Advanced Synthesis of Axially Chiral Alpha-Allene Alcohols for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex chiral scaffolds, particularly those possessing axial chirality which can be effectively transferred to central chirality in downstream transformations. Patent CN102675049B introduces a groundbreaking approach for the synthesis of optically active axially chiral α-allene alcohols, a class of versatile intermediates crucial for the construction of 2,5-dihydrofurans, vinyl oxiranes, and various functionalized allenes. This technology represents a significant leap forward in asymmetric synthesis, utilizing a zinc dibromide-promoted system that couples tert-butyldimethylsilyl (TBS) protected propargyl alcohols with aldehydes in the presence of chiral α,α-diphenylprolinol derivatives. The resulting products exhibit exceptional optical purity, with enantiomeric excess (ee) values frequently surpassing 96%, and in many optimized instances reaching up to 99% ee. For R&D directors and procurement specialists alike, this patent outlines a pathway that not only simplifies the synthetic route but also drastically improves the safety and scalability profile compared to legacy methods, positioning it as a cornerstone technology for the reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axially chiral α-allenols has been plagued by significant operational hazards and inefficiencies that hinder large-scale manufacturing. Traditional protocols often rely heavily on the use of highly reactive and dangerous organometallic reagents such as n-butyllithium (n-BuLi), ethylmagnesium bromide (EtMgBr), or lithium aluminum hydride (LiAlH4). These reagents necessitate stringent cryogenic conditions, often requiring temperatures well below -70°C to maintain control over reactivity and stereoselectivity, which imposes a massive energy burden and requires specialized, expensive reactor infrastructure. Furthermore, the multi-step nature of conventional routes often leads to cumulative yield losses, resulting in overall low total yields that are economically unsustainable for commercial production. The handling of pyrophoric reagents also introduces severe safety risks, complicating logistics and increasing the cost of compliance with environmental and safety regulations. Consequently, finding a reliable agrochemical intermediate supplier or pharma partner who can deliver these complex motifs using safer chemistry has been a persistent challenge in the supply chain.
The Novel Approach
The methodology disclosed in CN102675049B fundamentally reshapes the landscape of allene synthesis by replacing hazardous organometallics with a benign zinc salt promoter system. As illustrated in the general reaction scheme below, the process employs zinc bromide (ZnBr2) as a Lewis acid promoter in conjunction with a chiral secondary amine catalyst, specifically α,α-diphenylprolinol derivatives like (S)-3a. This innovation allows the reaction to proceed in common organic solvents such as toluene at elevated temperatures ranging from 100°C to 130°C, completely eliminating the need for cryogenic cooling. The reaction demonstrates remarkable substrate universality, accommodating a wide range of aldehydes including aliphatic, cyclic, and aromatic variants, while maintaining high stereocontrol. Following the coupling step, a simple deprotection using tetra-n-butylammonium fluoride (TBAF) yields the final axially chiral α-allenol. This streamlined two-step sequence (coupling followed by deprotection) significantly reduces processing time and waste generation, offering a clear path for cost reduction in pharmaceutical intermediate manufacturing through simplified operations and higher throughput.
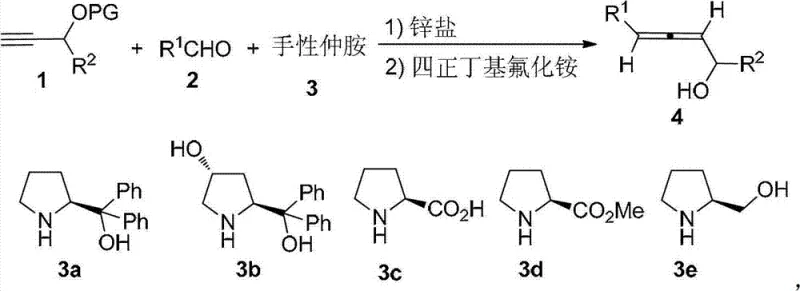
Mechanistic Insights into ZnBr2-Promoted Asymmetric Allenylation
The core of this technological advancement lies in the synergistic interaction between the zinc salt promoter and the chiral amine catalyst, which orchestrates the stereochemical outcome of the allene formation. The zinc bromide acts as a Lewis acid to activate the silyl-protected propargyl alcohol, facilitating the nucleophilic attack on the aldehyde carbonyl carbon. Simultaneously, the chiral α,α-diphenylprolinol creates a sterically defined environment that directs the approach of the reactants, ensuring the formation of the desired axial chirality (R or S configuration) with high fidelity. The mechanism involves a concerted transition state where the bulky phenyl groups of the catalyst shield one face of the reacting species, thereby enforcing high enantioselectivity. Experimental data from the patent indicates that using (S)-configured prolinol predominantly yields the (R)-axial chirality allene, while the (R)-configured catalyst produces the (S)-isomer, demonstrating predictable stereocontrol. This level of mechanistic understanding is critical for R&D teams aiming to optimize impurity profiles, as the high diastereoselectivity (de > 97% in many cases) ensures that downstream purification is straightforward, often requiring only standard column chromatography rather than complex chiral separations.
Furthermore, the stability of the intermediate silyl-protected species allows for flexible processing windows, reducing the risk of decomposition that often plagues sensitive allene intermediates. The subsequent deprotection step using TBAF is mild and highly selective, cleaving the silyl ether without affecting the sensitive allene double bond system or the newly formed stereocenters. This robustness is essential for maintaining the integrity of the chiral information throughout the synthesis. The ability to tune the steric and electronic properties of the aldehyde component (R1 group) and the propargyl alcohol (R2 group) without compromising the optical purity provides chemists with a powerful toolbox for generating diverse libraries of chiral building blocks. Such versatility is invaluable for medicinal chemistry programs exploring structure-activity relationships (SAR) where rapid access to varied chiral scaffolds is required to accelerate drug discovery timelines.
How to Synthesize Axially Chiral Alpha-Allene Alcohols Efficiently
The practical implementation of this synthesis is designed for ease of execution in standard laboratory and pilot plant settings. The process begins with the preparation of a dry reaction vessel under an inert atmosphere, into which the zinc bromide promoter and chiral amine are introduced. The silyl-protected propargyl alcohol and the chosen aldehyde are then added in a molar ratio that favors high conversion, typically utilizing a slight excess of the aldehyde. The reaction mixture is heated to reflux in toluene, allowing the thermodynamic driving force to push the equilibrium towards the protected allenol intermediate. After the coupling is complete, the crude material is passed through a short silica plug to remove metal salts before undergoing the deprotection step. The detailed standardized synthesis steps, including specific stoichiometric ratios, temperature profiles, and workup procedures optimized for maximum yield and purity, are outlined in the guide below.
- Combine zinc bromide, chiral secondary amine, silyl-protected propargyl alcohol, and aldehyde in dry toluene.
- Heat the mixture to 100-130°C and stir for 5-20 hours under inert atmosphere to form the protected intermediate.
- Treat the crude product with tetra-n-butylammonium fluoride (TBAF) in THF at 0°C to room temperature to remove the silyl group.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ZnBr2-promoted synthesis offers tangible strategic benefits that extend beyond mere chemical elegance. The elimination of hazardous reagents like n-BuLi removes the need for specialized storage and handling protocols, significantly lowering the operational overhead and insurance costs associated with raw material management. Moreover, the use of commodity chemicals such as toluene and zinc bromide ensures a stable and resilient supply chain, mitigating the risks of shortages that often affect exotic or highly regulated reagents. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in temperature or mixing efficiency, leading to more consistent batch-to-batch quality and reducing the rate of rejected lots. This reliability is paramount for maintaining continuous manufacturing schedules and meeting tight delivery deadlines for downstream API production.
- Cost Reduction in Manufacturing: The shift from cryogenic conditions to standard heating (100-130°C) results in substantial energy savings, as maintaining reactors at -78°C is exponentially more expensive than refluxing in toluene. Additionally, the high yields (often exceeding 60-70%) and excellent stereoselectivity minimize the loss of valuable starting materials and reduce the volume of solvent required for purification. By avoiding expensive chiral HPLC separations through high intrinsic ee values, the overall cost of goods sold (COGS) is drastically reduced, making the final intermediate more competitive in the global market.
- Enhanced Supply Chain Reliability: The reliance on readily available raw materials such as substituted aldehydes and protected alkynes ensures that production is not bottlenecked by scarce reagents. The simplified workflow, which combines coupling and deprotection in a linear fashion without complex intermediate isolations, shortens the overall cycle time. This efficiency translates directly into shorter lead times for customers, allowing for more responsive inventory management and the ability to scale up production rapidly in response to market demand without extensive process re-validation.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from milligram to multi-gram scales in the patent examples with consistent results. The use of zinc salts, which are less toxic and easier to dispose of than heavy transition metals or pyrophoric organometallics, aligns with green chemistry principles and simplifies waste treatment. This environmental compatibility reduces the regulatory burden and facilitates smoother audits, ensuring long-term sustainability of the supply source for high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and sourcing of these axially chiral intermediates. The answers are derived directly from the experimental data and scope defined in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the fit of this technology within their specific project requirements and quality standards.
Q: What are the advantages of using ZnBr2 over traditional organolithium reagents?
A: ZnBr2 promotion avoids the use of hazardous reagents like n-BuLi or LiAlH4, operates at safer temperatures (100-130°C vs cryogenic), and offers superior scalability and safety profiles for industrial production.
Q: What level of enantioselectivity can be achieved with this method?
A: The method consistently achieves high enantiomeric excess (ee) values, typically exceeding 96% and often reaching 98-99% ee, depending on the specific aldehyde substrate and chiral amine catalyst used.
Q: Can this synthesis be scaled for commercial supply?
A: Yes, the process utilizes common solvents like toluene and standard heating conditions without requiring specialized cryogenic equipment, making it highly suitable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Alpha-Allene Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to delivering products with stringent purity specifications, leveraging rigorous QC labs to verify enantiomeric excess and chemical purity for every batch. Our facility is equipped to handle the specific requirements of this ZnBr2-promoted chemistry, guaranteeing a secure and consistent supply of these valuable building blocks for your drug development programs.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how optimizing this pathway can reduce your overall project costs. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your supply chain with reliable, high-performance chemical solutions.
