Advanced Synthesis of Halogenated Conjugated Dienes for High-Value Agrochemical Intermediates
Advanced Synthesis of Halogenated Conjugated Dienes for High-Value Agrochemical Intermediates
The landscape of agrochemical intermediate manufacturing is constantly evolving, driven by the need for more efficient, scalable, and cost-effective synthetic routes. Patent CN111372914A introduces a significant advancement in this domain by disclosing a novel class of halogenated conjugated diene compounds and their versatile preparation methods. These compounds serve as critical building blocks for the synthesis of high-value biologically active molecules, most notably herbicides such as Pinoxaden. The innovation lies not only in the structural novelty of the halogenated intermediates but also in the robustness of the synthetic pathway, which allows for precise control over functional group installation. This technical breakthrough offers a reliable agrochemical intermediate supplier with the capability to deliver complex molecular architectures that were previously challenging to access with high purity and consistency.
At the core of this invention is the strategic design of Compound 1, a multifunctional halogenated conjugated diene that acts as a pivotal junction in the synthetic tree. The structural flexibility of this compound, defined by variable substituents R1 through R5 and functional groups Y1 and Y2, enables chemists to tailor the molecule for specific downstream applications. 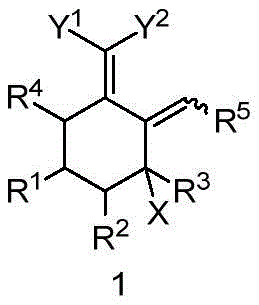 By leveraging this modular scaffold, manufacturers can access a diverse library of derivatives without needing to reinvent the wheel for each new target. The patent explicitly highlights the utility of these intermediates in producing final products with practical application value, positioning this technology as a cornerstone for modern herbicide manufacturing pipelines.
By leveraging this modular scaffold, manufacturers can access a diverse library of derivatives without needing to reinvent the wheel for each new target. The patent explicitly highlights the utility of these intermediates in producing final products with practical application value, positioning this technology as a cornerstone for modern herbicide manufacturing pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of arylmalonic acid derivatives, which are essential precursors for many herbicides, has relied on routes that often suffer from poor atom economy, harsh reaction conditions, or the generation of difficult-to-remove impurities. Conventional aromatization strategies might require high-energy inputs or expensive transition metal catalysts that introduce contamination risks, necessitating costly purification steps to meet stringent pharmaceutical or agrochemical grade specifications. Furthermore, older methods often lack the regioselectivity required to install halogen atoms at precise positions on the cyclohexene ring, leading to mixtures of isomers that complicate downstream processing. These inefficiencies translate directly into higher production costs and longer lead times, creating bottlenecks for procurement managers seeking to optimize their supply chains for cost reduction in herbicide manufacturing.
The Novel Approach
In contrast, the methodology outlined in CN111372914A offers a streamlined alternative that addresses these historical pain points through a clever two-stage sequence involving halogenation followed by dehydrohalogenation. By starting with a readily available conjugated diene precursor (Compound 2), the process utilizes common halogenating agents to install the necessary functionality under mild conditions. This approach minimizes the formation of side products and allows for the isolation of a stable, well-defined intermediate (Compound 1) that can be stored or transported before the final aromatization step. The ability to perform this transformation with high selectivity ensures that the resulting aromatic product (Compound 3) possesses the exact substitution pattern required for biological activity, thereby enhancing the overall yield and purity of the final agrochemical active ingredient.
Mechanistic Insights into Halogenation and Dehydrohalogenation Aromatization
The first stage of this synthetic route involves the electrophilic halogenation of the conjugated diene system. As illustrated in the reaction scheme below, the process converts the olefinic bonds of Compound 2 into the halogenated structure of Compound 1. 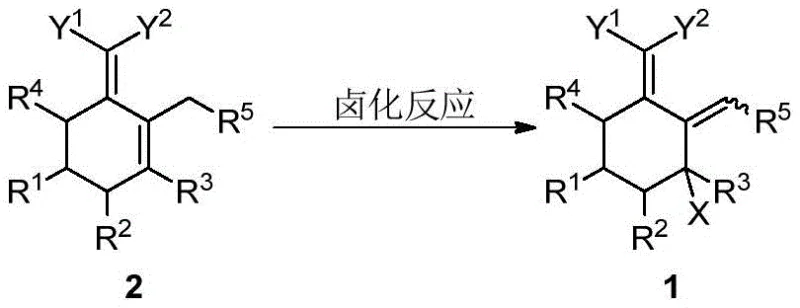 The mechanism likely proceeds through an electrophilic addition where the halogen source, whether it be chlorine gas, sulfuryl chloride, or hypohalous acids generated in situ, attacks the electron-rich double bond. The presence of electron-withdrawing groups such as cyano or carbonyl moieties at the exocyclic position influences the regioselectivity of this attack, directing the halogen to the allylic or vinylic position as defined by the specific R groups. This step is critical because it sets the stereochemical and constitutional foundation for the subsequent aromatization, ensuring that the halogen atom is positioned correctly to act as a leaving group in the next phase.
The mechanism likely proceeds through an electrophilic addition where the halogen source, whether it be chlorine gas, sulfuryl chloride, or hypohalous acids generated in situ, attacks the electron-rich double bond. The presence of electron-withdrawing groups such as cyano or carbonyl moieties at the exocyclic position influences the regioselectivity of this attack, directing the halogen to the allylic or vinylic position as defined by the specific R groups. This step is critical because it sets the stereochemical and constitutional foundation for the subsequent aromatization, ensuring that the halogen atom is positioned correctly to act as a leaving group in the next phase.
Following the halogenation, the second key transformation is the dehydrohalogenation aromatization, which converts the cyclic halogenated diene into a fully aromatic benzene derivative.  This step is catalyzed by simple alkali metal halides like lithium chloride or sodium chloride, which facilitate the elimination of hydrogen halide (HX) to restore aromaticity. The driving force for this reaction is the thermodynamic stability gained by forming the aromatic ring system. From an impurity control perspective, this mechanism is advantageous because the elimination is highly specific; the base-catalyzed removal of the proton adjacent to the halogen ensures that only the desired aromatic isomer is formed, significantly reducing the burden on downstream purification teams. This mechanistic clarity allows R&D directors to predict the impurity profile with high confidence, facilitating faster regulatory filings.
This step is catalyzed by simple alkali metal halides like lithium chloride or sodium chloride, which facilitate the elimination of hydrogen halide (HX) to restore aromaticity. The driving force for this reaction is the thermodynamic stability gained by forming the aromatic ring system. From an impurity control perspective, this mechanism is advantageous because the elimination is highly specific; the base-catalyzed removal of the proton adjacent to the halogen ensures that only the desired aromatic isomer is formed, significantly reducing the burden on downstream purification teams. This mechanistic clarity allows R&D directors to predict the impurity profile with high confidence, facilitating faster regulatory filings.
How to Synthesize Halogenated Conjugated Diene Efficiently
Implementing this synthesis in a commercial setting requires careful attention to reaction parameters such as temperature, solvent choice, and reagent stoichiometry to maximize yield and safety. The patent provides detailed embodiments demonstrating the versatility of the method across different scales and reagent systems, proving its adaptability for industrial production. For instance, the halogenation can be conducted in acetic acid at low temperatures (e.g., 15°C) using chlorine gas, or alternatively in THF using aqueous sodium hypochlorite with pH control. The subsequent aromatization typically requires heating the intermediate in polar aprotic solvents like N-methylpyrrolidone or chlorobenzene to temperatures between 110°C and 150°C.
- Perform halogenation of the conjugated diene starting material (Compound 2) using reagents such as chlorine gas, sulfuryl chloride, or sodium hypochlorite in solvents like acetic acid or THF at controlled low temperatures.
- Isolate the novel halogenated intermediate (Compound 1) or proceed directly in a one-pot fashion depending on the desired purity and downstream requirements.
- Execute dehydrohalogenation aromatization by heating the halogenated intermediate with a catalyst such as lithium chloride or sodium chloride in polar aprotic solvents like DMF to generate the final aromatic product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology represents a strategic opportunity to enhance supply security and reduce overall manufacturing costs. The reliance on commodity chemicals such as chlorine, sulfuryl chloride, and simple salts like lithium chloride means that the raw material supply chain is robust and less susceptible to the volatility associated with specialized reagents. This accessibility translates into significant cost reduction in herbicide manufacturing, as the input costs are stabilized and the need for expensive catalysts is eliminated. Furthermore, the simplicity of the workup procedures described in the examples, which often involve straightforward concentration and washing steps, reduces the consumption of solvents and energy, contributing to a leaner and more sustainable production model.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the use of inexpensive halogenating agents drastically lower the bill of materials for producing these key intermediates. By avoiding precious metals, manufacturers also save on the substantial costs associated with metal scavenging and waste disposal, which are often hidden expenses in traditional synthetic routes. The high selectivity of the reaction minimizes the loss of valuable starting materials to side reactions, ensuring that the theoretical yield is closely approached in practice, which directly improves the cost-per-kilogram metric for the final product.
- Enhanced Supply Chain Reliability: The use of widely available reagents like chlorine gas and acetic acid ensures that production is not held hostage by the supply constraints of niche chemicals. This reliability is crucial for maintaining continuous operation of large-scale plants, reducing the risk of production stoppages due to raw material shortages. Additionally, the stability of the intermediate Compound 1 allows for flexible scheduling; it can be produced in batches and stored, decoupling the halogenation and aromatization steps to optimize plant throughput and manage inventory levels more effectively.
- Scalability and Environmental Compliance: The reaction conditions described, such as moderate temperatures and the absence of highly toxic reagents, make this process highly amenable to scale-up from pilot plant to commercial tonnage. The ability to control exotherms during the halogenation step, as demonstrated in the examples, ensures safe operation at scale. Moreover, the generation of simpler waste streams, primarily consisting of salt byproducts and recoverable solvents, simplifies wastewater treatment and aligns with increasingly strict environmental regulations, reducing the compliance burden on the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential partners.
Q: What are the preferred halogenating agents for this synthesis?
A: The patent specifies several effective halogenating agents including elemental halogens like chlorine gas or liquid bromine, as well as sulfonyl halides such as sulfuryl chloride. Sodium hypochlorite combined with hydrochloric acid is also a viable option for generating the active halogen species in situ.
Q: How is the aromatization step catalyzed effectively?
A: The dehydrohalogenation aromatization reaction is facilitated by alkali metal halides or alkaline earth metal halides. Specifically, lithium chloride and sodium chloride are highlighted as preferred catalysts, operating effectively at temperatures ranging from 110°C to 150°C in solvents like N-methylpyrrolidone or chlorobenzene.
Q: Can this method be applied to Pinoxaden synthesis?
A: Yes, the resulting 2-arylmalonic acid derivatives obtained from this process are key precursors for synthesizing biologically active compounds, specifically the herbicide Pinoxaden (8-(2,6-diethyl-4-methylphenyl)-7-oxo-1,2,4,5-tetrahydro-7H-pyrazolo[1,2-d][1,4,5]oxadiazepin-9-yl pivalate).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogenated Conjugated Diene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic routes disclosed in CN111372914A for the production of next-generation agrochemicals. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry from the laboratory bench to full-scale manufacturing. Our facilities are equipped with the rigorous QC labs and analytical capabilities necessary to ensure stringent purity specifications for every batch of halogenated intermediates we produce, guaranteeing that your downstream synthesis of herbicides like Pinoxaden proceeds without interruption or quality issues.
We invite you to collaborate with us to leverage this innovative technology for your supply chain needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how switching to this route can optimize your bottom line. Please contact our technical procurement team today to request specific COA data for our pilot batches and to discuss route feasibility assessments for your upcoming projects, ensuring a seamless transition to this superior manufacturing process.
