Industrial Scale Production of Optically Active Spiro Quaternary Ammonium Salts for Asymmetric Synthesis
Industrial Scale Production of Optically Active Spiro Quaternary Ammonium Salts for Asymmetric Synthesis
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the need for more efficient and cost-effective chiral phase transfer catalysts. Patent CN101146812A introduces a groundbreaking methodology for the production of optically active spiro quaternary ammonium salts, specifically designed to overcome the economic and synthetic limitations of prior art. Unlike traditional approaches that rely on the coupling of two distinct, expensive optically active binaphthyl derivatives, this invention leverages a sophisticated kinetic resolution strategy. By reacting a racemic bibenzyl derivative with an optically active azepine derivative, or vice versa, the process not only yields the desired chiral catalyst with high optical purity but also allows for the recovery of the unreacted enantiomer. This dual benefit addresses critical pain points for R&D directors seeking high-purity intermediates and supply chain managers focused on raw material efficiency.
For procurement professionals and technical decision-makers in the fine chemical sector, understanding the nuances of this technology is paramount. The ability to produce high-purity optically active ammonium salts through a streamlined process represents a substantial opportunity for cost reduction in chiral catalyst manufacturing. The patent details a versatile family of compounds where substituents such as fluorine, methoxy, and trifluoromethyl groups can be tuned to optimize catalytic performance. This flexibility ensures that the resulting catalysts are not merely academic curiosities but robust tools for the commercial scale-up of complex pharmaceutical intermediates, particularly in the asymmetric alkylation of α-amino acid derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active spiro quaternary ammonium salts has been hindered by prohibitive costs and complex supply chains. Prior art, such as that described in Japanese patent documents 2001-48866 and 2002-326992, typically necessitates the convergence of two different optically active binaphthyl or biphenyl derivatives. This requirement creates a bottleneck because sourcing or synthesizing two distinct chiral building blocks significantly inflates the bill of materials. Furthermore, these conventional routes often suffer from long reaction times and limited design flexibility, as the structural diversity of the catalyst is constrained by the availability of specific chiral precursors. For industrial applications, where tonnage and consistency are key, relying on such expensive and rigid synthetic pathways is often economically unviable, limiting the widespread adoption of these powerful catalysts in large-scale API production.
The Novel Approach
The methodology disclosed in CN101146812A revolutionizes this landscape by introducing a kinetic resolution-based production method. This novel approach allows for the use of racemic starting materials, which are significantly cheaper and easier to produce than their optically pure counterparts. In this process, a racemic bibenzyl compound reacts with an optically active azepine derivative. Due to the chiral environment provided by the azepine, one enantiomer of the racemic bibenzyl reacts preferentially, forming the desired optically active quaternary ammonium salt. Crucially, the unreacted enantiomer of the bibenzyl derivative remains in the solution and can be recovered as an optically active substance. This means that even if the starting material is racemic, the process yields a product with two asymmetric axes that are all optically active, while simultaneously generating valuable chiral byproducts. 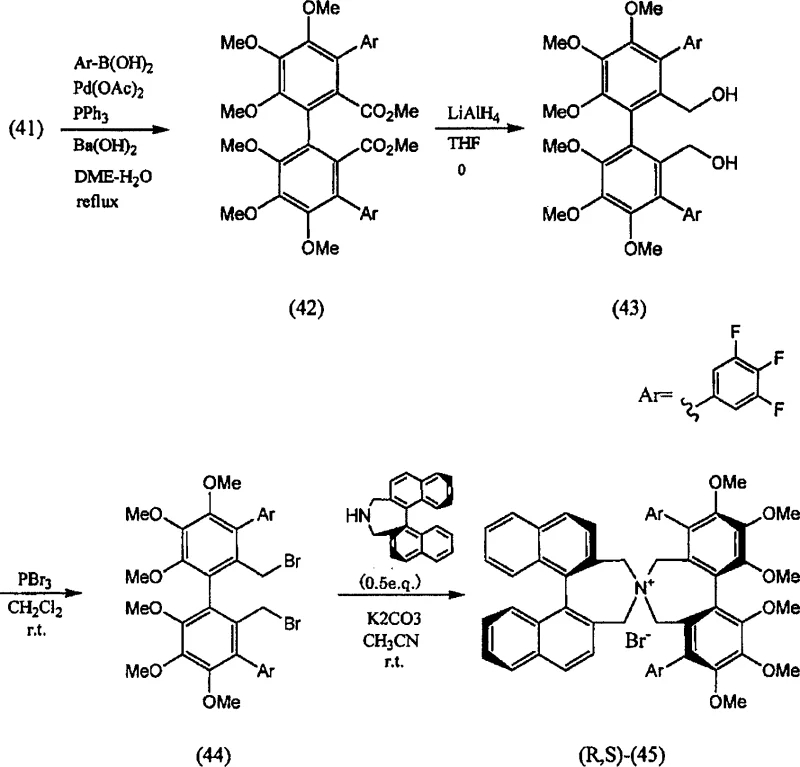
Mechanistic Insights into Kinetic Resolution and N-Benzylation
The core of this technology lies in the precise control of stereochemistry during the N-benzylation reaction. The mechanism involves the nucleophilic attack of the nitrogen atom in the azepine ring upon the benzylic carbon of the bibenzyl derivative, which bears a leaving group such as a bromine atom. When an optically active azepine is employed, its chiral backbone creates a diastereomeric transition state with the enantiomers of the racemic bibenzyl. One transition state is energetically favored, leading to a faster reaction rate for one specific enantiomer. This difference in reaction kinetics is the essence of kinetic resolution. The patent specifies that the molar ratio of the racemic modification to the optically active substance can range from 1.0 to 10 times, with 1.5 to 3.0 times being particularly preferable for industrial optimization. This flexibility allows process chemists to balance reaction speed against the recovery yield of the unreacted enantiomer.
Furthermore, the structural integrity of the catalyst is maintained through the rigidity of the spiro-cycle formed upon quaternization. The presence of bulky substituents at the 3,3'-positions of the biphenyl or binaphthyl cores prevents free rotation around the aryl-aryl bond, locking the molecule into a specific axial chirality. This axial asymmetry is critical for inducing high enantioselectivity in downstream reactions, such as the alkylation of glycine Schiff bases. The patent provides extensive data on various substituents (R1 to R6), demonstrating that electron-withdrawing groups like fluorine or trifluoromethyl can enhance the electrophilicity of the benzylic position, thereby facilitating the quaternization step under milder conditions. This mechanistic understanding is vital for R&D teams aiming to replicate or modify the synthesis for specific catalytic applications.
How to Synthesize Optically Active Quaternary Ammonium Salt Efficiently
The synthesis of these high-value chiral catalysts follows a logical progression from simple aromatic precursors to the final spiro-quaternized structure. The process begins with the preparation of the bibenzyl intermediate, typically involving the reduction of diesters to diols followed by halogenation to install the leaving groups. Concurrently, the azepine ring system is constructed, often through the cyclization of biphenyl diamines or related precursors with ammonia. The final and most critical step is the coupling of these two fragments. As detailed in the patent examples, this quaternization is performed in solvents like acetonitrile with inorganic bases. The following guide outlines the standardized operational procedure derived from the patent's preferred embodiments, ensuring reproducibility and high optical purity.
- Preparation of the Bibenzyl Intermediate: Synthesize the 2,2'-bis(substituted methyl)biaryl compound containing leaving groups (such as bromine) at the benzylic positions through reduction of diesters followed by halogenation.
- Synthesis of the Azepine Derivative: React the corresponding biphenyl diamine or di-halomethyl precursor with ammonia or a secondary amine to form the seven-membered azepine ring structure.
- Kinetic Resolution and Quaternization: React the racemic bibenzyl derivative with an optically active azepine derivative (or vice versa) in the presence of a base like potassium carbonate in acetonitrile to yield the final optically active quaternary ammonium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this kinetic resolution technology offers transformative benefits that extend beyond mere technical performance. The primary advantage is the drastic simplification of the raw material supply chain. By enabling the use of racemic starting materials, manufacturers can bypass the expensive and time-consuming resolution steps typically required to obtain optically pure binaphthyl or biphenyl precursors. This shift fundamentally alters the cost structure of catalyst production, making high-performance chiral phase transfer catalysts accessible for broader industrial applications. Additionally, the ability to recover unreacted enantiomers adds a secondary revenue stream or internal recycling loop, further enhancing the overall economic viability of the process.
- Cost Reduction in Manufacturing: The elimination of the need for two distinct optically active starting materials results in significant cost savings. Traditional methods require the purchase or synthesis of two expensive chiral building blocks, whereas this novel method requires only one, with the other being introduced as a cheaper racemate. Furthermore, the recovery of the unreacted enantiomer means that nearly 100% of the chiral information introduced into the system is utilized either in the product or as a recoverable asset. This high atom economy translates directly to lower cost per kilogram of the final catalyst, providing a competitive edge in the pricing of fine chemical intermediates.
- Enhanced Supply Chain Reliability: Racemic bibenzyl and biphenyl derivatives are structurally simpler and can be synthesized from commodity chemicals like xylenes and phenols through well-established industrial processes such as Ullmann coupling or Suzuki-Miyaura cross-coupling. This reliance on robust, scalable chemistry reduces the risk of supply disruptions associated with niche chiral pool materials. For supply chain planners, this means shorter lead times for raw material acquisition and greater predictability in production scheduling. The process conditions, utilizing common solvents like acetonitrile and bases like potassium carbonate, are also compatible with existing multipurpose reactor infrastructure, eliminating the need for specialized equipment investments.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are inherently scalable, operating effectively at temperatures ranging from room temperature to 80°C without the need for cryogenic cooling or high-pressure vessels. This thermal profile is ideal for large-scale batch processing. Moreover, the use of inorganic bases and the potential for solvent recovery aligns well with modern green chemistry principles. The high yields reported in the examples, such as 85% to 94% for the final quaternization steps, minimize waste generation. For environmental health and safety (EHS) teams, the predictable nature of the exotherm and the absence of hazardous transition metal catalysts in the final quaternization step simplify waste treatment and regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this optically active ammonium salt technology. These insights are derived directly from the experimental data and claims within patent CN101146812A, providing a reliable foundation for feasibility assessments. Understanding these details is essential for technical teams evaluating the integration of these catalysts into existing synthetic routes for amino acid production.
Q: What is the primary advantage of using kinetic resolution in this synthesis?
A: Kinetic resolution allows for the production of optically active quaternary ammonium salts even when starting with racemic materials. Crucially, the unreacted enantiomer of the racemic starting material can be recovered in an optically active form, significantly improving atom economy and reducing raw material waste compared to traditional methods requiring two chiral starting materials.
Q: Can these catalysts be used for synthesizing unnatural amino acids?
A: Yes, the patent data explicitly demonstrates that these optically active spiro quaternary ammonium salts act extremely effectively as phase transfer catalysts for synthesizing both natural and unnatural optically active α-amino acids, achieving high enantiomeric excess (ee) values such as 97% ee in reference examples.
Q: What are the typical reaction conditions for the final quaternization step?
A: The final N-benzylation reaction is typically conducted in polar aprotic solvents like acetonitrile or DMF, using inorganic bases such as potassium carbonate or sodium hydroxide. The reaction temperature generally ranges from room temperature to 80°C, making it highly suitable for standard stainless steel reactor setups without requiring cryogenic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Ammonium Salt Supplier
The technological breakthroughs detailed in CN101146812A represent a significant leap forward in the field of asymmetric catalysis, offering a pathway to more sustainable and cost-effective chiral synthesis. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these patented methodologies into commercial reality. Our team specializes in the process development and scale-up of complex chiral intermediates, ensuring that the theoretical advantages of kinetic resolution are fully realized in production. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, adhering to stringent purity specifications and utilizing rigorous QC labs to guarantee the optical purity and chemical integrity of every batch.
We invite pharmaceutical and agrochemical companies to explore the potential of these advanced catalysts for their asymmetric synthesis needs. Whether you require custom synthesis of specific derivatives mentioned in the patent or optimization of the kinetic resolution parameters for your specific application, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis tailored to your project. We encourage you to reach out to our technical procurement team to obtain specific COA data and route feasibility assessments, ensuring that your transition to this superior catalytic technology is seamless and commercially sound.
