Scalable Manufacturing of Protected 1,8-Diazaspiro[4.5]decane via Novel Hydrogenation Strategy
The pharmaceutical industry's relentless pursuit of potent orexin receptor antagonists has placed significant demand on the supply chain for high-quality heterocyclic scaffolds, specifically the 1,8-diazaspiro[4.5]decane core structure. As detailed in patent CN102070633A, a groundbreaking synthetic methodology has been developed that fundamentally alters the economic and technical landscape for producing these critical intermediates. This innovation addresses the longstanding bottlenecks of traditional synthesis, which have historically plagued manufacturers with low yields, hazardous reagents, and complex purification protocols. By shifting the paradigm towards a streamlined Michael addition followed by a catalytic hydrogenation-cyclization sequence, this technology offers a robust pathway for generating protected diazaspiro compounds with exceptional efficiency. For R&D directors and procurement strategists alike, understanding this shift is crucial for securing a reliable pharmaceutical intermediate supplier capable of meeting the rigorous demands of modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,8-diazaspiro[4.5]decane skeleton has been fraught with significant technical and economic challenges that hindered mass production capabilities. As illustrated in prior art such as WO2007/025069A1, traditional routes often relied on multi-step sequences involving highly toxic reagents like sodium cyanide, posing severe safety risks and environmental compliance burdens for manufacturing facilities. Furthermore, these legacy processes frequently necessitated extreme reaction conditions, including cryogenic temperatures as low as -78°C, which drastically increased energy consumption and required specialized, expensive equipment. The reliance on precious metal catalysts like palladium in complex coupling steps not only inflated raw material costs but also introduced difficulties in removing trace metal impurities to meet stringent pharmaceutical standards. Consequently, the overall yields remained suboptimal, and the purification of intermediates was often labor-intensive, rendering these methods economically unviable for large-scale commercial application.
The Novel Approach
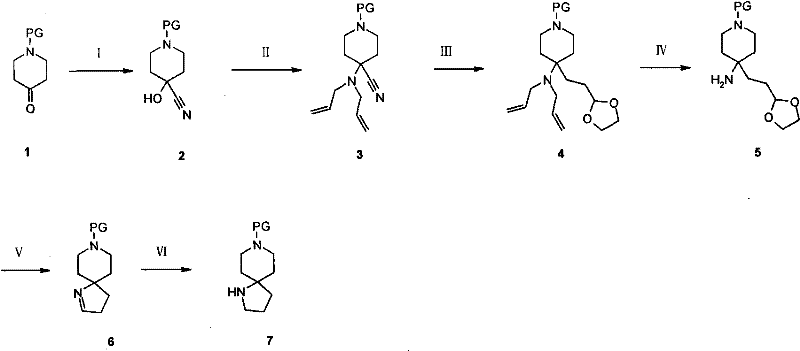
In stark contrast to these cumbersome legacy methods, the novel approach disclosed in the patent introduces a remarkably concise and efficient synthetic strategy that leverages readily available starting materials. The core innovation lies in the utilization of N-PG-4-nitropiperidine derivatives, which undergo a base-catalyzed Michael addition with acrolein to form a key aldehyde intermediate. This is followed by a elegant one-pot hydrogenation and ring-closing reaction, typically employing cost-effective catalysts like Raney Nickel under mild pressure and temperature conditions. This strategic simplification eliminates the need for hazardous cyanide chemistry and cryogenic operations, thereby streamlining the entire production workflow. The result is a process that not only achieves superior overall yields but also significantly reduces the operational complexity, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing while maintaining high product integrity.
Mechanistic Insights into Reductive Cyclization and Michael Addition
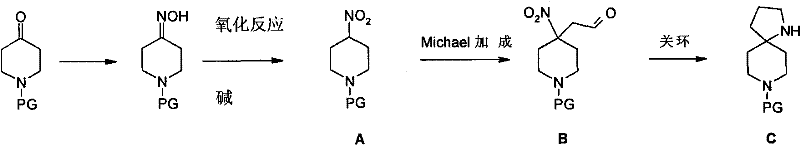
The mechanistic elegance of this new pathway is rooted in the precise control of chemoselectivity during the transformation of the nitro-aldehyde precursor into the final spirocyclic amine. The initial Michael addition step involves the deprotonation of the alpha-position of the nitropiperidine by a mild organic base, such as triethylamine or DIPEA, generating a nucleophilic species that attacks the beta-carbon of acrolein. This step is critical as it installs the necessary three-carbon chain bearing the aldehyde functionality required for the subsequent cyclization. Careful optimization of the solvent system, typically acetonitrile or alcohols, ensures that polymerization of the acrolein is minimized, thereby maximizing the conversion to the desired linear intermediate. The reaction proceeds smoothly at ambient temperatures, avoiding the thermal degradation issues often associated with more aggressive alkylation methods.
Following the Michael addition, the reductive cyclization represents the pivotal transformation where the spiro-center is established. Under hydrogen atmosphere in the presence of a heterogeneous catalyst like Raney Nickel, the nitro group is selectively reduced to a primary amine. This newly formed amine then spontaneously undergoes an intramolecular condensation with the pendant aldehyde group to form an imine, which is immediately reduced in situ to the secondary amine, locking the spiro[4.5]decane structure. This tandem reduction-cyclization mechanism is highly advantageous because it avoids the isolation of unstable imine intermediates and drives the equilibrium towards the thermodynamically stable cyclic product. From an impurity control perspective, the use of Raney Nickel is particularly beneficial as it effectively suppresses side reactions such as over-reduction of the piperidine ring or incomplete cyclization, ensuring a clean crude profile that simplifies downstream purification efforts.
How to Synthesize N-PG-1,8-Diazaspiro[4.5]decane Efficiently
The execution of this synthesis requires careful attention to reaction parameters to ensure optimal conversion and safety, particularly during the oxidation and hydrogenation stages. The process begins with the conversion of commercially available N-protected-4-piperidone into its corresponding oxime, followed by oxidation to the nitro compound using reagents like m-CPBA or peracetic acid. Once the nitro intermediate is secured, the Michael addition with acrolein is performed under inert atmosphere to prevent polymerization, yielding the key aldehyde precursor. The final step involves charging the reactor with the aldehyde intermediate and the chosen catalyst, pressurizing with hydrogen, and maintaining moderate temperatures to effect the ring closure. For a comprehensive breakdown of the specific stoichiometric ratios, solvent choices, and workup procedures validated in the patent examples, please refer to the standardized protocol below.
- Convert N-PG-4-piperidone to the corresponding oxime using hydroxylamine hydrochloride in alcoholic solution.
- Oxidize the oxime intermediate to N-PG-4-nitropiperidine using oxidants like m-CPBA or peracetic acid.
- Perform a Michael addition reaction between the nitropiperidine and acrolein in the presence of a base catalyst.
- Execute a one-pot hydrogenation and ring-closing reaction using Raney Nickel to form the final spiro cycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route translates directly into tangible operational efficiencies and risk mitigation strategies. By replacing hazardous reagents and extreme conditions with benign alternatives, the process inherently lowers the barrier to entry for manufacturing partners, expanding the pool of qualified suppliers and enhancing supply chain resilience. The elimination of cryogenic steps and toxic cyanide waste streams significantly reduces the environmental footprint and associated disposal costs, aligning with modern green chemistry initiatives and regulatory compliance standards. Furthermore, the use of robust, non-precious metal catalysts like Raney Nickel minimizes exposure to volatile commodity prices for noble metals, providing greater stability in long-term cost forecasting for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The streamlined nature of this process, which consolidates multiple transformations into fewer steps, inherently drives down manufacturing costs by reducing labor hours, solvent consumption, and energy usage. The avoidance of expensive palladium catalysts and the ability to operate at ambient or moderately elevated temperatures rather than cryogenic conditions results in substantial utility savings. Additionally, the simplified workup procedures, often requiring only filtration and concentration, minimize the need for complex chromatographic purifications, further enhancing the economic viability of the process for large-volume production.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as acrolein, acetonitrile, and standard protecting group reagents ensures that raw material sourcing remains stable and unaffected by niche supply disruptions. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process is less sensitive to minor fluctuations in temperature or pressure compared to the finicky prior art methods. This reliability is crucial for maintaining continuous production runs and meeting tight delivery timelines for downstream API synthesis, effectively reducing lead times for complex pharmaceutical intermediates.
- Scalability and Environmental Compliance: The transition from batch processes requiring specialized low-temperature equipment to standard hydrogenation reactors facilitates seamless scale-up from pilot plant to commercial tonnage. The reduction in hazardous waste generation, particularly the elimination of cyanide-containing effluents, simplifies wastewater treatment requirements and lowers the regulatory burden on manufacturing sites. This environmental compatibility not only future-proofs the supply chain against tightening regulations but also enhances the corporate sustainability profile of the final drug product, a key consideration for modern pharmaceutical stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the practical advantages of the new route over existing methodologies, focusing on safety, scalability, and product quality. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this intermediate into their broader drug substance manufacturing strategies.
Q: How does this new method improve safety compared to conventional routes?
A: The novel route eliminates the use of highly toxic sodium cyanide and avoids cryogenic conditions (-78°C) required in prior art, significantly enhancing operational safety and reducing hazardous waste disposal costs.
Q: What catalysts are suitable for the final cyclization step?
A: The patent specifies the use of cost-effective heterogeneous catalysts such as Raney Nickel, palladium on carbon, or platinum dioxide, with Raney Nickel being particularly effective for large-scale operations.
Q: Is this process suitable for multi-ton commercial production?
A: Yes, the process features mild reaction conditions (25-65°C, 10-55 psi), simple workup procedures involving filtration, and high overall yields, making it highly amenable to industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,8-Diazaspiro[4.5]decane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel therapeutics depends heavily on the availability of high-quality, cost-effective building blocks. Our technical team has extensively analyzed the pathway described in CN102070633A and possesses the expertise to implement this advanced hydrogenation-cyclization strategy at scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of 1,8-diazaspiro[4.5]decane derivative meets the exacting standards required for clinical and commercial API manufacturing.
We invite you to collaborate with us to leverage these process improvements for your specific project requirements. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this superior synthetic route for your supply chain. We encourage you to reach out today to discuss your specific volume requirements, obtain specific COA data for our reference standards, and review detailed route feasibility assessments tailored to your development timeline.
