Advanced Stereoselective Synthesis of Pine Sawfly Sex Pheromone Intermediates for Commercial Scale-Up
The development of highly effective pest control agents relies heavily on the precise synthesis of insect sex pheromones, where stereochemistry dictates biological activity. Patent CN1233608C introduces a robust and economically viable method for synthesizing high enantiomeric pure pine sawfly sex pheromones, specifically targeting the (2S, 3S, 7S)-3,7-dimethyl-2-pentadecanol ester series. This technology leverages natural (S)-malic acid as a chiral starting material, bypassing the need for expensive asymmetric catalysis or tedious resolution steps that have plagued previous generations of pheromone synthesis. By utilizing a chiral pool strategy, the process ensures that the critical stereocenters at positions 2, 3, and 7 are established with high fidelity from the outset. This approach not only guarantees the biological efficacy of the final product but also streamlines the manufacturing workflow, making it an ideal candidate for industrial scale-up. For R&D teams focused on agrochemical intermediates, this pathway represents a significant advancement in balancing stereochemical precision with process efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pine sawfly sex pheromones has been fraught with challenges related to step count, yield, and stereochemical control. Early methodologies, such as those reported by the Mori research group in the late 1970s, relied on starting materials like (R)-(+)-geraniol or tartrates, which often necessitated long synthetic sequences involving organocopper lithium reagents and epoxide ring openings. These routes were not only lengthy but also suffered from moderate overall yields due to the accumulation of losses over multiple steps. Furthermore, other approaches utilized chiral auxiliaries or asymmetric hydrogenation with nickel catalysts, which introduced additional complexity regarding catalyst removal and metal contamination. Some methods even employed sulfur-containing compounds that posed significant environmental disposal issues, conflicting with modern green chemistry principles. The reliance on racemic mixtures followed by resolution further exacerbated cost inefficiencies, as half of the synthesized material was often discarded as the unwanted enantiomer.
The Novel Approach
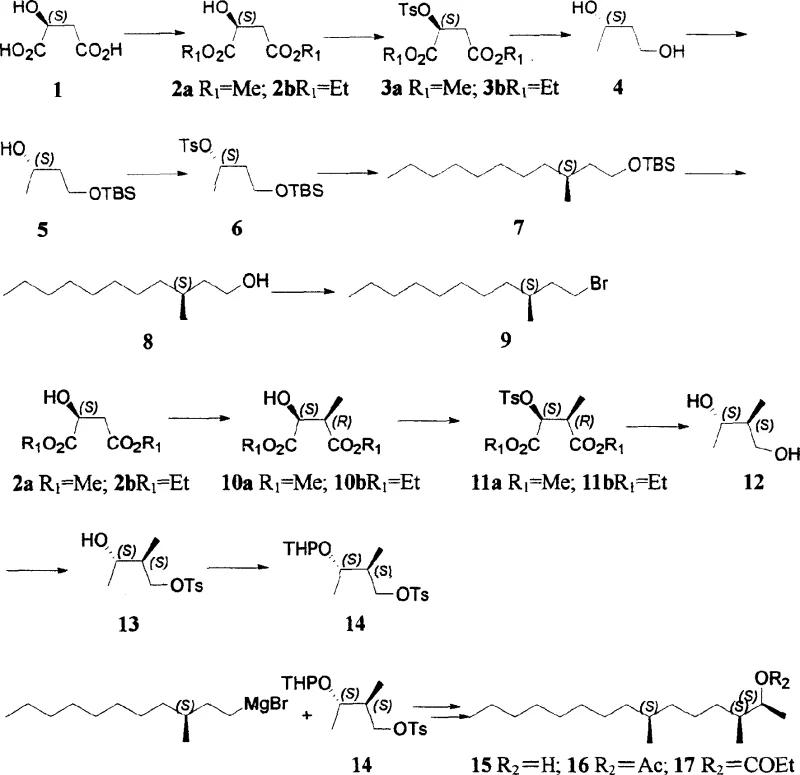
In stark contrast, the methodology disclosed in CN1233608C offers a streamlined solution by anchoring the synthesis on natural (S)-malic acid. This abundant and inexpensive feedstock inherently possesses the required chirality, allowing for the direct construction of the carbon skeleton with minimal stereochemical manipulation. The route is characterized by the use of common, scalable reagents such as thionyl chloride for esterification, tosyl chloride for activation, and lithium aluminium hydride for reduction. A key innovation lies in the strategic use of Grignard coupling catalyzed by cuprous iodide to join the two major fragments of the molecule, a reaction known for its reliability and scalability in fine chemical manufacturing. By avoiding exotic catalysts and minimizing purification steps, this novel approach drastically reduces the operational burden on production facilities. The result is a synthesis that is not only shorter in terms of step count but also demonstrates superior atom economy and environmental compatibility, addressing the core pain points of traditional pheromone manufacturing.
Mechanistic Insights into Stereoselective Alkylation and Cuprate Coupling
The success of this synthetic route hinges on the precise control of stereochemistry during the formation of the carbon-carbon bonds. The initial phase involves the conversion of (S)-malic acid into a protected diol intermediate, where the stereocenter is preserved through careful selection of reaction conditions that avoid racemization. Subsequent alkylation steps, particularly the introduction of the methyl group at the C-2 position using LDA and methyl iodide at cryogenic temperatures (-78°C), are critical for establishing the (2S, 3S) configuration. The use of strong, non-nucleophilic bases like lithium diisopropylamide ensures kinetic control over the enolate formation, favoring the desired diastereomer with high selectivity. This level of control is essential because any deviation in stereochemistry at these early stages would propagate through the synthesis, rendering the final pheromone biologically inactive. The rigorous temperature control and stoichiometry management described in the patent highlight the importance of process parameters in maintaining optical purity throughout the sequence.
The convergence of the two molecular fragments is achieved through a copper-catalyzed cross-coupling reaction between a long-chain Grignard reagent and a chiral tosylate. In this mechanistic step, cuprous iodide plays a pivotal role by forming an organocuprate species in situ, which is less basic and more nucleophilic than the corresponding Grignard reagent. This modification suppresses competing side reactions such as elimination or direct attack on the ester functionalities, ensuring that the coupling occurs selectively at the intended electrophilic center. The reaction proceeds with inversion of configuration at the coupling site, a predictable outcome that allows chemists to design the precursor stereochemistry accordingly. Following the coupling, the removal of protecting groups like the tert-butyldimethylsilyl (TBS) ether and tetrahydropyranyl (THP) ether is performed under mild acidic conditions, revealing the final triol structure without compromising the integrity of the newly formed carbon backbone. This mechanistic robustness is what makes the process suitable for reliable [agrochemical intermediate] supply.
How to Synthesize (2S,3S,7S)-3,7-dimethyl-2-pentadecanol Efficiently
The synthesis of this high-value pheromone intermediate requires a disciplined approach to reaction sequencing and purification. The process begins with the activation of the chiral pool material and proceeds through distinct phases of chain elongation and functional group interconversion. Operators must pay close attention to the exclusion of moisture during the Grignard formation and coupling steps, as water can quench the organometallic reagents and drastically reduce yields. The detailed protocol involves specific workup procedures, such as the use of saturated ammonium chloride for quenching and silica gel column chromatography for purification, which are standard in fine chemical laboratories but require optimization for larger batches. Understanding the nuances of each transformation, from the initial esterification to the final acylation, is crucial for achieving the reported high yields and optical purity. For a comprehensive breakdown of the specific operational parameters, temperatures, and stoichiometric ratios required for each step, please refer to the standardized synthesis guide below.
- Preparation of Chiral Building Blocks: Convert natural (S)-malic acid into protected diol intermediates (S)-1,3-butanediol and (2S,3S)-2-methyl-1,3-butanediol through esterification, tosylation, and selective reduction.
- Chain Elongation and Functionalization: Transform the chiral diols into alkyl halides and organometallic reagents, specifically preparing (S)-3-methyl-undecyl bromide and the corresponding Grignard reagent.
- Final Coupling and Esterification: Perform a copper-catalyzed coupling between the long-chain Grignard reagent and the protected chiral tosylate, followed by deprotection and acylation to yield the final acetate or propionate pheromones.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the shift towards this malic acid-based synthesis offers profound strategic benefits. Traditional methods often relied on specialized chiral reagents or catalysts that were subject to volatile pricing and limited availability, creating bottlenecks in the supply of critical [agrochemical intermediate] materials. By pivoting to a feedstock like (S)-malic acid, which is produced on a massive scale for the food and beverage industry, manufacturers can secure a stable and cost-effective raw material base. This transition effectively decouples pheromone production from the fluctuations of the specialty chemical market, ensuring greater supply continuity even during periods of global raw material scarcity. Furthermore, the use of commodity chemicals for reagents means that sourcing is simplified, reducing the administrative burden on purchasing departments and minimizing the risk of single-source supplier dependency.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the elimination of expensive chiral catalysts and the reduction in total processing time. Since the chirality is derived from the starting material rather than induced through costly asymmetric transformations, the direct material costs are significantly lowered. Additionally, the shorter reaction sequence implies fewer unit operations, less solvent consumption, and reduced energy usage for heating and cooling cycles. The high yields reported in most steps further contribute to cost efficiency by maximizing the output per batch, thereby lowering the cost of goods sold (COGS). This structural cost advantage allows for more competitive pricing in the [cost reduction in agrochemical manufacturing] landscape without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and robustness of the chemical transformations involved. The reliance on standard reagents like thionyl chloride, pyridine, and magnesium metal means that backup suppliers can be easily identified and qualified, mitigating the risk of disruption. Unlike processes requiring custom-synthesized ligands or sensitive organometallic catalysts that degrade upon storage, the reagents here are shelf-stable and widely traded. This flexibility enables [reducing lead time for high-purity pheromones] by allowing for rapid ramp-up of production capacity in response to seasonal demand spikes typical in the pest control sector. The predictability of the reaction outcomes also reduces the incidence of batch failures, ensuring consistent delivery schedules to downstream formulators.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of hazardous heavy metals or toxic sulfur byproducts that characterize older synthetic routes. The waste streams generated are primarily organic salts and solvents, which can be managed through standard recovery and incineration protocols, aligning with increasingly stringent environmental regulations. The process avoids the generation of difficult-to-treat sulfide wastes, simplifying the permitting process for new manufacturing lines. Moreover, the high stereoselectivity minimizes the formation of diastereomeric impurities, reducing the need for extensive recycling loops or complex chromatographic separations at scale. This clean profile supports the [commercial scale-up of complex chiral intermediates] while maintaining a sustainable operational footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from a detailed analysis of the patent specifications and are intended to clarify the operational feasibility and strategic value of the process for potential partners. Understanding these aspects is vital for integrating this technology into existing production portfolios and assessing its fit within current quality assurance frameworks. The answers reflect the specific advantages outlined in the intellectual property documentation.
Q: Why is natural (S)-malic acid chosen as the starting material for this synthesis?
A: Natural (S)-malic acid is selected because it is a cheap, readily available chiral pool resource with high inherent enantiomeric purity. Using it eliminates the need for expensive chiral catalysts or resolution steps required in older methods, significantly simplifying the process and reducing raw material costs while ensuring the correct stereochemistry for the final pheromone activity.
Q: How does this method improve upon previous synthetic routes for pine sawfly pheromones?
A: Previous methods, such as those by Mori or Akira, often involved excessive reaction steps, complex fractionation processes, or environmentally hazardous sulfur compounds. This patented route minimizes the number of steps, utilizes common reagents like thionyl chloride and lithium aluminium hydride, and achieves high stereoselectivity without the need for complex chiral auxiliaries, resulting in higher overall yields and better environmental compliance.
Q: What are the final products obtained from this synthetic pathway?
A: The primary target of this synthesis is the optically active alcohol (2S,3S,7S)-3,7-dimethyl-2-pentadecanol. This key intermediate is subsequently acylated to produce the biologically active sex pheromones: (2S,3S,7S)-3,7-dimethyl-2-pentadecanol acetate and its propionate ester, which are critical for pest control applications against Diprionidae species.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pine Sawfly Sex Pheromone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity pheromones play in modern integrated pest management strategies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of (2S,3S,7S)-3,7-dimethyl-2-pentadecanol ester meets the exacting standards required for biological efficacy. Our commitment to quality extends beyond mere compliance; we actively optimize processes to enhance stability and shelf-life, providing our clients with a superior product that performs consistently in the field.
We invite you to collaborate with us to leverage this advanced synthesis technology for your agrochemical portfolio. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can drive value and efficiency in your supply chain. Together, we can advance the availability of sustainable and effective pest control solutions globally.
