Advanced Synthesis of Annona Lactone Analogues for Oncology Drug Development
The development of potent anticancer agents often hinges on the ability to synthesize complex natural product scaffolds efficiently. Patent CN1109678C introduces a groundbreaking class of annonaceolide analogues that retain the high biological activity of natural Annonaceae acetogenins while significantly simplifying their molecular architecture. Traditional natural products in this class are characterized by long carbon chains containing multiple tetrahydrofuran (THF) rings and numerous chiral centers, which pose immense challenges for total synthesis and commercial manufacturing. This patent proposes a strategic structural modification where the complex THF segments are replaced by polyethylene glycol (PEG) chains or sugar fragments. This innovation preserves the critical oxygen-containing functional groups responsible for ion-coordination and cell membrane penetration, which are hypothesized to be the mechanism of action for tumor cell death, while removing the stereochemical complexity that hinders large-scale production. For pharmaceutical developers, this represents a pivotal shift towards more accessible and cost-effective oncology intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of natural annonaceolides has been plagued by extreme stereochemical complexity. Natural molecules typically contain five to ten chiral centers and one or two tetrahydrofuran rings arranged in specific configurations. Constructing these THF rings with high diastereoselectivity requires lengthy sequences of protecting group manipulations, cyclizations, and purifications. Furthermore, the absolute configuration of these natural products is often difficult to determine directly, leading to uncertainties in synthetic planning. The reliance on extracting these compounds from natural sources is equally problematic due to their extremely low content in plant tissues, making it impossible to secure sufficient quantities for clinical development or commercial drug supply. Consequently, conventional routes suffer from low overall yields, high material costs, and significant batch-to-batch variability, rendering them unsuitable for the reliable supply chains required by the global pharmaceutical industry.
The Novel Approach
The methodology described in the patent circumvents these bottlenecks by decoupling biological activity from structural complexity. By substituting the THF rings with linear polyether chains derived from polyethylene glycol, the synthesis eliminates multiple chiral centers in that segment of the molecule. This simplification allows for the use of robust, high-yielding reactions such as aldol condensations and etherifications. A key feature of this streamlined route is the utilization of mild oxidation protocols to install necessary carbonyl functionalities without compromising sensitive protecting groups. 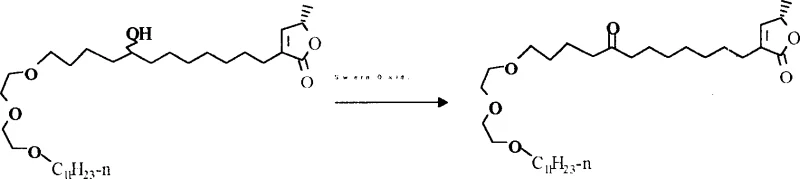 As illustrated in the reaction scheme, the conversion of hydroxyl groups to ketones using Swern oxidation conditions (DMSO/Oxalyl Chloride) proceeds efficiently at low temperatures. This approach avoids the harsh conditions often associated with traditional oxidants, thereby minimizing side reactions and improving the purity profile of the intermediate. The result is a synthetic pathway that is not only shorter but also far more predictable and scalable for industrial applications.
As illustrated in the reaction scheme, the conversion of hydroxyl groups to ketones using Swern oxidation conditions (DMSO/Oxalyl Chloride) proceeds efficiently at low temperatures. This approach avoids the harsh conditions often associated with traditional oxidants, thereby minimizing side reactions and improving the purity profile of the intermediate. The result is a synthetic pathway that is not only shorter but also far more predictable and scalable for industrial applications.
Mechanistic Insights into Chiral Lactone and Polyether Coupling
The core of this synthetic strategy relies on the precise assembly of two distinct fragments: a chiral gamma-butyrolactone head and a polyether tail. The lactone fragment is synthesized from enantiopure lactic acid derivatives, ensuring that the critical chirality at the C4 position of the butenolide ring is established early and maintained throughout the sequence. The process involves the generation of a chiral aldehyde via LDA reduction followed by Swern oxidation, which then undergoes an aldol-type reaction with the enolate of a long-chain unsaturated ester. This step constructs the carbon backbone with high fidelity. Subsequent protection and deprotection steps reveal the lactone ring, which is then functionalized to an epoxide. 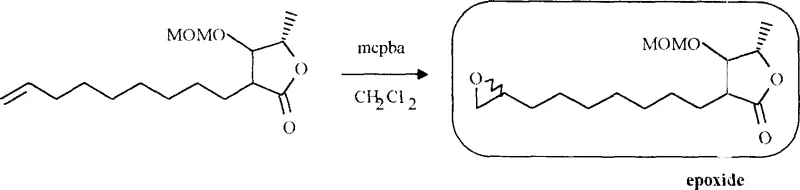 The epoxidation step, utilizing meta-chloroperoxybenzoic acid (mcpba) as shown in the scheme, is crucial for activating the side chain for the final coupling event. The epoxide serves as an electrophilic handle that can be opened by nucleophilic attack, allowing for the attachment of the polyether chain. This mechanistic design ensures that the oxygen-rich pharmacophore is correctly positioned relative to the lactone warhead, mimicking the spatial arrangement found in the natural bioactive conformation.
The epoxidation step, utilizing meta-chloroperoxybenzoic acid (mcpba) as shown in the scheme, is crucial for activating the side chain for the final coupling event. The epoxide serves as an electrophilic handle that can be opened by nucleophilic attack, allowing for the attachment of the polyether chain. This mechanistic design ensures that the oxygen-rich pharmacophore is correctly positioned relative to the lactone warhead, mimicking the spatial arrangement found in the natural bioactive conformation.
Impurity control is inherently built into this mechanism through the use of defined starting materials and selective reagents. Unlike natural extraction, which yields a mixture of congeners, this chemical synthesis produces a single, well-defined molecular entity. The use of Lewis acid catalysis during the coupling of the alkynyl lithium reagent with the epoxy intermediate ensures regioselective ring opening, preventing the formation of unwanted regioisomers. Furthermore, the final elimination step using DBU (1,8-Diazabicyclo[5.4.0]undec-7-ene) cleanly generates the alpha,beta-unsaturated lactone system without affecting the ether linkages in the tail. This high level of chemoselectivity reduces the burden on downstream purification, resulting in a final product with a superior impurity profile that meets the stringent requirements for pharmaceutical intermediates intended for preclinical and clinical evaluation.
How to Synthesize Annona Lactone Analogues Efficiently
The synthesis of these high-value anticancer intermediates follows a convergent strategy that maximizes yield and minimizes operational complexity. The process begins with the preparation of the chiral lactone epoxide and the polyether alkyne fragment in parallel, which are then united in the final stages. This modular approach allows for the optimization of each fragment independently before the final coupling. The detailed standardized synthesis steps, including specific stoichiometry, temperature controls, and workup procedures for each transformation, are outlined in the guide below to ensure reproducibility and safety during scale-up operations.
- Preparation of chiral epoxy intermediates containing gamma-butyrolactone via THP protection, LDA reduction, Swern oxidation, and aldol reaction followed by epoxidation.
- Synthesis of alkynyl lithium reagents from polyethylene glycol or sugar fragments via dipropargyl etherification and monolithiation.
- Coupling of the alkyne reagent with the epoxy intermediate under Lewis acid catalysis, followed by hydrogenation and DBU elimination to yield the final analogue.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from natural extraction or complex total synthesis to this simplified analogue route offers substantial strategic benefits. The primary advantage lies in the drastic reduction of process steps and the elimination of low-yielding stereochemical constructions. By replacing the difficult-to-synthesize THF rings with commercially abundant polyethylene glycols, the raw material costs are significantly lowered. Additionally, the reliance on commodity chemicals rather than rare natural extracts insulates the supply chain from agricultural volatility, seasonal variations, and geopolitical risks associated with sourcing plant materials from specific tropical regions. This stability is critical for maintaining continuous production schedules for downstream drug manufacturing.
- Cost Reduction in Manufacturing: The streamlined synthetic route eliminates the need for expensive chiral catalysts or resolution steps associated with constructing multiple THF rings. The use of standard reagents like LDA, mcpba, and DBU, which are available in bulk at competitive prices, further drives down the cost of goods sold (COGS). Moreover, the higher overall yield resulting from fewer purification steps means less solvent consumption and waste generation, leading to significant operational expenditure savings. The removal of transition metal catalysts in favor of organolithium and Lewis acid chemistry also simplifies the purification process, avoiding the costly and time-consuming heavy metal scavenging steps often required in palladium-catalyzed couplings.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as ethyl lactate and various grades of polyethylene glycol, are produced on a massive global scale by multiple suppliers. This commoditization ensures that there is no single point of failure in the raw material supply chain. Unlike natural products which may face supply shortages due to crop failures or regulatory restrictions on plant harvesting, these synthetic precursors offer guaranteed availability. This reliability allows for accurate long-term capacity planning and reduces the risk of production delays that could impact the timelines of critical oncology drug development programs.
- Scalability and Environmental Compliance: The reactions employed in this patent, including reductions, oxidations, and eliminations, are well-understood unit operations that translate easily from laboratory glassware to industrial reactors. The process avoids the use of highly toxic reagents or extreme conditions that would require specialized containment equipment. Furthermore, the simplified structure results in a cleaner reaction profile with fewer byproducts, facilitating easier wastewater treatment and waste disposal. This alignment with green chemistry principles not only reduces environmental compliance costs but also enhances the sustainability profile of the manufacturing process, a key metric for modern pharmaceutical supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these annonaceolide analogues. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on the feasibility and performance of this synthetic technology.
Q: Why simplify the tetrahydrofuran ring in natural annonaceolides?
A: Natural annonaceolides possess complex tetrahydrofuran rings with multiple chiral centers (5-10), making total synthesis arduous and low-yielding. The patented approach replaces these with polyethylene glycol chains, retaining the oxygen coordination necessary for ion-carrying biological activity while eliminating difficult-to-control chiral centers, thus drastically simplifying production.
Q: What is the biological activity profile of these analogues?
A: According to National Cancer Institute (NCI) data cited in the patent, specific analogues such as 2-(8'-hydroxy-13',16',19'22'-tetraoxa)-triacontyl-4-methyl-butene-2-lactone exhibit potent cytotoxicity against leukemia, lung cancer, and melanoma cell lines, with GI50 values in the nanomolar range, comparable to or exceeding natural variants.
Q: Are the raw materials for this synthesis commercially scalable?
A: Yes, the synthesis utilizes readily available starting materials such as enantiopure lactate esters and polyethylene glycols. The avoidance of exotic natural extraction and the use of standard organic transformations like Swern oxidation and aldol condensation ensure that the process is highly amenable to commercial scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Annona Lactone Analogue Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex academic innovations into commercial reality. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we possess the technical expertise to master the chiral synthesis and polyether functionalization required for these potent anticancer intermediates. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch meets stringent purity specifications, guaranteeing the consistency required for clinical trials and eventual market launch. We understand the critical nature of oncology supply chains and are committed to delivering high-quality intermediates that accelerate your drug development timeline.
We invite research and procurement leaders to engage with our technical team to discuss how this simplified synthetic route can optimize your project economics. By leveraging our manufacturing capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples to evaluate the quality of our Annona Lactone Analogues for your next generation of anticancer therapeutics.
