Advanced Chiral Synthesis of Piperidine Intermediates for Pharmaceutical Manufacturing
Advanced Chiral Synthesis of Piperidine Intermediates for Pharmaceutical Manufacturing
The pharmaceutical industry's relentless pursuit of potent neurokinin-1 (NK1) receptor antagonists has placed significant demand on the supply chain for high-purity chiral intermediates. Patent CN1189458C introduces a transformative synthetic methodology for producing (2S,3S)-1-tert-butoxycarbonyl-3-hydroxy-2-phenylpiperidine, a critical precursor for the synthesis of L-733060 and related Substance P antagonists. Unlike conventional approaches that rely on the resolution of racemic mixtures, this innovation leverages the inherent chirality of L-glutamic acid, a ubiquitous and cost-effective natural amino acid. By anchoring the stereochemistry early in the synthesis, the process achieves exceptional enantiomeric selectivity while streamlining the operational workflow. For R&D directors and procurement strategists, this represents a pivotal shift towards more sustainable and economically viable manufacturing pathways for complex heterocyclic scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-phenylpiperidine derivatives relied heavily on the condensation of benzaldehyde with methyl 4-nitrobutyrate, a pathway famously disclosed in earlier European patents. This traditional route fundamentally suffers from a lack of stereocontrol, inevitably generating a racemic mixture of cis and trans isomers. As illustrated in the reaction scheme below, the process necessitates a cumbersome resolution step using chiral acids like dibenzoyltartrate to isolate the desired enantiomer. 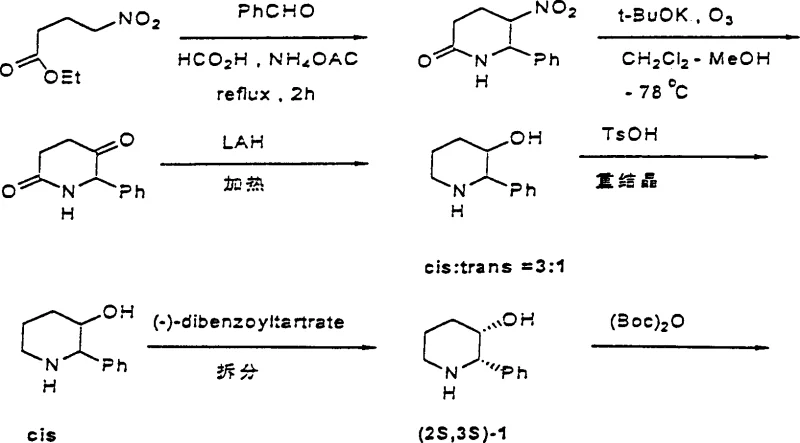 This intrinsic inefficiency results in a theoretical maximum yield loss of nearly 50% for the unwanted enantiomer, drastically inflating the cost of goods sold (COGS). Furthermore, the reliance on nitro compounds and ozonolysis introduces significant safety hazards and environmental burdens, complicating waste management and regulatory compliance for large-scale facilities.
This intrinsic inefficiency results in a theoretical maximum yield loss of nearly 50% for the unwanted enantiomer, drastically inflating the cost of goods sold (COGS). Furthermore, the reliance on nitro compounds and ozonolysis introduces significant safety hazards and environmental burdens, complicating waste management and regulatory compliance for large-scale facilities.
The Novel Approach
In stark contrast, the methodology detailed in CN1189458C circumvents these bottlenecks by utilizing L-glutamic acid as the chiral template. This chiral pool strategy ensures that the stereochemical integrity is maintained throughout the synthetic sequence, effectively bypassing the need for late-stage resolution. The novel route replaces hazardous ozonolysis with safer reduction and cyclization protocols, utilizing common reagents such as thionyl chloride, borohydrides, and Lewis acids. By eliminating the resolution step and avoiding the handling of unstable nitro intermediates, the new approach offers a streamlined pathway that is inherently safer and more atom-economical. This transition from racemic synthesis to asymmetric construction marks a significant technological upgrade for manufacturers seeking to optimize their supply chains for NK1 antagonist production.
Mechanistic Insights into L-Glutamic Acid Derived Cyclization
The core of this synthetic innovation lies in the precise manipulation of the glutamic acid backbone to construct the piperidine ring while preserving the chiral center. The process initiates with the diazotization of L-glutamic acid, converting the alpha-amino group into a hydroxyl functionality which subsequently cyclizes to form a lactone intermediate. This step is critical as it locks the stereochemistry derived from the natural amino acid. Subsequent amidation with p-methoxybenzylamine and base-mediated intramolecular Claisen-type condensation forge the six-membered piperidinedione core. The use of strong bases like LDA or potassium tert-butoxide at cryogenic temperatures ensures kinetic control, preventing epimerization at the alpha-position. This meticulous control over reaction conditions is paramount for maintaining the high diastereomeric ratio required for downstream efficacy.
Furthermore, the introduction of the phenyl group and the final reduction steps are engineered to minimize racemization risks. The protocol employs a Lewis acid-mediated transformation to install the phenyl moiety, followed by a selective reduction of the lactam carbonyl using lithium aluminum hydride or borane complexes. The final Boc-protection step is conducted under mild hydrogenation conditions, ensuring the stability of the sensitive hydroxyl group. From an impurity control perspective, this route is superior because the starting material's purity directly dictates the product's optical purity, reducing the complexity of the impurity profile. For quality assurance teams, this means fewer unknown peaks in HPLC chromatograms and a more robust validation process for the drug master file (DMF).
How to Synthesize (2S,3S)-1-tert-butoxycarbonyl-3-hydroxy-2-phenylpiperidine Efficiently
The synthesis of this high-value intermediate involves a multi-step sequence that transforms a commodity amino acid into a complex heterocycle. The process is designed for scalability, utilizing standard unit operations such as extraction, crystallization, and column chromatography where necessary. Each step has been optimized to balance yield with purity, ensuring that the cumulative loss across the eight-step sequence is minimized. The following guide outlines the critical operational phases required to execute this synthesis effectively in a GMP environment.
- Convert L-glutamic acid to the corresponding lactone derivative via diazotization with sodium nitrite and sulfuric acid.
- Perform amidation with p-methoxybenzylamine followed by base-mediated cyclization to form the piperidinedione core.
- Execute stereoselective reduction and Lewis acid-mediated phenylation to establish the final chiral piperidine scaffold.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this L-glutamic acid-based route offers compelling strategic advantages beyond mere technical elegance. The shift away from resolution-dependent processes fundamentally alters the cost structure of the intermediate. By removing the need for expensive chiral resolving agents and discarding half of the produced material, the effective throughput of the manufacturing plant is nearly doubled without increasing reactor volume. This efficiency gain translates directly into substantial cost savings, allowing pharmaceutical companies to negotiate more favorable pricing tiers with their CDMO partners. Additionally, the reliance on L-glutamic acid, a fermentation-derived commodity available in massive global quantities, insulates the supply chain from the volatility often associated with specialized petrochemical precursors.
- Cost Reduction in Manufacturing: The elimination of the resolution step is the primary driver for cost optimization in this process. In traditional racemic syntheses, the cost of the discarded enantiomer and the resolving agent must be amortized over the final product, significantly inflating the price. By achieving high stereoselectivity from the start, this novel route removes that financial burden entirely. Furthermore, the avoidance of ozonolysis reduces the capital expenditure required for specialized safety equipment and lowers operational costs related to hazard mitigation. The use of common solvents and reagents also simplifies procurement logistics, reducing lead times and inventory holding costs for the manufacturing facility.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit immensely from the use of L-glutamic acid as the starting material. Unlike specialized nitro-compounds or chiral catalysts that may have single-source suppliers, L-glutamic acid is produced globally by multiple manufacturers for the food and feed industries. This abundance ensures a stable and continuous supply, mitigating the risk of production stoppages due to raw material shortages. Moreover, the simplified synthetic route reduces the number of critical intermediates that need to be stocked, streamlining inventory management. For supply chain planners, this means greater predictability in delivery schedules and a reduced risk of bottlenecks during scale-up campaigns.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns perfectly with modern green chemistry principles. The removal of nitro groups from the synthesis eliminates the generation of nitrogenous waste streams, which are costly and difficult to treat. The replacement of ozonolysis with safer reduction methods reduces the energy footprint and safety risks associated with handling explosive ozone gas. These factors facilitate easier regulatory approval and permit acquisition for manufacturing sites. As pharmaceutical companies face increasing pressure to reduce their carbon footprint, adopting a synthesis that minimizes hazardous waste and energy consumption provides a significant competitive advantage in sustainability reporting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. Understanding these details is crucial for cross-functional teams evaluating the feasibility of integrating this intermediate into their broader API manufacturing strategies. The answers are derived directly from the technical specifications and experimental data provided in the patent literature.
Q: What is the primary advantage of using L-glutamic acid over traditional nitro-aldol routes?
A: Using L-glutamic acid as a chiral pool starting material eliminates the need for resolution of racemic mixtures, significantly improving overall yield and optical purity compared to traditional condensation methods.
Q: How does this process impact the impurity profile of the final API?
A: The chiral pool approach ensures high enantiomeric excess (up to 97% ee in downstream steps) from the outset, reducing the burden on downstream purification and minimizing genotoxic impurities associated with nitro compounds.
Q: Is this synthesis scalable for commercial production?
A: Yes, the process utilizes common reagents and avoids hazardous conditions like ozonation found in prior art, making it highly suitable for multi-kilogram to ton-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2S,3S)-1-tert-butoxycarbonyl-3-hydroxy-2-phenylpiperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals like L-733060 depends on a robust and technically proficient supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory bench to industrial reactor. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee the optical purity and chemical integrity of every batch. Our commitment to technical excellence means we can navigate the nuances of chiral pool synthesis to deliver intermediates that meet the exacting standards of global regulatory bodies.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can optimize your project economics. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage potential partners to request specific COA data and route feasibility assessments to verify our capabilities firsthand. Let us collaborate to secure a reliable supply of high-purity intermediates, driving your drug development program forward with confidence and efficiency.
