Scalable Synthesis of 3-(2-Aminophenyl)-2-Acrylate: A Cost-Effective Route for Pharmaceutical Manufacturing
Introduction to Advanced Synthetic Methodologies
The pharmaceutical industry constantly seeks robust, scalable, and cost-efficient pathways for critical intermediates, and the recent disclosure in patent CN113880724A offers a compelling solution for the synthesis of 3-(2-aminophenyl)-2-acrylate. This compound serves as a vital building block in the construction of various bioactive molecules, particularly within the cardiovascular therapeutic class. The disclosed methodology represents a significant departure from traditional synthetic strategies, addressing long-standing pain points regarding purity, waste generation, and operational complexity. By leveraging a sequential three-step protocol involving Knoevenagel condensation, salt-mediated decarboxylation, and metal-mediated reduction, this process achieves high chemical purity while utilizing readily available commodity chemicals. For R&D directors and process chemists, this patent provides a blueprint for optimizing the manufacturing of nitro-substituted cinnamate derivatives, ensuring that the final active pharmaceutical ingredient (API) precursors meet stringent quality standards without the burden of complex purification protocols associated with older technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha,beta-unsaturated esters like the target molecule has frequently relied upon the Wittig reaction, a classic carbon-carbon bond-forming transformation. However, as illustrated in the background art of the patent, this conventional approach suffers from inherent inefficiencies that are unacceptable in modern green chemistry and large-scale manufacturing contexts. The primary drawback lies in the generation of stoichiometric quantities of triphenylphosphine oxide (TPPO) as a byproduct. TPPO is a high-melting solid that is notoriously difficult to separate from the desired product, often requiring extensive chromatography or multiple recrystallizations, which drastically reduces overall yield and increases solvent consumption. Furthermore, the phosphonium ylides used in Wittig reactions are moisture-sensitive and require stringent anhydrous conditions, adding to the operational cost and complexity. The low atom economy of this route means that a significant portion of the reactant mass ends up as waste, creating substantial environmental disposal challenges and inflating the cost of goods sold (COGS) for the final intermediate.

The Novel Approach
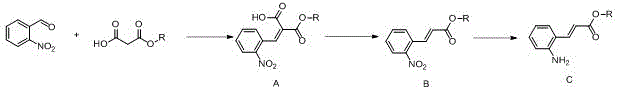
In stark contrast, the novel methodology presented in CN113880724A circumvents these issues by employing a Knoevenagel condensation followed by a specialized decarboxylation sequence. This strategy utilizes o-nitrobenzaldehyde and monomethyl malonate as starting materials, both of which are inexpensive and commercially abundant commodity chemicals. The initial condensation step proceeds under mild basic conditions using organic amines such as piperidine or pyridine, avoiding the need for strong, hazardous bases or cryogenic temperatures. The subsequent decarboxylation is elegantly facilitated by the presence of lithium salts, which promote the loss of carbon dioxide to form the mono-ester with high selectivity. Finally, the reduction of the nitro group is achieved using iron powder and acid, a method that is far more economical and safer to operate on a multi-ton scale compared to catalytic hydrogenation. This holistic approach not only improves the impurity profile by eliminating phosphine residues but also streamlines the workup procedures, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations aiming for cost reduction in API manufacturing.
Mechanistic Insights into Knoevenagel Condensation and Salt-Mediated Decarboxylation
The success of this synthetic route hinges on the precise control of reaction mechanisms, particularly during the formation of the alpha,beta-unsaturated system. The initial step involves a base-catalyzed Knoevenagel condensation where the active methylene group of monomethyl malonate is deprotonated by the organic base, such as piperidine, to form a nucleophilic enamine or carbanion species. This nucleophile attacks the electrophilic carbonyl carbon of o-nitrobenzaldehyde, followed by dehydration to yield the dicarbonyl intermediate (Compound A). The choice of solvent, typically an aprotic solvent like toluene or xylene, is critical here to facilitate the removal of water via azeotropic distillation, driving the equilibrium towards product formation. The electron-withdrawing nature of the ortho-nitro group enhances the electrophilicity of the aldehyde, ensuring rapid reaction kinetics even at moderate temperatures of 20-30°C, which minimizes side reactions such as polymerization or self-condensation of the aldehyde.
Following the condensation, the transformation of Compound A to Compound B involves a fascinating salt-mediated decarboxylation mechanism that is central to this patent's innovation. In the presence of lithium chloride or lithium bromide within a polar aprotic solvent system like DMSO or DMF containing water, the beta-keto acid moiety undergoes thermal decarboxylation. The lithium cation likely coordinates with the carbonyl oxygens, stabilizing the transition state and lowering the activation energy required for the loss of carbon dioxide. This step is remarkably efficient, converting the diester directly into the desired mono-ester without affecting the sensitive alkene geometry or the nitro group. The use of specific lithium salts is key; other alkali metal salts may not provide the same level of rate acceleration or selectivity. This mechanistic nuance ensures that the process yields high-purity 3-(2-nitrophenyl)-2-acrylate (Compound B), setting the stage for the final reduction step with minimal impurity carryover, which is essential for producing high-purity OLED material or pharmaceutical precursors.
How to Synthesize 3-(2-Aminophenyl)-2-Acrylate Efficiently
To implement this synthesis effectively in a pilot or production plant, operators must adhere to specific parameters regarding reagent ratios and temperature controls to maximize yield and safety. The process begins with the condensation of o-nitrobenzaldehyde and monomethyl malonate in toluene with piperidine, maintaining temperatures between 20-30°C to prevent exotherms. Following isolation of the intermediate, the decarboxylation is performed in DMSO/water with LiCl at 100°C, a step that requires careful monitoring to ensure complete gas evolution. The final reduction utilizes iron powder in ethyl acetate/water with hydrochloric acid, a heterogeneous reaction that benefits from vigorous stirring to maintain contact between the solid metal and the organic phase. Detailed standard operating procedures (SOPs) derived from the patent examples suggest that strict control of the acid addition rate during reduction is necessary to manage hydrogen evolution and heat generation, ensuring a safe and reproducible process suitable for commercial scale-up of complex intermediates.
- Perform Knoevenagel condensation between o-nitrobenzaldehyde and monomethyl malonate using an organic base like piperidine in toluene.
- Execute decarboxylation of the intermediate using lithium chloride or lithium bromide in a polar aprotic solvent like DMSO or DMF with water.
- Conduct nitro reduction using iron powder and hydrochloric acid in ethyl acetate or ethanol to yield the final amine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers transformative advantages that directly impact the bottom line and operational resilience. The elimination of triphenylphosphine and its associated oxide byproduct removes a major cost driver and waste disposal liability, leading to substantial cost savings in raw material procurement and effluent treatment. Furthermore, the reliance on commodity chemicals such as iron powder, hydrochloric acid, and simple organic solvents like ethyl acetate and toluene ensures a stable and secure supply chain, mitigating the risks associated with sourcing specialized or regulated reagents. This robustness allows for reducing lead time for high-purity pharmaceutical intermediates, as the raw materials are universally available and not subject to the geopolitical or logistical constraints often faced by exotic catalysts. The process stability also implies fewer batch failures and less downtime for cleaning, enhancing overall equipment effectiveness (OEE) in manufacturing facilities.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of expensive phosphorus-based reagents with inexpensive malonate derivatives and the use of iron powder instead of noble metal catalysts like palladium or platinum. By avoiding the generation of triphenylphosphine oxide, the downstream purification costs are drastically simplified, as there is no need for complex chromatographic separations or excessive solvent usage to remove this stubborn impurity. Additionally, the high atom economy of the Knoevenagel-decarboxylation sequence means that a greater proportion of the input mass is converted into valuable product, reducing the cost per kilogram of the final API intermediate significantly. These factors combine to create a leaner manufacturing process that is highly competitive in price-sensitive markets.
- Enhanced Supply Chain Reliability: The supply chain security for this route is exceptionally high due to the use of bulk commodity chemicals that are produced globally in massive quantities. Reagents such as o-nitrobenzaldehyde, monomethyl malonate, and lithium chloride are standard inventory items for most chemical distributors, ensuring that production schedules are not disrupted by raw material shortages. Moreover, the process does not rely on single-source proprietary catalysts or enzymes, giving procurement managers the flexibility to source materials from multiple vendors to negotiate better pricing and terms. This diversification of supply sources strengthens the resilience of the manufacturing operation against market volatility and logistical bottlenecks, ensuring consistent delivery to downstream customers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to multi-ton production is straightforward due to the absence of hazardous high-pressure hydrogenation steps or pyrophoric reagents. The iron-mediated reduction operates at atmospheric pressure and moderate temperatures, reducing the capital expenditure required for specialized high-pressure reactors and safety systems. From an environmental standpoint, the process generates less hazardous waste; the iron sludge produced can often be recycled or disposed of more easily than heavy metal catalyst waste, and the avoidance of phosphine oxides reduces the organic load in wastewater. This alignment with green chemistry principles facilitates easier regulatory approval and permits, accelerating the time-to-market for new drug formulations relying on this intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims within CN113880724A, providing clarity on process robustness and product quality. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for their specific supply chain needs. By addressing these key areas, stakeholders can make informed decisions about integrating this technology into their existing manufacturing portfolios to achieve better efficiency and compliance.
Q: Why is the Knoevenagel route preferred over the Wittig reaction for this intermediate?
A: The traditional Wittig reaction generates stoichiometric amounts of triphenylphosphine oxide (TPPO), which is notoriously difficult to remove and lowers atom economy. The patented Knoevenagel route avoids phosphine reagents entirely, simplifying purification and significantly reducing waste disposal costs.
Q: What role do lithium salts play in the decarboxylation step?
A: Lithium chloride or lithium bromide acts as a crucial catalyst/promoter in the decarboxylation of the alpha,beta-unsaturated dicarbonyl intermediate. This salt-mediated process allows the reaction to proceed efficiently in polar aprotic solvents like DMSO or DMF at moderate temperatures, ensuring high conversion rates without requiring harsh acidic or basic conditions that might degrade the sensitive double bond.
Q: Is the iron powder reduction method suitable for large-scale production?
A: Yes, the use of iron powder and hydrochloric acid is highly advantageous for industrial scale-up. Unlike catalytic hydrogenation which requires high-pressure equipment and expensive noble metal catalysts, the iron-mediated reduction operates at atmospheric pressure with inexpensive reagents. The resulting iron sludge is easily filtered off, making the process robust, safe, and economically viable for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(2-Aminophenyl)-2-Acrylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for complex pharmaceutical intermediates like 3-(2-aminophenyl)-2-acrylate. Our team of expert process chemists has extensively analyzed the pathway described in CN113880724A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to ensure every batch complies with international pharmacopeia standards. Our facility is equipped to handle the specific solvent systems and reaction conditions, such as the lithium salt-mediated decarboxylation and iron reductions, ensuring consistent quality and supply continuity for your global operations.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through this advanced synthetic route. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how switching to this method can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for high-quality fine chemical intermediates in the global market.
