Advanced Synthesis of Benzopyrazine Compounds: A Safe, Scalable Route for Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for complex heterocyclic scaffolds that balance efficacy with manufacturing safety. Patent CN113754666A introduces a groundbreaking methodology for synthesizing 1,2,3,4,6,11,12,12a-octahydrobenzo[e]pyrazino[1,2-a]azepine derivatives, a class of benzopyrazine compounds with significant therapeutic potential. This innovation addresses critical bottlenecks in the production of benzo-heterocyclic medicines, specifically targeting the construction of the challenging seven-membered azepine ring. By leveraging modern organometallic catalysis, particularly Ring-Closing Metathesis (RCM), the disclosed route offers a superior alternative to traditional methods that rely on hazardous reagents. For R&D directors and process chemists, this patent represents a pivotal shift towards safer, more efficient manufacturing of high-value API intermediates.
The structural versatility of these compounds allows for extensive modification at the R1, R2, and R3 positions, enabling the fine-tuning of pharmacokinetic properties such as lipid solubility and target binding affinity. The core framework serves as a privileged structure for developing agents against parasitic diseases, bacterial infections, and neurological disorders like Parkinson's disease. As the demand for novel active molecules grows to combat drug resistance, having access to a reliable, scalable synthesis for these scaffolds is paramount for maintaining a competitive pipeline in the pharmaceutical sector.
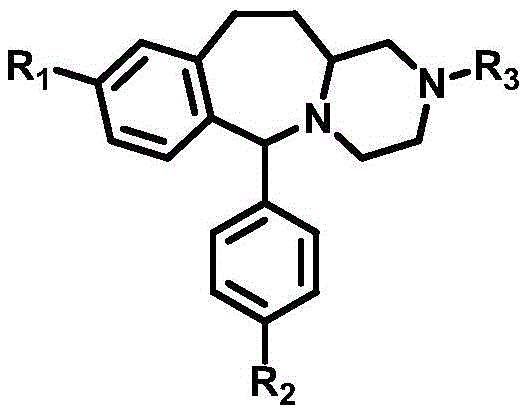
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzo-heterocyclic compounds like those described in patent CN101497612A has been plagued by significant safety and efficiency challenges. Traditional routes often employ sodium azide for ring expansion or construction steps, a reagent notorious for its high toxicity and potential to form explosive metal azides. This not only creates severe occupational health hazards but also complicates waste disposal and regulatory compliance for large-scale facilities. Furthermore, conventional cyclization strategies frequently utilize harsh conditions, such as high-temperature reactions with polyphosphoric acid, which can lead to substrate carbonization, complex impurity profiles, and disappointingly low yields, often hovering around 39.4% for key ring-forming steps. These factors collectively render older methods economically unviable and environmentally unsustainable for modern commercial manufacturing.
The Novel Approach
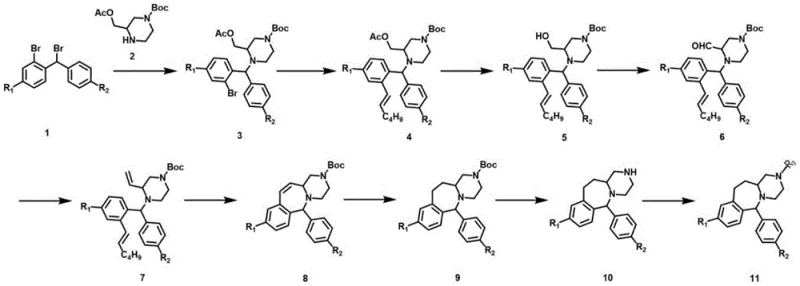
In stark contrast, the methodology disclosed in CN113754666A adopts a strategic divergence by constructing the benzhydrylpiperazine intermediate first, followed by a sophisticated ring-closing sequence. The cornerstone of this novel approach is the utilization of a diene precursor subjected to Ring-Closing Metathesis (RCM) using a Grubbs second-generation catalyst. This transition metal-catalyzed reaction proceeds under mild reflux conditions, typically in solvents like toluene or dichloroethane, avoiding the extreme thermal stress of previous methods. The result is a dramatic improvement in reaction selectivity and yield, with the key cyclization step achieving yields exceeding 60% in optimized examples. By eliminating sodium azide and harsh acidic cyclization, this route significantly lowers the E-factor of the process and enhances the overall safety profile, making it ideally suited for industrial scale-up.
Mechanistic Insights into Grubbs-Catalyzed Ring-Closing Metathesis
The mechanistic elegance of this synthesis lies in the formation of the seven-membered azepine ring via olefin metathesis. The process begins with the generation of a diene intermediate (Compound 7) through a sequence involving Suzuki coupling, deprotection, oxidation, and a Wittig reaction. This diene possesses two terminal olefinic groups strategically positioned to facilitate intramolecular cyclization. Upon exposure to the Grubbs second-generation catalyst, a ruthenium carbene complex initiates the reaction by coordinating with one of the olefins to form a metallacyclobutane intermediate. Through a series of [2+2] cycloaddition and cycloreversion steps, the catalyst mediates the exchange of alkylidene units, ultimately releasing ethylene gas and forging the new carbon-carbon double bond within the seven-membered ring structure (Compound 8). This mechanism is highly tolerant of various functional groups present on the piperazine and phenyl rings, ensuring high fidelity in the final product structure.
Impurity control is inherently managed through the specificity of the catalytic cycle and the mild reaction conditions employed. Unlike acid-catalyzed cyclizations that promote rearrangement and polymerization side reactions, the RCM pathway is clean and predictable. The use of a phosphine-ligated ruthenium catalyst ensures rapid initiation and propagation, minimizing the residence time of reactive intermediates that could otherwise degrade. Furthermore, the subsequent hydrogenation step (Compound 8 to 9) using platinum dioxide or palladium carbon effectively saturates the newly formed double bond, locking the conformation of the azepine ring and removing any residual olefinic impurities. This rigorous control over the reaction trajectory results in a final product with high purity, often exceeding 98%, which is critical for meeting the stringent specifications required for pharmaceutical intermediates.
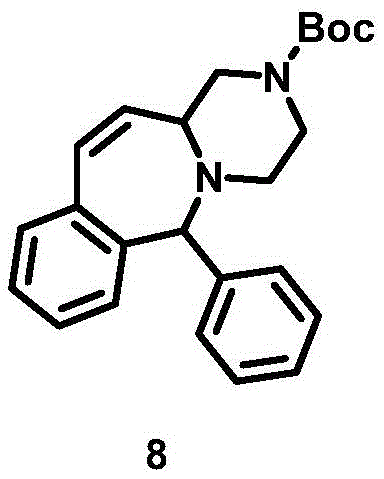
How to Synthesize 1,2,3,4,6,11,12,12a-octahydrobenzo[e]pyrazino[1,2-a]azepine Efficiently
The synthesis of this complex heterocyclic scaffold requires precise control over stoichiometry, temperature, and catalyst loading to maximize efficiency. The process initiates with a nucleophilic substitution to build the core piperazine framework, followed by a palladium-catalyzed cross-coupling to install the necessary carbon chain. Critical attention must be paid to the oxidation and Wittig steps to ensure the correct diene geometry for the subsequent metathesis. The detailed standardized synthesis steps, including specific solvent choices, molar ratios, and workup procedures for each of the nine transformations, are outlined below to guide process implementation.
- Perform nucleophilic substitution between a dibromo-benzene derivative and a protected piperazine to form the benzhydrylpiperazine intermediate.
- Execute a Suzuki coupling reaction with 1-alkenyl hexyl boric acid to introduce the olefinic side chain required for ring closure.
- Convert the alcohol to an aldehyde, perform a Wittig reaction to generate a diene, and finally utilize Grubbs II catalyst for Ring-Closing Metathesis to form the seven-membered azepine ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers substantial strategic benefits beyond mere technical feasibility. The elimination of sodium azide removes a major logistical and regulatory burden associated with the storage, handling, and disposal of explosive precursors. This simplification of the safety protocol translates directly into reduced operational overhead and lower insurance costs for manufacturing facilities. Additionally, the reliance on commercially available reagents such as boronic acids, Grubbs catalysts, and standard solvents ensures a stable and resilient supply chain, mitigating the risk of production delays caused by specialty chemical shortages. The robustness of the route allows for flexible sourcing of raw materials without compromising the quality of the final intermediate.
- Cost Reduction in Manufacturing: The significant improvement in reaction yields, particularly in the critical ring-closing step, leads to a drastic reduction in raw material consumption per kilogram of product. By avoiding low-yielding steps that generate excessive waste, the overall material cost is substantially lowered. Furthermore, the mild reaction conditions reduce energy consumption associated with heating and cooling, contributing to a more economical production process. The simplified purification requirements, driven by cleaner reaction profiles, also decrease the volume of chromatography media and solvents needed, further driving down the cost of goods sold.
- Enhanced Supply Chain Reliability: The synthetic route utilizes widely available starting materials and catalysts that are produced by multiple global suppliers, reducing dependency on single-source vendors. The stability of the intermediates allows for potential campaign manufacturing, where key precursors can be stockpiled safely, ensuring continuity of supply even during market fluctuations. This reliability is crucial for long-term contracts with pharmaceutical partners who require guaranteed delivery schedules for their clinical and commercial programs.
- Scalability and Environmental Compliance: Designed with industrial scale-up in mind, this process avoids the use of highly toxic reagents that would necessitate expensive containment systems and specialized waste treatment facilities. The aqueous workups and standard organic extractions are easily adaptable to large-scale reactors, facilitating a smooth transition from pilot plant to commercial tonnage production. The greener profile of the synthesis aligns with increasingly strict environmental regulations, future-proofing the manufacturing asset against tightening emissions and effluent standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these benzopyrazine derivatives. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on process capabilities and product specifications. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your specific drug development pipeline.
Q: Why is the Grubbs catalyst preferred for constructing the seven-membered ring in this synthesis?
A: Traditional methods often rely on high-temperature cyclization with polyphosphoric acid or toxic sodium azide, leading to low yields and safety hazards. The Grubbs II catalyst enables Ring-Closing Metathesis under mild conditions with high selectivity, significantly improving yield and operational safety.
Q: How does this new route address the safety concerns of prior art benzopyrazine synthesis?
A: Prior art methods frequently utilize sodium azide, a highly toxic and explosive reagent posing severe industrial risks. This patented route eliminates sodium azide entirely, replacing it with safer reagents like boronic acids and standard oxidants, making it suitable for large-scale commercial production.
Q: What are the potential therapeutic applications of these benzopyrazine derivatives?
A: These compounds serve as potent building blocks for drugs targeting parasitic infections (schistosomiasis, tapeworm), bacterial infections, and neurological disorders such as Parkinson's disease, acting as selective dopaminergic D1 agonists or antagonists.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzopyrazine Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality, complex intermediates for the development of next-generation therapeutics. Our team of expert process chemists has thoroughly analyzed the synthetic route disclosed in CN113754666A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art reactors capable of handling sensitive organometallic reactions, such as the Grubbs-catalyzed metathesis described herein, ensuring that every batch meets stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify the identity and purity of the benzopyrazine scaffold, guaranteeing consistency for your R&D and manufacturing needs.
We invite you to collaborate with us to leverage this innovative synthesis for your pharmaceutical projects. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing expertise can accelerate your timeline to market while optimizing your supply chain economics.
