Advanced Synthesis of Pyrazolinone Derivatives: Overcoming Regioselectivity Challenges for Commercial Scale-Up
The pharmaceutical and agrochemical industries rely heavily on the precise synthesis of heterocyclic scaffolds, among which pyrazolinone compounds occupy a critical position due to their potent biological activities, particularly in plant disease control. A pivotal advancement in this domain is documented in Chinese Patent CN1204647A, which discloses a robust method for preparing substituted pyrazolinone derivatives with exceptional regioselectivity. This patent addresses a longstanding synthetic challenge: the competitive formation of unwanted O-alkylated byproducts during the functionalization of the pyrazolone ring. By shifting from traditional alkyl halide alkylation to a sophisticated lithiation-sulfonate ester coupling strategy, the inventors have unlocked a pathway that significantly enhances the purity profile of the final active ingredients. For R&D directors and process chemists, understanding this shift is crucial, as it represents a move from empirical trial-and-error to a mechanistically driven approach that ensures consistent quality. The implications for supply chain stability are profound, as higher selectivity directly translates to simplified downstream processing and reduced waste generation, aligning perfectly with modern green chemistry mandates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the alkylation of pyrazolinone rings has been plagued by poor regiocontrol, leading to complex mixtures that are costly and difficult to separate. Traditional protocols often employ alkyl halides in the presence of bases such as potassium carbonate or sodium hydride in alcoholic or aromatic solvents. As illustrated in the reaction scheme below, these conditions frequently result in the nucleophilic attack occurring not only at the desired nitrogen atom but also at the carbonyl oxygen atom. 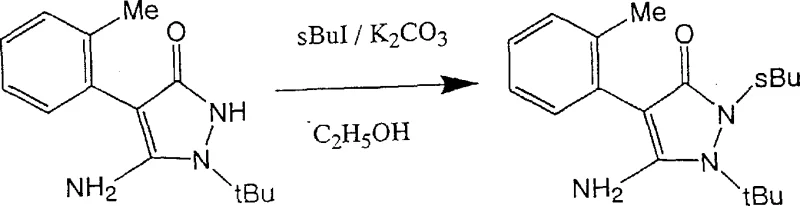 This lack of specificity generates substantial quantities of O-alkylated impurities, which, as shown in structural analyses within the patent data, can constitute the major product in some scenarios. For a procurement manager, this inefficiency is a nightmare; it means that a significant portion of raw materials is converted into worthless waste, driving up the cost of goods sold (COGS). Furthermore, separating these structural isomers often necessitates rigorous purification techniques like column chromatography, which are feasible in a laboratory setting but economically prohibitive and operationally hazardous when scaled to metric ton quantities for commercial agrochemical production.
This lack of specificity generates substantial quantities of O-alkylated impurities, which, as shown in structural analyses within the patent data, can constitute the major product in some scenarios. For a procurement manager, this inefficiency is a nightmare; it means that a significant portion of raw materials is converted into worthless waste, driving up the cost of goods sold (COGS). Furthermore, separating these structural isomers often necessitates rigorous purification techniques like column chromatography, which are feasible in a laboratory setting but economically prohibitive and operationally hazardous when scaled to metric ton quantities for commercial agrochemical production.
The Novel Approach
In stark contrast to the conventional limitations, the novel approach detailed in the patent utilizes a lithium salt intermediate to dictate the reaction trajectory with high precision. Instead of relying on the ambiguous reactivity of alkyl halides, the method employs sulfonate esters as the alkylating agents in the presence of the pre-formed lithium salt of the pyrazolinone. 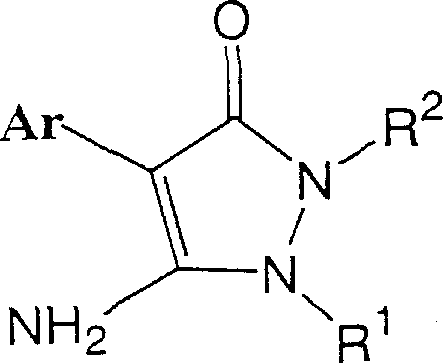 This strategic modification fundamentally alters the electronic environment of the nucleophile, favoring attack at the nitrogen center over the oxygen center. The use of ether solvents, such as tetrahydrofuran or 1,4-dioxane, is critical in this regime, as they stabilize the lithium cation and enhance the nucleophilicity of the nitrogen anion. For supply chain heads, this innovation offers a compelling value proposition: the ability to produce high-purity intermediates without the bottleneck of complex purification. The reaction conditions, typically ranging from 60°C to 150°C, are compatible with standard industrial reactor setups, ensuring that the transition from benchtop discovery to commercial manufacturing is seamless and risk-mitigated.
This strategic modification fundamentally alters the electronic environment of the nucleophile, favoring attack at the nitrogen center over the oxygen center. The use of ether solvents, such as tetrahydrofuran or 1,4-dioxane, is critical in this regime, as they stabilize the lithium cation and enhance the nucleophilicity of the nitrogen anion. For supply chain heads, this innovation offers a compelling value proposition: the ability to produce high-purity intermediates without the bottleneck of complex purification. The reaction conditions, typically ranging from 60°C to 150°C, are compatible with standard industrial reactor setups, ensuring that the transition from benchtop discovery to commercial manufacturing is seamless and risk-mitigated.
Mechanistic Insights into Lithium-Mediated Regioselective Alkylation
The core of this technological breakthrough lies in the unique coordination chemistry of the lithium ion compared to larger alkali metals like sodium or potassium. When the pyrazolinone precursor is treated with a lithium base, such as lithium hydroxide monohydrate under azeotropic dehydration or lithium alkylides, it forms a tight ion pair where the lithium cation coordinates strongly with the carbonyl oxygen. 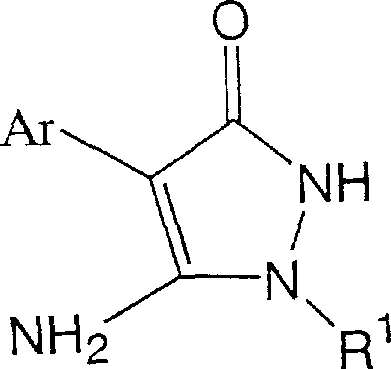 This coordination effectively blocks the oxygen atom from acting as a nucleophile, sterically and electronically shielding it from the incoming electrophile. Consequently, the negative charge density is localized more effectively on the nitrogen atom, making it the dominant nucleophilic site. This phenomenon explains the dramatic shift in product distribution observed in the experimental data, where the ratio of N-alkylate to O-alkylate improves drastically compared to sodium-mediated processes. For technical teams, this mechanistic understanding provides a blueprint for optimizing reaction parameters, such as solvent polarity and temperature, to further maximize yield and minimize impurity profiles in future derivative campaigns.
This coordination effectively blocks the oxygen atom from acting as a nucleophile, sterically and electronically shielding it from the incoming electrophile. Consequently, the negative charge density is localized more effectively on the nitrogen atom, making it the dominant nucleophilic site. This phenomenon explains the dramatic shift in product distribution observed in the experimental data, where the ratio of N-alkylate to O-alkylate improves drastically compared to sodium-mediated processes. For technical teams, this mechanistic understanding provides a blueprint for optimizing reaction parameters, such as solvent polarity and temperature, to further maximize yield and minimize impurity profiles in future derivative campaigns.
Furthermore, the choice of the leaving group in the electrophile plays a pivotal role in the success of this transformation. The patent specifies the use of sulfonate esters (R2-O-SO2R3), where R3 can be a C1-C10 alkyl or substituted phenyl group. Unlike halides, which can sometimes participate in side reactions or exhibit variable leaving group abilities depending on the specific halogen, sulfonate esters offer a consistent and highly reactive leaving group profile that complements the lithium salt nucleophile. The reaction proceeds through a concerted SN2-type mechanism where the sulfonate group departs cleanly, minimizing the formation of elimination byproducts that are common with bulky alkyl halides. This cleanliness of reaction is essential for maintaining a narrow impurity spectrum, a key requirement for regulatory approval in both pharmaceutical and agrochemical sectors. By controlling the stoichiometry, typically using 1.1 to 2.0 moles of sulfonate ester per mole of lithium salt, the process ensures complete conversion while avoiding excess reagent waste.
How to Synthesize N-Alkylated Pyrazolinones Efficiently
Implementing this synthesis route requires careful attention to the preparation of the lithium salt intermediate, as moisture sensitivity can impact the overall yield. The process begins with the dehydration of the pyrazolinone starting material using anhydrous lithium hydroxide in an aromatic solvent like toluene, ensuring the complete removal of water before the introduction of the ether solvent. Once the lithium salt is formed, the reaction mixture is transferred or directly treated with the sulfonate ester in a solvent system rich in ethers, such as 1,4-dioxane. The detailed standardized synthesis steps, including specific molar ratios, temperature gradients, and workup procedures, are outlined in the guide below to ensure reproducibility and safety in your pilot plant operations.
- Prepare the lithium salt of the pyrazolinone precursor by reacting with anhydrous lithium hydroxide or lithium alkylide under azeotropic dehydration conditions.
- React the resulting lithium salt with a sulfonate ester (R2-O-SO2R3) in an ether solvent such as tetrahydrofuran or 1,4-dioxane at elevated temperatures.
- Perform standard aqueous workup and extraction to isolate the N-alkylated product, avoiding complex chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and operational continuity, the adoption of this lithiation-based methodology offers transformative benefits that extend far beyond simple yield improvements. The primary economic driver is the drastic simplification of the purification workflow. In conventional processes, the presence of significant O-alkylated impurities often forces manufacturers to utilize preparative chromatography or multiple recrystallization cycles, both of which are time-consuming and solvent-intensive. By virtually eliminating these impurities at the source, the new method allows for straightforward isolation via extraction and concentration, significantly reducing solvent consumption and waste disposal costs. This efficiency gain directly contributes to cost reduction in agrochemical intermediate manufacturing, allowing producers to maintain healthy margins even in volatile raw material markets.
- Cost Reduction in Manufacturing: The elimination of expensive and time-consuming purification steps is the most immediate financial benefit. Conventional routes often require column chromatography to separate N- and O-alkylated isomers, a technique that is notoriously difficult to scale and expensive to operate due to high silica and solvent usage. The novel method's high regioselectivity means the crude product is already of high purity, often requiring only a simple wash or recrystallization to meet specifications. This reduction in processing complexity lowers utility costs, labor hours, and equipment occupancy time, leading to substantial cost savings per kilogram of finished product without compromising on quality standards.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for downstream formulation companies. The new method utilizes readily available sulfonate esters and common ether solvents, reducing dependency on specialized or scarce alkylating agents that might face supply disruptions. Furthermore, the robustness of the reaction conditions—tolerating a broad range of temperatures and concentrations—ensures consistent batch-to-batch quality. This predictability allows supply chain managers to optimize inventory levels and reduce safety stock requirements, knowing that the production process is stable and less prone to failure or deviation compared to the finicky conventional alkylation methods.
- Scalability and Environmental Compliance: Scaling chemical processes often exacerbates environmental challenges, particularly regarding solvent waste and energy consumption. The streamlined nature of this synthesis, which avoids the massive solvent volumes associated with chromatographic purification, inherently reduces the environmental footprint of the manufacturing process. The use of recoverable ether solvents like THF or dioxane further aligns with sustainability goals, as these can be efficiently distilled and reused. For facilities operating under strict environmental regulations, this method offers a clearer path to compliance, minimizing the generation of hazardous waste streams and simplifying the permitting process for capacity expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps R&D and procurement teams evaluate the feasibility of integrating this route into their existing portfolios.
Q: What is the primary technical advantage of the lithiation method described in CN1204647A?
A: The primary advantage is the significant suppression of O-alkylation byproducts. Conventional methods using alkyl halides often yield a mixture of N- and O-alkylated products, requiring difficult separation. The lithium salt method drastically improves regioselectivity towards the desired N-alkylated pyrazolinone.
Q: Which solvents are recommended for this regioselective alkylation process?
A: The patent specifies the use of ether solvents containing at least one ether bond. Preferred solvents include tetrahydrofuran (THF), 1,4-dioxane, and tetrahydropyran. These solvents facilitate the formation and stability of the lithium salt intermediate essential for high selectivity.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is highly suitable for scale-up. By minimizing the formation of O-alkylated impurities, the process reduces the need for resource-intensive purification steps like column chromatography, which are impractical on a multi-ton scale. The use of standard ether solvents and accessible sulfonate esters further supports commercial viability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazolinone Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercially viable process requires deep technical expertise and state-of-the-art infrastructure. Our team of process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the lithiation-sulfonate method are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of pyrazolinone intermediate meets the exacting standards required by global agrochemical and pharmaceutical leaders. Our commitment to quality assurance means that you receive a product that is not only chemically pure but also consistent in its physical properties, facilitating smooth downstream processing.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data rather than assumptions. Let us help you secure a competitive advantage in the market through superior chemistry and reliable supply chain execution.
