept to clinic.
