Scalable Synthesis of N-3-Isoxazole Carbamic Acid Tert-Butyl Ester for Global Pharma Supply Chains
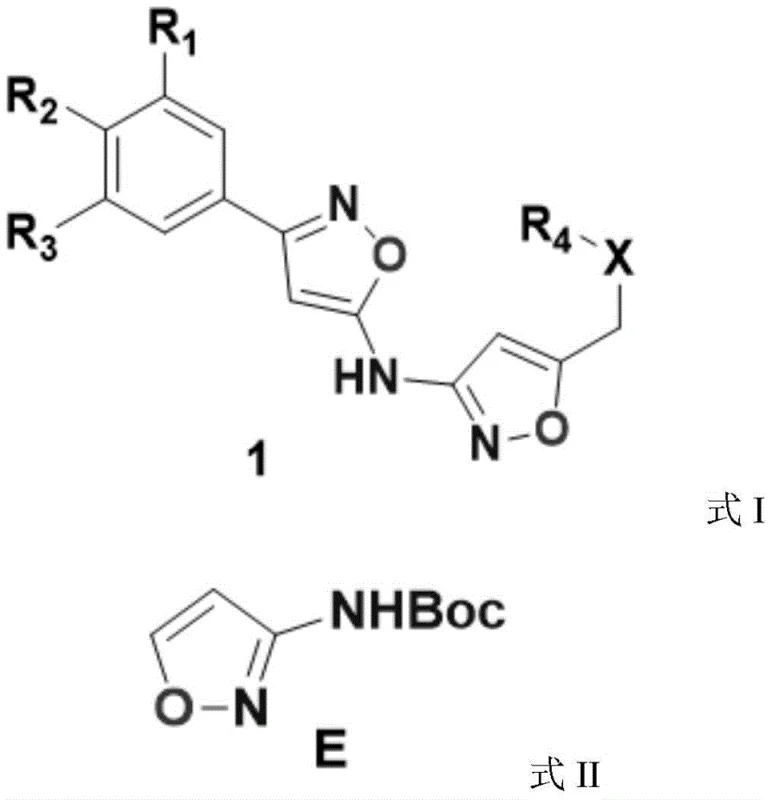
The pharmaceutical industry continuously seeks robust pathways for constructing heterocyclic scaffolds that serve as critical building blocks for novel therapeutic agents. Patent CN109369553B introduces a highly efficient method for synthesizing N-3-isoxazole carbamic acid tert-butyl ester, a pivotal intermediate used in the development of anti-infective active molecules. This specific chemical architecture allows for the strategic introduction of aminoisoxazole structures into complex drug candidates, a capability notably utilized by major pharmaceutical entities such as AstraZeneca AB in their proprietary pipelines. The significance of this patent lies not merely in the chemical transformation itself, but in its deliberate engineering for industrial applicability, addressing the long-standing gap between laboratory feasibility and commercial viability. By leveraging readily available starting materials and optimizing reaction conditions for safety and yield, this technology offers a reliable pharmaceutical intermediate supplier pathway that aligns with modern Good Manufacturing Practice (GMP) standards. The structural versatility of the resulting isoxazole derivative enables medicinal chemists to explore new chemical space with confidence in the supply chain's ability to deliver consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-aminoisoxazole derivatives has been plagued by methodologies that are difficult to translate from benchtop experiments to manufacturing floors. Prior art, such as the processes described in US 2003207899A1, often relies on conditions that are technically feasible on a gram scale but become hazardous or economically prohibitive when scaled up. Traditional routes frequently involve expensive reagents, harsh reaction environments that compromise safety, or purification steps that result in significant material loss, thereby inflating the cost of goods sold. Furthermore, many legacy methods lack rigorous control over impurity profiles, leading to batch-to-batch variability that is unacceptable for regulated pharmaceutical production. The reliance on specialized catalysts or solvents that are difficult to recover also contributes to environmental burdens and operational complexity. These limitations create bottlenecks in the supply chain, causing delays in drug development timelines and increasing the financial risk for procurement teams managing tight budgets. Consequently, there has been a persistent demand for a synthesis route that balances chemical efficiency with economic and operational practicality.
The Novel Approach
The methodology disclosed in CN109369553B represents a paradigm shift by prioritizing simplicity and scalability without sacrificing chemical integrity. This novel approach utilizes acrylonitrile and hydroxyurea as primary feedstocks, both of which are commodity chemicals with stable global supply chains, ensuring cost reduction in pharmaceutical manufacturing. The process eliminates the need for exotic catalysts or extreme conditions, instead employing a straightforward three-step sequence involving bromination, cyclization, and protection. By optimizing the reaction parameters, such as maintaining specific low-temperature ranges during exothermic steps, the process minimizes side reactions and maximizes the yield of the desired intermediate. The workup procedures are designed for ease of execution, utilizing common solvents like ethyl acetate and dichloromethane which can be efficiently recovered and recycled. This streamlined workflow not only reduces the operational footprint but also enhances the overall throughput, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates. The result is a robust manufacturing protocol that delivers high-purity products consistently, meeting the stringent requirements of downstream drug synthesis.
Mechanistic Insights into Bromination and Cyclization Strategy
The core of this synthesis lies in the precise control of electrophilic addition and subsequent heterocyclic ring formation. The initial step involves the reaction of acrylonitrile with liquid bromine, where temperature regulation between 10°C and 20°C is critical to manage the exothermic nature of the bromination. This careful thermal management prevents the formation of poly-brominated byproducts and ensures the selective generation of the dibromo intermediate. Following this, the introduction of hydroxyurea in an alkaline aqueous medium facilitates a nucleophilic attack that drives the cyclization process. The mechanism proceeds through the formation of an oxime intermediate which subsequently undergoes intramolecular condensation to close the isoxazole ring. The use of sodium hydroxide not only acts as a base to deprotonate the hydroxyurea but also helps in neutralizing the hydrogen bromide generated during the reaction, driving the equilibrium towards product formation. This mechanistic pathway is inherently cleaner than alternative methods, as it avoids the use of heavy metal catalysts that often leave toxic residues requiring extensive purification. The result is a reaction profile that is both chemically elegant and industrially pragmatic, ensuring high conversion rates with minimal waste generation.
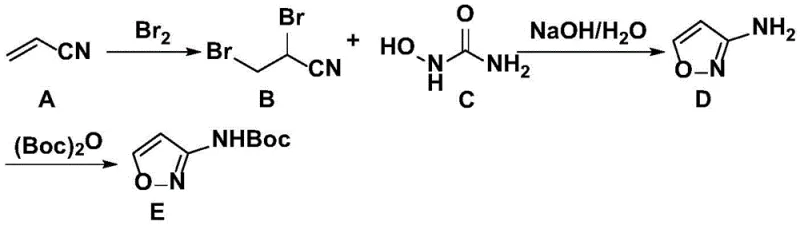
Impurity control is further enhanced in the final protection step, where the crude 3-aminoisoxazole is reacted with di-tert-butyl dicarbonate. The presence of 4-dimethylaminopyridine (DMAP) acts as a nucleophilic catalyst, accelerating the formation of the carbamate bond while operating under mild conditions of 5-10°C. This low-temperature environment is crucial for suppressing potential decomposition of the sensitive isoxazole ring and preventing the formation of urea-based side products. The subsequent purification strategy involves a series of acidic and basic washes followed by recrystallization, which effectively removes unreacted starting materials and organic impurities. By adjusting the pH to 8-9 during the workup, insoluble salts are filtered off, and the organic phase is dried thoroughly to prevent hydrolysis during concentration. This rigorous attention to detail in the mechanistic execution ensures that the final N-3-isoxazole carbamic acid tert-butyl ester meets high-purity specifications required for API synthesis. The process demonstrates how fundamental chemical principles, when applied with precision, can solve complex manufacturing challenges and deliver superior product quality.
How to Synthesize N-3-Isoxazole Carbamic Acid Tert-Butyl Ester Efficiently
Implementing this synthesis route requires adherence to specific operational protocols to maximize yield and safety. The process begins with the careful handling of liquid bromine, necessitating appropriate corrosion-resistant equipment and ventilation systems. Operators must monitor the dropwise addition of acrylonitrile closely to maintain the exotherm within the specified 10-20°C window, ensuring the reaction mixture remains homogeneous. Detailed standardized synthetic steps see the guide below for exact parameters regarding stoichiometry and agitation speeds. Following the bromination, the transfer of the reaction mixture to the alkaline hydroxyurea solution must be performed slowly to control gas evolution and heat generation. The final isolation involves precise solvent swaps and temperature-controlled crystallization to obtain the product as a white solid. Adhering to these procedural nuances guarantees reproducibility and aligns with the quality expectations of a reliable pharmaceutical intermediate supplier.
- Bromination of acrylonitrile with liquid bromine at controlled low temperatures (10-20°C) to form the dibromo intermediate.
- Cyclization reaction with hydroxyurea in aqueous NaOH solution, maintaining temperature below 20°C to yield 3-aminoisoxazole.
- Protection of the amine group using di-tert-butyl dicarbonate (Boc2O) in dichloromethane with DMAP catalyst to finalize the ester.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial value by addressing key pain points in the chemical supply chain. The reliance on commodity raw materials such as acrylonitrile and hydroxyurea significantly mitigates the risk of supply disruptions caused by the scarcity of specialized reagents. This accessibility translates directly into enhanced supply chain reliability, allowing procurement managers to secure long-term contracts with greater confidence in continuity. Furthermore, the elimination of expensive transition metal catalysts removes the need for costly scavenging steps and heavy metal testing, leading to substantial cost savings in the overall manufacturing budget. The simplified operational workflow reduces the burden on production facilities, enabling faster turnaround times and reducing lead time for high-purity pharmaceutical intermediates. By minimizing the number of unit operations and utilizing standard solvents, the process also lowers the environmental compliance burden, facilitating smoother regulatory approvals and audits. These factors combine to create a compelling economic case for adopting this technology in large-scale production environments.
- Cost Reduction in Manufacturing: The utilization of inexpensive, bulk-available starting materials drastically lowers the direct material costs associated with production. By avoiding the use of precious metal catalysts, the process eliminates the significant expense related to catalyst procurement and the subsequent removal of metal residues. The high yields achieved in each step minimize waste disposal costs and maximize the output per batch, contributing to a more favorable cost structure. Additionally, the ability to recycle solvents like dichloromethane and ethyl acetate further enhances the economic efficiency of the operation. These cumulative effects result in a highly competitive pricing model that supports margin improvement for downstream drug manufacturers without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent production output, which is critical for maintaining uninterrupted supply to pharmaceutical clients. The use of stable raw materials reduces the volatility associated with sourcing niche chemicals, thereby stabilizing the supply chain against market fluctuations. The scalability of the process means that production volumes can be ramped up quickly to meet surges in demand without requiring significant capital investment in new equipment. This flexibility provides a strategic advantage in managing inventory levels and responding to dynamic market requirements. Ultimately, the reliability of this manufacturing method fosters stronger partnerships between suppliers and buyers, built on a foundation of trust and dependability.
- Scalability and Environmental Compliance: The process is inherently designed for scale, with reaction conditions that are easily managed in large reactors using standard industrial control systems. The absence of hazardous reagents and the generation of manageable waste streams simplify the environmental permitting process and reduce the ecological footprint of the facility. Efficient workup procedures minimize solvent consumption and energy usage, aligning with green chemistry principles and corporate sustainability goals. The straightforward purification steps ensure that the final product meets strict regulatory limits for impurities, facilitating smoother registration filings. This alignment with environmental and safety standards future-proofs the supply chain against tightening global regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this isoxazole intermediate. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation. They serve to clarify the operational advantages and quality assurances associated with this synthesis method. Understanding these details helps stakeholders make informed decisions regarding the integration of this intermediate into their development pipelines.
Q: What are the critical temperature controls for the bromination step?
A: The bromination of acrylonitrile requires strict temperature control between 10°C and 20°C to prevent over-bromination and ensure safety during the exothermic reaction.
Q: How is the purity of the final isoxazole ester ensured?
A: Purity is maintained through precise pH adjustment during workup, multiple extraction cycles with ethyl acetate, and final recrystallization using petroleum ether.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent explicitly demonstrates scalability from laboratory to multi-kilogram batches using inexpensive, readily available raw materials like acrylonitrile and hydroxyurea.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-3-Isoxazole Carbamic Acid Tert-Butyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. We are committed to delivering products that adhere to stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques for comprehensive characterization. Our infrastructure is designed to handle complex chemistries safely, providing a secure environment for the manufacture of sensitive pharmaceutical building blocks. By partnering with us, you gain access to a supply chain that is both resilient and responsive to the evolving demands of the global healthcare market.
We invite you to engage with our technical procurement team to discuss your specific requirements and explore how our capabilities can support your objectives. Request a Customized Cost-Saving Analysis to understand the economic benefits of switching to our optimized synthesis route. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our commitment to quality and transparency. Let us collaborate to accelerate your drug development timeline with a supply partner you can trust.
