Optimized Anhydrous Synthesis of Bis(2-dimethylaminoethyl) Ether for Industrial Polyurethane Applications
The global demand for high-performance polyurethane catalysts continues to drive innovation in fine chemical synthesis, specifically for tertiary amine ethers like bis(2-dimethylaminoethyl) ether. Patent CN102786427B introduces a transformative anhydrous methodology that addresses longstanding purity and yield challenges associated with conventional etherification routes. This technical breakthrough leverages a unique combination of thionyl chloride chlorination and metallic sodium activation to create a completely water-free reaction environment. By fundamentally altering the neutralization mechanism, the process prevents the degradation of sensitive chloro-intermediates that typically plagues aqueous-based syntheses. The resulting product achieves exceptional purity levels exceeding 99%, making it an ideal candidate for demanding polymer applications where trace impurities can compromise foam stability.
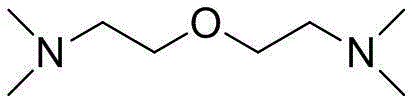
Furthermore, the strategic use of the starting amine as both reactant and solvent represents a significant leap towards green chemistry principles in industrial manufacturing. This approach not only streamlines the operational workflow by removing solvent recovery steps but also drastically reduces the volume of hazardous waste generated during production. For R&D teams focused on scaling complex amine derivatives, this patent offers a robust framework for achieving consistent batch-to-batch quality. The integration of quaternary ammonium catalysts further refines the kinetic profile of the reaction, ensuring that commercial scale-up can be achieved with predictable efficiency and minimal energy consumption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bis(2-dimethylaminoethyl) ether has been hindered by reliance on processes such as those described in US3084675, which utilize toluene as a solvent and aqueous sodium hydroxide for neutralization. These legacy methods suffer from inherent thermodynamic and kinetic inefficiencies, primarily due to the introduction of water into the reaction system. The presence of water during the neutralization of the chloro-intermediate leads to significant hydrolysis, converting valuable reactants into useless alcohol by-products and drastically lowering overall yield. Additionally, the use of toluene necessitates complex distillation and recovery systems to meet environmental regulations, adding substantial capital and operational expenditures to the manufacturing process.
Moreover, conventional approaches often require harsh reaction conditions or extended reaction times to drive the etherification to completion, which can promote the formation of unwanted side products and degrade product quality. The separation of inorganic salts, such as sodium chloride, from organic phases in aqueous systems is frequently inefficient, leading to product contamination and requiring additional purification steps like extensive washing or recrystallization. These cumulative inefficiencies result in a process that is not only economically burdensome due to low atom economy but also environmentally unsustainable due to the generation of large volumes of saline wastewater. For procurement managers, these factors translate into higher raw material costs and unpredictable supply chain volatility caused by waste disposal constraints.
The Novel Approach
In stark contrast, the novel methodology outlined in CN102786427B eliminates water from the entire synthetic sequence, thereby preserving the integrity of the reactive chloro-species. By generating sodium alkoxide in situ using metallic sodium and excess 2-dimethylethanolamine, the process creates a strongly nucleophilic environment without introducing protons that could trigger hydrolysis. The chloride hydrochloride salt is neutralized directly by the alkoxide, producing sodium chloride which precipitates out of the organic matrix due to its poor solubility in the amine solvent. This elegant design allows for simple mechanical filtration to remove inorganic by-products, bypassing the need for aqueous extraction entirely.
The introduction of quaternary ammonium salts as catalysts further distinguishes this approach, enabling the etherification to proceed rapidly at moderate temperatures between 50°C and 90°C. This catalytic enhancement reduces the thermal load on the reactor and minimizes the risk of thermal decomposition of the product. The use of excess starting amine as the reaction medium removes the necessity for external volatile organic compounds, aligning the process with modern sustainability goals. For supply chain heads, this translates to a simplified logistics profile with fewer hazardous materials to transport and store, while R&D directors benefit from a cleaner impurity profile that facilitates easier regulatory approval for downstream applications.
Mechanistic Insights into Anhydrous Etherification Catalyzed by Quaternary Ammonium Salts
The core of this synthesis lies in the precise control of nucleophilic substitution dynamics within an anhydrous organic phase. The initial chlorination step utilizes thionyl chloride to convert the hydroxyl group of 2-dimethylethanolamine into a good leaving group, forming the corresponding chloride hydrochloride salt. Unlike reactions using hydrochloric acid, thionyl chloride decomposes into gaseous by-products (SO2 and HCl) which are easily removed, driving the equilibrium forward and ensuring high conversion. The subsequent formation of the sodium alkoxide involves the direct reaction of metallic sodium with the alcohol functionality, releasing hydrogen gas and generating a potent alkoxide nucleophile that is free from hydration shells.
During the critical etherification stage, the quaternary ammonium catalyst plays a pivotal role in facilitating the interaction between the ionic alkoxide species and the organic chloride substrate. Although both species are in the same organic phase (the excess amine), the catalyst likely aids in disrupting ion pairing or stabilizing the transition state, thereby lowering the activation energy required for the SN2 displacement. This mechanistic advantage allows the reaction to achieve high yields, reported in the range of 66% to 78% in specific embodiments, without the need for extreme temperatures. The absence of water ensures that the chloride intermediate remains stable until it encounters the alkoxide, maximizing the efficiency of the carbon-oxygen bond formation.
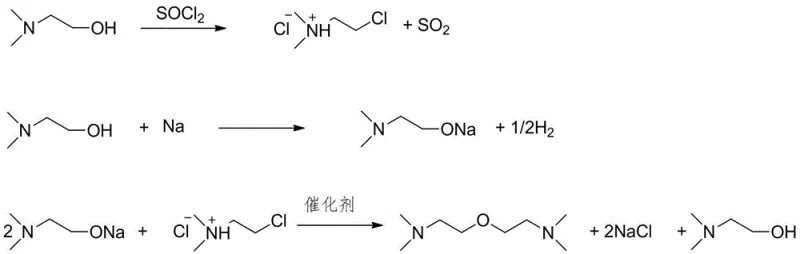
Impurity control is inherently built into this mechanism through the physical properties of the by-products. Since sodium chloride is insoluble in the reaction medium, it precipitates immediately upon formation, effectively removing itself from the equilibrium and preventing reverse reactions or side interactions. Any unreacted starting material can be easily recovered via vacuum distillation due to the significant boiling point difference between the amine alcohol and the final ether product. This high degree of selectivity ensures that the final distillate contains minimal structural analogs or degradation products, which is critical for applications in polyurethane foaming where catalyst consistency dictates cell structure uniformity. The rigorous exclusion of moisture throughout the process chain guarantees that the final specification meets the stringent requirements of high-end industrial users.
How to Synthesize Bis(2-dimethylaminoethyl) Ether Efficiently
Implementing this synthesis route requires careful attention to moisture exclusion and temperature control during the alkoxide formation stage. The process begins with the dropwise addition of 2-dimethylethanolamine to thionyl chloride at low temperatures to manage exothermicity, followed by the removal of volatiles under reduced pressure to isolate the dry chloride salt. Subsequently, metallic sodium is reacted with a molar excess of the amine alcohol under reflux to generate the alkoxide solution, which is then cooled prior to the addition of the chloride salt.
- Chlorinate 2-dimethylethanolamine with thionyl chloride at -10 to 50°C to form the chloride hydrochloride salt.
- React excess 2-dimethylethanolamine with metallic sodium at 80 to 140°C to generate the sodium alkoxide solution.
- Combine the chloride salt with the alkoxide solution in the presence of a quaternary ammonium catalyst at 50 to 90°C, followed by vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this anhydrous synthesis route offers profound advantages in cost structure and operational reliability compared to traditional aqueous methods. The elimination of toluene and other organic solvents removes a significant line item from the raw material budget and eradicates the costs associated with solvent recovery and loss. Furthermore, the simplified workup procedure, which relies on filtration rather than complex aqueous extractions and washes, reduces labor hours and utility consumption per batch. For procurement managers, this means a more stable cost base that is less susceptible to fluctuations in solvent markets and waste disposal fees.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing the starting material as the solvent, thereby negating the need to purchase, store, and recover large volumes of external organic solvents. The high atom economy and improved yield resulting from the prevention of hydrolysis directly lower the effective cost per kilogram of the final active ingredient. Additionally, the ability to filter off solid sodium chloride simplifies the purification train, reducing energy consumption associated with distillation and wastewater treatment. These cumulative efficiencies result in a leaner manufacturing process with substantially lower variable costs.
- Enhanced Supply Chain Reliability: By removing dependence on aqueous neutralization agents and complex solvent systems, the supply chain becomes more resilient and easier to manage. The raw materials required, specifically thionyl chloride and metallic sodium, are commodity chemicals with robust global availability, ensuring consistent feedstock supply. The simplified process flow reduces the number of unit operations, decreasing the likelihood of equipment failure or bottlenecks that could disrupt production schedules. This reliability allows for more accurate lead time forecasting and strengthens the ability to meet just-in-time delivery commitments for downstream polymer manufacturers.
- Scalability and Environmental Compliance: The anhydrous nature of the reaction significantly reduces the volume of hazardous wastewater generated, easing the burden on effluent treatment plants and ensuring compliance with increasingly strict environmental regulations. The process is inherently scalable because the heat management and mixing requirements are less demanding than those for heterogeneous aqueous-organic systems. The use of vacuum distillation for purification is a standard unit operation that scales linearly from pilot plant to commercial production, facilitating rapid capacity expansion without the need for specialized or exotic equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this specific synthetic pathway. Understanding these details is crucial for evaluating the feasibility of adopting this technology for large-scale production.
Q: How does this method prevent hydrolysis of the chloro-intermediate?
A: Unlike traditional methods using aqueous NaOH, this process utilizes sodium alkoxide generated from metallic sodium in an anhydrous environment. The neutralization of the chloride hydrochloride occurs without water generation, effectively eliminating the risk of hydrolyzing the sensitive 2-dimethylaminoethyl chloride intermediate.
Q: What is the role of the quaternary ammonium salt in this synthesis?
A: The quaternary ammonium salt acts as a phase-transfer-like catalyst that significantly accelerates the etherification reaction rate. It allows the reaction to proceed efficiently at lower temperatures (50-90°C) compared to uncatalyzed thermal methods, thereby improving overall yield and selectivity.
Q: Why is excess 2-dimethylethanolamine used in the reaction?
A: Excess 2-dimethylethanolamine serves a dual purpose: it acts as the reactant for sodium alkoxide formation and simultaneously functions as the reaction solvent. This eliminates the need for external organic solvents like toluene, simplifying downstream recovery and reducing environmental waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis(2-dimethylaminoethyl) Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity catalysts play in the performance of advanced polyurethane materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102786427B are fully realized in practical manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of bis(2-dimethylaminoethyl) ether meets the exacting standards required for sensitive catalytic applications. Our commitment to quality assurance ensures that your downstream processes remain stable and efficient.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific application needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and product performance.
