Advanced Synthesis of 2-Substituent-4-Bromophenol for High-Volume Pharmaceutical Manufacturing
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for safer, more efficient, and highly selective synthetic routes. A significant breakthrough in this domain is documented in patent CN115724721A, which details a novel preparation method for 2-substituent-4-bromophenol, a critical intermediate used in the synthesis of complexes, specialty polymers, and catalysts. Traditional methods for brominating phenols often struggle with regioselectivity, frequently yielding unwanted ortho-substituted or polybrominated by-products that complicate downstream purification and inflate production costs. This new technology addresses these challenges by utilizing N-bromosuccinimide (NBS) in specific polar organic solvents, creating a reaction environment that dramatically enhances both conversion rates and target selectivity. For R&D directors and procurement managers seeking reliable fine chemical intermediates supplier partnerships, understanding this shift towards milder, high-selectivity chemistry is essential for securing long-term supply chain stability and cost efficiency.
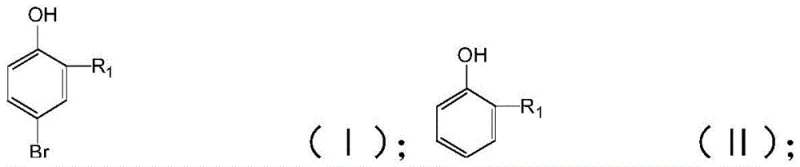
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the bromination of phenolic compounds has relied heavily on the use of molecular bromine (Br2), often in the presence of Lewis acid catalysts or in non-polar solvents. While effective in introducing a bromine atom, this classical approach suffers from significant drawbacks regarding safety and selectivity. Molecular bromine is highly corrosive, toxic, and difficult to handle on a large industrial scale, requiring specialized equipment and rigorous safety protocols that drive up capital expenditure. Furthermore, the high reactivity of molecular bromine often leads to uncontrolled polybromination, generating substantial amounts of 2-substituent-4,6-dibromophenol and other impurities. These side reactions not only reduce the overall yield of the desired 4-bromo isomer but also create a complex impurity profile that necessitates energy-intensive purification steps, such as multiple recrystallizations or column chromatography, which are impractical for commercial scale-up of complex fine chemical intermediates.
The Novel Approach
In stark contrast, the method disclosed in patent CN115724721A utilizes N-bromosuccinimide (NBS) as the brominating agent within a carefully selected polar organic solvent system. This approach fundamentally changes the reaction kinetics, allowing for a much milder process that operates effectively at temperatures ranging from -30°C to 60°C. By optimizing the solvent polarity to be greater than 5.0, specifically using solvents like acetonitrile or methanol, the process achieves a remarkable raw material conversion rate of up to 99% and a target product selectivity exceeding 96%. This high level of control minimizes the formation of the problematic 2-substituent-6-bromophenol and dibrominated by-products. For procurement teams focused on cost reduction in fine chemical intermediates manufacturing, this translates to a streamlined workflow where the crude reaction mixture requires less aggressive purification, directly lowering operational expenses and reducing the environmental footprint associated with waste solvent disposal.
Mechanistic Insights into Regioselective NBS Bromination
The success of this synthesis lies in the intricate interplay between the electronic effects of the phenolic hydroxyl group and the steric hindrance provided by the 2-position substituent, all modulated by the solvent environment. The phenolic hydroxyl group is a strong activating group that directs electrophilic substitution to the ortho and para positions. However, the presence of a substituent at the 2-position sterically hinders the adjacent ortho position, theoretically favoring the para position. Despite this, conventional conditions often fail to fully suppress ortho-attack or subsequent dibromination. The use of polar solvents with a dielectric constant greater than 5.0 is critical because it stabilizes the polar transition states involved in the electrophilic attack by the bromonium species generated from NBS. This stabilization lowers the activation energy for the para-substitution pathway relative to the ortho-pathway, effectively steering the reaction towards the desired 4-bromo isomer with high fidelity.
Furthermore, the mechanistic pathway involves a competition between mono-bromination and di-bromination, as illustrated in the reaction schemes provided in the patent documentation. The formation of the main product (Equation 1) competes directly with the formation of the ortho-isomer (Equation 2) and the dibrominated side product (Equation 3). The generation of the dibrominated by-product is particularly detrimental as it consumes two equivalents of the expensive NBS reagent for every molecule of starting material lost. By strictly controlling the molar ratio of 2-substituent phenol to NBS between 0.8:0.8 and 1.2:1.2, and maintaining the reaction temperature within the optimal window, the process kinetically favors the termination of the reaction after the first bromination event. This precise control over the reaction stoichiometry and thermodynamics ensures that the valuable starting material is not wasted on over-bromination, maximizing the atom economy of the process.
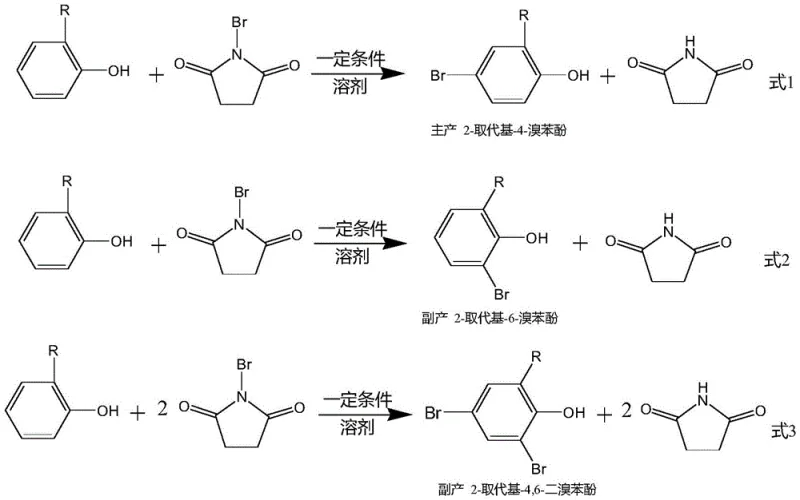
How to Synthesize 2-Substituent-4-Bromophenol Efficiently
Implementing this patented methodology requires strict adherence to the specified reaction parameters to replicate the high selectivity and conversion rates reported. The process begins with the dissolution of N-bromosuccinimide in a polar solvent, followed by its controlled addition to the phenol substrate to manage the exotherm and local concentration of the brominating agent. Following the reaction, a unique workup procedure involving evaporation and crystallization in non-polar solvents is employed to remove succinimide and trace impurities before final distillation. This sequence is designed to be robust and scalable, ensuring that the high purity observed in laboratory examples can be maintained during commercial production. The detailed standardized synthesis steps see the guide below for specific operational parameters.
- Dissolve N-bromosuccinimide (NBS) in a polar organic solvent such as acetonitrile or methanol with polarity greater than 5.0.
- Slowly add the NBS solution to 2-substituent phenol under stirring at temperatures between -30°C and 60°C to ensure regioselectivity.
- Concentrate the reaction mixture, crystallize impurities using non-polar solvents like hexane, and distill to isolate the pure target product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this NBS-based synthesis route offers tangible strategic advantages beyond mere chemical yield. The shift away from hazardous molecular bromine to solid NBS simplifies logistics and storage, as NBS is easier to transport and handle safely, reducing the regulatory burden and insurance costs associated with hazardous materials. Additionally, the mild reaction conditions, which can proceed at ambient temperature and pressure in many embodiments, drastically reduce energy consumption compared to processes requiring extreme heating or cooling. This energy efficiency, combined with the high selectivity that minimizes waste generation, aligns perfectly with modern green chemistry initiatives and corporate sustainability goals, making the supply of these intermediates more resilient against future environmental regulations.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous molecular bromine handling systems, coupled with the high selectivity that reduces the need for complex purification, leads to substantial cost savings. By minimizing the formation of by-products that consume additional reagents, the overall material cost per kilogram of the final product is significantly optimized, providing a competitive pricing structure for bulk buyers.
- Enhanced Supply Chain Reliability: The use of readily available and stable reagents like NBS and common polar solvents ensures that raw material sourcing is not a bottleneck. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by sensitive process deviations, guaranteeing consistent lead times for high-purity fine chemical intermediates and supporting just-in-time manufacturing models for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional bromination methods, primarily consisting of succinimide which can often be recovered or disposed of with lower environmental impact. The simplicity of the workup, involving standard evaporation and crystallization techniques, facilitates easy scale-up from pilot plants to multi-ton production facilities without the need for specialized exotic equipment, ensuring long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-substituent-4-bromophenol. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy for R&D and quality assurance teams evaluating this technology for their own processes. Understanding these nuances is vital for assessing the feasibility of integrating this intermediate into broader synthetic pathways for agrochemicals or pharmaceuticals.
Q: What is the primary advantage of using NBS over molecular bromine for this synthesis?
A: Using N-bromosuccinimide (NBS) eliminates the handling hazards associated with molecular bromine and significantly reduces the formation of polybrominated by-products, leading to higher selectivity for the 4-position.
Q: How does solvent polarity affect the reaction outcome?
A: Solvents with polarity greater than 5.0, such as acetonitrile and methanol, are critical for stabilizing the transition state that favors para-substitution, thereby suppressing the formation of ortho-brominated impurities.
Q: What purity levels can be achieved with this patented method?
A: The optimized process allows for raw material conversion rates reaching 99% and target product selectivity exceeding 96%, resulting in final distilled products with purity levels suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Substituent-4-Bromophenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final drug substances and specialty chemicals. Our technical team has extensively analyzed emerging technologies like the one described in CN115724721A to ensure our manufacturing capabilities remain at the forefront of the industry. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need kilogram quantities for clinical trials or metric tons for commercial launch, our supply remains uninterrupted. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-substituent-4-bromophenol meets the exacting standards required for pharmaceutical and fine chemical applications.
We invite you to discuss how our advanced manufacturing capabilities can support your specific project requirements. By leveraging our expertise in regioselective bromination and process optimization, we can offer a Customized Cost-Saving Analysis tailored to your volume needs. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to quality and efficiency can become a cornerstone of your supply chain strategy.
