Advanced Manufacturing of p-Methylsulfurylbenzaldehyde for Global Pharmaceutical Supply Chains
Introduction to Advanced Intermediate Synthesis
The global demand for broad-spectrum antibiotics continues to drive innovation in the manufacturing of key pharmaceutical intermediates, particularly for veterinary and human applications. Patent CN102827041B discloses a highly efficient preparation method for p-methylsulfurylbenzaldehyde, a critical precursor in the synthesis of Thiamphenicol. This specific chemical entity serves as the foundational scaffold for a class of antibiotics known for their efficacy against a wide range of pathogens, including Hemophilus influenzae and various enterobacteriaceae. The disclosed technology represents a significant departure from legacy synthetic routes, introducing a streamlined bromination-hydrolysis sequence that addresses long-standing issues regarding yield, purity, and environmental impact. By integrating a closed-loop reagent recovery system, this method not only enhances the economic viability of production but also aligns with increasingly stringent global environmental regulations regarding heavy metal discharge and waste management.
For R&D directors and procurement specialists, understanding the nuances of this patented pathway is essential for securing a reliable supply chain. The transition from hazardous oxidants to a recyclable bromine-based system offers a compelling value proposition. The process achieves a hydrolysis yield enhancement from historical averages of roughly 85% to over 94%, while simultaneously elevating product purity to exceed 98%. These improvements are not merely incremental; they represent a fundamental optimization of the reaction thermodynamics and kinetics, ensuring that the final API intermediate meets the rigorous quality standards required by international regulatory bodies. As we delve deeper into the technical specifics, it becomes clear that this methodology offers a robust solution for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of p-methylsulfurylbenzaldehyde has been plagued by significant technical and environmental hurdles that compromise both cost-efficiency and operational safety. Traditional pathways often rely on the Stephen aldehyde synthesis, which utilizes tin protochloride and hydrochloric acid in diethyl ether, a solvent combination that presents substantial fire hazards and yields relatively low conversion rates around 68%. Alternatively, oxidation methods employing chromium trioxide have been widely used; however, these processes generate vast quantities of toxic heavy metal waste, creating a massive liability for waste treatment and disposal. The reliance on chromium-based oxidants not only inflates the cost of production due to expensive remediation requirements but also poses severe risks of soil and water contamination, making such facilities targets for regulatory scrutiny. Furthermore, other oxidative routes using hydrogen peroxide on sulfide precursors suffer from raw material scarcity, as specific sulfide starting materials are difficult to source commercially in bulk quantities, leading to supply chain bottlenecks and price volatility.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a direct bromination-hydrolysis strategy starting from p-methylsulfonyltoluene, a readily available and cost-effective raw material. This innovative route bypasses the need for toxic heavy metals and hazardous ether solvents entirely, replacing them with a controlled bromination followed by an aqueous hydrolysis step. The core of this advancement lies in the seamless integration of the bromination byproduct, hydrogen bromide, directly into the hydrolysis stage, thereby creating a synergistic reaction environment that maximizes atom economy. By operating under reduced pressure and elevated temperatures, the process ensures complete conversion of the starting material while minimizing side reactions that typically lead to impurity formation. This shift in synthetic strategy effectively decouples production costs from the volatile markets of specialty oxidants and rare precursors, establishing a more stable and predictable manufacturing baseline.
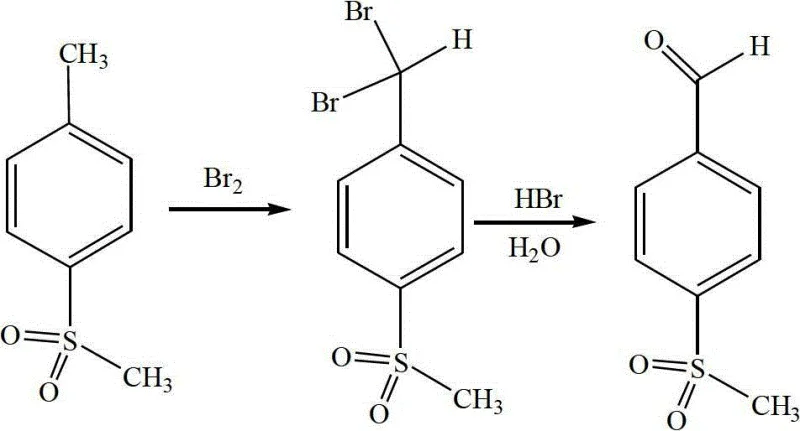
Mechanistic Insights into Bromination-Hydrolysis Cascade
The chemical elegance of this process is rooted in the precise control of radical bromination followed by nucleophilic substitution. In the initial phase, p-methylsulfonyltoluene undergoes free-radical bromination at the benzylic position. The reaction conditions are meticulously tuned, with the temperature maintained between 150°C and 160°C under a vacuum of -40 to -50 KPa. This specific thermal and pressure regime is critical for driving the substitution of two hydrogen atoms on the methyl group with bromine atoms, forming the gem-dibromide intermediate, p-methylsulfonyl dibromomethylbenzene. The use of a slight molar excess of bromine (ratio 1:2.10 to 1:2.25) ensures that the reaction proceeds to completion, as monitored by HPLC, preventing the accumulation of mono-brominated species which could act as persistent impurities in downstream processing. The evolution of hydrogen bromide gas during this exothermic step is carefully managed, not as a waste stream, but as a valuable reagent for the subsequent stage.
Following bromination, the crude dibromide intermediate is subjected to hydrolysis in the presence of the captured hydrogen bromide. This step converts the unstable gem-dibromide into the target aldehyde functionality. The mechanism involves the nucleophilic attack of water on the electron-deficient benzylic carbon, facilitated by the acidic environment provided by the HBr. Maintaining the hydrolysis temperature between 95°C and 102°C is paramount; this range provides sufficient activation energy for the hydrolysis to proceed rapidly while preventing the over-oxidation of the aldehyde to the corresponding carboxylic acid, a common side reaction in aldehyde synthesis. The final refinement involves a delicate pH adjustment to neutrality (pH 7-8) using sodium hydroxide, which precipitates the product while leaving soluble inorganic salts in the aqueous phase. This simple yet effective workup procedure ensures high purity without the need for complex chromatographic separations, making it ideal for large-scale industrial application.
How to Synthesize p-Methylsulfurylbenzaldehyde Efficiently
The practical implementation of this synthesis requires strict adherence to the thermal and stoichiometric parameters defined in the patent embodiments to ensure reproducibility and safety. The process is designed as a semi-continuous operation where the output of the bromination reactor feeds directly into the hydrolysis vessel, minimizing material handling and exposure. Operators must monitor the vacuum levels and temperature gradients closely during the bromine addition phase to prevent runaway reactions. Following the reaction, the recovery of unreacted bromine and the regeneration of bromine from waste hydrogen bromide using sodium chlorate are critical unit operations that sustain the economic viability of the plant. For a comprehensive breakdown of the specific equipment setups, stirring rates, and exact addition times required to replicate the high yields reported in the patent, please refer to the standardized synthesis protocol below.
- Bromination Phase: Melt p-methylsulfonyltoluene and heat to 150°C under vacuum, then dropwise add liquid bromine (molar ratio 1:2.10-2.25) while maintaining temperature to form the dibromide intermediate.
- Hydrolysis Phase: Transfer the brominated product into a hydrolysis kettle containing 15-25Wt.% hydrobromic acid, heat to 95-102°C, and stir for 5-8 hours to convert the dibromide to the crude aldehyde.
- Refinement and Recycling: Adjust the crude product pH to 7-8 using sodium hydroxide, centrifuge, and dry. Simultaneously, recover excess hydrogen bromide by oxidizing it with sodium chlorate to regenerate bromine for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this bromination-hydrolysis technology offers profound advantages in terms of cost structure and supply chain resilience. The elimination of chromium trioxide and ether solvents removes the necessity for expensive hazardous waste disposal contracts and specialized containment infrastructure, leading to substantial operational expenditure savings. Furthermore, the ability to recycle hydrogen bromide back into elemental bromine creates a closed-loop material flow that insulates the manufacturer from fluctuations in the global bromine market. This self-sufficiency in reagent generation means that production can continue uninterrupted even during periods of raw material scarcity, providing a level of supply security that is increasingly valued by multinational pharmaceutical buyers who prioritize business continuity.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the drastic simplification of the waste treatment workflow. By avoiding heavy metal contaminants, the facility avoids the high costs associated with neutralizing and disposing of toxic sludge, which can often account for a significant portion of the total manufacturing cost in traditional oxidation routes. Additionally, the regeneration of bromine from byproduct acids means that the net consumption of this expensive halogen is minimized, directly lowering the variable cost per kilogram of the finished intermediate. The use of p-methylsulfonyltoluene, a commodity chemical with a stable supply chain, further anchors the production costs, preventing the price spikes often seen with niche starting materials.
- Enhanced Supply Chain Reliability: Supply chain leaders benefit from the robustness of the raw material base, as p-methylsulfonyltoluene is produced by multiple manufacturers globally, reducing the risk of single-source dependency. The process design inherently supports continuous operation through its efficient recycling loops, ensuring that production throughput is not limited by the availability of fresh reagents. This reliability translates into shorter and more predictable lead times for customers, allowing pharmaceutical companies to optimize their own inventory levels and reduce the need for safety stock. The stability of the process also minimizes the risk of batch failures, ensuring a consistent flow of high-quality intermediate to downstream API manufacturers.
- Scalability and Environmental Compliance: The environmental profile of this technology facilitates easier permitting and expansion of production capacity. With wastewater discharge significantly reduced and free from persistent organic pollutants or heavy metals, the process aligns perfectly with modern green chemistry principles and strict environmental regulations. This compliance reduces the regulatory burden on the supply chain, preventing potential shutdowns due to environmental violations. The scalability is further evidenced by the successful demonstration in multi-hundred-kilogram batches, proving that the heat transfer and mixing dynamics are manageable at a commercial scale, paving the way for seamless expansion to meet growing market demand for Thiamphenicol.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and quality of p-methylsulfurylbenzaldehyde using this advanced methodology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing transparency for potential partners. Understanding these details is crucial for assessing the fit of this intermediate within your specific formulation or synthesis requirements.
Q: How does this new synthesis method improve environmental compliance compared to traditional routes?
A: Traditional methods often utilize chromium trioxide or cyanide-based reagents, generating hazardous heavy metal waste and toxic effluents. This patented process eliminates heavy metal catalysts entirely and implements a closed-loop hydrogen bromide recovery system, significantly reducing wastewater discharge and avoiding the complex disposal protocols associated with chromium contamination.
Q: What represents the primary cost-saving mechanism in this manufacturing process?
A: The primary economic advantage stems from the efficient recycling of reagents. By capturing hydrogen bromide generated during bromination and converting excess acid back into elemental bromine using sodium chlorate, the process drastically reduces the consumption of fresh bromine. Additionally, the use of readily available p-methylsulfonyltoluene avoids the premium pricing of scarce precursors required by older synthetic pathways.
Q: Is this process scalable for industrial production of Thiamphenicol intermediates?
A: Yes, the process is designed for scalability, utilizing standard unit operations such as vacuum bromination and aqueous hydrolysis which are easily adaptable to large-scale reactors. The patent demonstrates successful execution in 1000L reactors with high yields, indicating robust thermal control and manageable exothermic profiles suitable for commercial metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Methylsulfurylbenzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is critical for the future of the pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patented process are fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of p-methylsulfurylbenzaldehyde meets the highest international standards for impurity profiles and physical properties. Our commitment to technical excellence allows us to deliver a product that not only performs reliably in downstream synthesis but also supports our clients' sustainability goals.
We invite procurement managers and R&D leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized production capabilities, we can help you reduce the total cost of ownership for this critical intermediate. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. We are ready to demonstrate how our advanced manufacturing platform can become a strategic asset to your supply chain, delivering both quality and value in equal measure.
