Scalable Synthesis of 2-Chloro-5-Thiophenecarboxylic Acid for Antithrombotic Drug Production
Introduction to Advanced Intermediate Synthesis
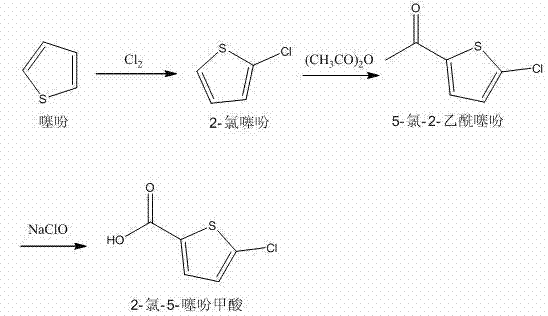
The pharmaceutical landscape for antithrombotic agents continues to evolve, driven by the critical need for safer, oral anticoagulants like Razaxaban. At the heart of this therapeutic class lies the key building block, 2-chloro-5-thiophenecarboxylic acid. Patent CN102993164A discloses a robust and environmentally conscious preparation method that addresses the longstanding challenges of yield optimization and impurity control in heterocyclic chemistry. This technical disclosure outlines a streamlined three-step sequence starting from readily available thiophene, utilizing electrophilic chlorination, acetylation, and a final oxidative cleavage. For global procurement leaders and R&D directors, understanding the nuances of this specific pathway is essential for securing a reliable supply chain for next-generation cardiovascular medications. The methodology emphasizes operational simplicity and high atom economy, positioning it as a superior alternative to traditional routes that often rely on hazardous reagents or complex purification protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functionalized thiophene carboxylic acids has been plagued by significant technical hurdles that impact both cost and safety profiles. Traditional routes frequently employ harsh oxidizing agents such as potassium permanganate or chromic acid derivatives, which generate substantial quantities of heavy metal waste sludge, creating severe environmental compliance liabilities for manufacturers. Furthermore, direct lithiation strategies, while effective in laboratory settings, often suffer from poor scalability due to the requirement for cryogenic conditions and pyrophoric reagents, leading to inconsistent batch-to-batch quality. The lack of regioselectivity in early-stage functionalization often results in complex mixtures of isomers, necessitating energy-intensive chromatographic separations that drastically erode profit margins. These legacy processes are increasingly untenable in a modern regulatory environment that demands greener chemistry and stricter control over residual metal impurities in active pharmaceutical ingredients.
The Novel Approach
The methodology detailed in CN102993164A introduces a paradigm shift by leveraging a sequential chlorination-acetylation-oxidation strategy that circumvents these traditional bottlenecks. By initiating the synthesis with controlled gas-phase chlorination of thiophene, the process ensures high regioselectivity for the 2-position, establishing a clean foundation for subsequent transformations. The introduction of an acetyl group serves as a strategic handle for the final oxidative cleavage, allowing for the use of sodium hypochlorite—a cost-effective and commercially ubiquitous oxidant—instead of expensive transition metal catalysts. This approach not only simplifies the workup procedures through straightforward phase separations and crystallizations but also inherently reduces the toxicological burden of the manufacturing process. The result is a synthetic route that balances high chemical efficiency with operational safety, making it ideally suited for the rigorous demands of cGMP production environments.
Mechanistic Insights into Electrophilic Substitution and Hypochlorite Oxidation
The core of this synthesis relies on precise control over electrophilic aromatic substitution mechanisms, particularly during the initial chlorination step. Thiophene, being an electron-rich heterocycle, is highly susceptible to electrophilic attack; however, controlling the degree of substitution is paramount to preventing the formation of di- or tri-chlorinated byproducts. The patent specifies a careful thermal profile, initiating the reaction at 35-40°C and ramping to 50-55°C, which kinetically favors mono-chlorination at the alpha position while minimizing thermal degradation or over-reaction. The stoichiometry of chlorine gas is tightly regulated at a molar ratio of 1.1:1 to 1.3:1 relative to thiophene, ensuring that the electrophile is the limiting reagent to drive selectivity. Following this, the acetylation step utilizes phosphoric acid as a mild Lewis acid catalyst to activate acetic anhydride, facilitating the Friedel-Crafts acylation without the aggressive conditions associated with aluminum chloride, thereby reducing corrosion risks and waste generation.
The final transformation involves the oxidative cleavage of the methyl ketone moiety in 5-chloro-2-acetyl thiophene to yield the target carboxylic acid. This reaction proceeds via a haloform-type mechanism where sodium hypochlorite acts as both the base and the oxidant. Under the specified conditions of 40-50°C, the hypochlorite ion sequentially halogenates the alpha-methyl group, rendering it susceptible to nucleophilic attack by hydroxide ions generated in situ. This leads to the cleavage of the carbon-carbon bond and the formation of the carboxylate salt, which is subsequently acidified to precipitate the free acid. The use of aqueous sodium sulfite in the workup is a critical quality control measure, serving to quench excess oxidant and prevent potential over-oxidation of the sensitive thiophene ring sulfur atom. This mechanistic elegance ensures that the final product maintains high purity with minimal risk of ring-opening degradation.
How to Synthesize 2-Chloro-5-Thiophenecarboxylic Acid Efficiently
Implementing this synthesis requires strict adherence to the thermal and stoichiometric parameters defined in the patent to ensure optimal yield and safety. The process begins with the preparation of 2-chlorothiophene, followed by its conversion to the acetyl derivative, and concludes with the oxidative cleavage. Each stage demands precise monitoring of pH and temperature to maintain reaction fidelity. The following guide outlines the critical operational phases derived from the exemplary embodiments, providing a framework for process engineers to establish robust manufacturing protocols. Detailed standardized synthesis steps are provided in the section below.
- Chlorinate thiophene in dichloroethane at 35-55°C with controlled chlorine gas flow to obtain 2-chlorothiophene.
- Perform Friedel-Crafts acetylation on 2-chlorothiophene using acetic anhydride and phosphoric acid catalyst under reflux.
- Oxidize 5-chloro-2-acetyl thiophene using sodium hypochlorite solution at 40-50°C, followed by acidification and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond simple yield metrics. The elimination of precious metal catalysts and the reliance on commodity chemicals like chlorine, acetic anhydride, and bleach fundamentally alters the cost structure of the intermediate. By removing the need for specialized catalyst recovery systems or extensive heavy metal scavenging steps, the overall processing time is significantly reduced, leading to faster throughput and lower utility consumption. Furthermore, the use of dichloroethane and toluene, which are standard solvents with well-established recovery and recycling infrastructures, minimizes solvent procurement risks and volatility. This process design inherently supports a more resilient supply chain by reducing dependency on niche reagents that are prone to market shortages.
- Cost Reduction in Manufacturing: The substitution of expensive oxidizing agents with sodium hypochlorite represents a drastic reduction in raw material costs, as bleach is produced globally at massive scale with stable pricing. Additionally, the avoidance of cryogenic conditions and pyrophoric reagents eliminates the need for specialized low-temperature reactors and inert atmosphere handling equipment, resulting in substantial capital expenditure savings. The simplified workup procedure, which relies on phase separation and crystallization rather than chromatography, further drives down operational expenses by reducing labor hours and consumable usage. These cumulative efficiencies translate into a highly competitive cost profile for the final API intermediate.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including thiophene, chlorine gas, and acetic anhydride, are bulk commodities available from multiple global suppliers, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions, which tolerate minor fluctuations without catastrophic failure, ensures consistent production output even in varying operational environments. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, preventing costly production stoppages. The ability to source reagents locally in most major chemical hubs further shortens lead times and reduces logistics complexity.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations such as gas-liquid reactions and reflux distillation that are easily translated from pilot to commercial scale. The environmental footprint is markedly lower compared to traditional methods, as the primary byproduct is sodium chloride, which is benign and easy to treat. This alignment with green chemistry principles facilitates smoother regulatory approvals and reduces the burden of waste disposal fees. The inherent safety of the process, avoiding highly exothermic or unstable intermediates, also lowers insurance premiums and enhances facility safety ratings.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-chloro-5-thiophenecarboxylic acid. These insights are derived directly from the patent specifications and practical manufacturing considerations. They are intended to clarify the feasibility and advantages of this specific synthetic route for stakeholders evaluating potential partnerships.
Q: What is the primary advantage of using sodium hypochlorite for oxidation in this route?
A: Using sodium hypochlorite avoids the use of heavy metal oxidants like chromium or manganese, significantly reducing toxic waste disposal costs and environmental compliance burdens while maintaining high conversion rates.
Q: How is regioselectivity controlled during the initial chlorination of thiophene?
A: Regioselectivity is managed by strict temperature control (35-55°C) and precise molar ratios of chlorine to thiophene (1.1:1 to 1.3:1), minimizing the formation of polychlorinated byproducts.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes common industrial solvents like dichloroethane and toluene, and avoids exotic catalysts, making it highly scalable from pilot plants to multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-5-Thiophenecarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antithrombotic therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering 2-chloro-5-thiophenecarboxylic acid with stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest international standards. Our facility is equipped to handle the specific safety and environmental requirements of chlorination and oxidation chemistries, guaranteeing a secure and compliant supply for your global operations.
We invite you to collaborate with us to optimize your supply chain for Razaxaban and related cardiovascular drugs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's success and accelerate your time to market.
