Advanced Manufacturing of High Purity Hexaphenoxycyclotriphosphazene for Global Specialty Chemical Markets
The global demand for high-performance flame retardant materials and specialized organophosphorus intermediates has driven intense research into optimizing synthetic routes for cyclic phosphazenes. Patent CN101648978A introduces a groundbreaking preparation method for high purity hexaphenoxycyclotriphosphazene, a critical compound widely utilized in modifying the fire resistance of polyethylene and viscose fibers. Unlike conventional methods that often struggle with purity levels hovering around 70% to 80%, this novel technology leverages a sophisticated phase transfer catalysis system to consistently achieve product purity exceeding 99%. The innovation lies not merely in the final substitution step but in the holistic integration of precursor synthesis and downstream purification, creating a seamless workflow that addresses the chronic pain points of yield loss and solvent contamination. For R&D directors and procurement strategists, this patent represents a pivotal shift towards more sustainable and economically viable manufacturing protocols for complex nitrogen-phosphorus organic compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenoxy-substituted cyclotriphosphazenes has been plagued by inefficient solvent systems and costly catalytic reagents that hinder industrial scalability. Prior art, such as the methods disclosed in CN1284508, frequently relies on low-boiling, volatile solvents like acetonitrile or acetone, which pose significant safety hazards and complicate solvent recovery processes due to their high vapor pressure. Furthermore, traditional approaches often necessitate the use of expensive acid binding agents such as triethylamine or 4-dimethylaminopyridine (DMAP) in stoichiometric excess, leading to cumbersome post-reaction separation procedures and inflated production costs. The reliance on multiple solvent switches—using THF for reaction and ethyl acetate for purification—introduces additional unit operations that increase energy consumption and reduce overall throughput. These legacy methods also suffer from poor selectivity, often generating substantial amounts of tetrameric phosphonitrile byproducts that are difficult to remove, ultimately capping the achievable purity and limiting the material's application in high-end sectors.
The Novel Approach
The methodology outlined in patent CN101648978A fundamentally reengineers the synthesis landscape by adopting a unified chlorobenzene solvent system that persists through both the precursor formation and the final substitution stages. This single-solvent strategy eliminates the need for intermediate solvent exchanges, drastically simplifying the operational workflow and enhancing the feasibility of large-scale continuous production. By replacing expensive amine bases with recyclable polyethylene glycol (PEG-500~1000) as a phase transfer catalyst, the process not only reduces raw material expenditures but also improves the exposure of reactive sodium phenolate ions, thereby accelerating reaction kinetics. The result is a highly selective transformation that suppresses the formation of unwanted oligomers, allowing for the direct isolation of the target hexaphenoxycyclotriphosphazene with purity levels greater than 99% through simple concentration crystallization. This approach effectively resolves the trade-off between high purity and high yield, delivering a robust protocol that is ideally suited for the rigorous demands of modern chemical supply chains.
Mechanistic Insights into PEG-Catalyzed Nucleophilic Substitution
The core chemical innovation driving this superior performance is the utilization of polyethylene glycol (PEG) to facilitate the nucleophilic substitution of chlorine atoms on the phosphazene ring. In the reaction mixture, the ether oxygen atoms within the PEG chain form stable complexes with sodium cations from the sodium phenolate, effectively stripping away the ion pairing and exposing the highly nucleophilic phenoxide anions. This "naked anion" effect significantly enhances the reactivity of the phenolate species towards the electrophilic phosphorus centers in hexachlorocyclotriphosphazene (HCTP), allowing the substitution to proceed efficiently at moderate temperatures ranging from 60°C to 140°C. The steric environment created by the PEG catalyst also contributes to the remarkable regioselectivity observed, ensuring that the substitution proceeds uniformly across all six chlorine positions without triggering ring-opening polymerization or forming linear oligomeric impurities. This mechanistic precision is critical for maintaining the structural integrity of the cyclic phosphazene core, which is essential for its subsequent performance as a flame retardant additive.
Furthermore, the control of residual chlorine content is meticulously managed through this catalytic system, ensuring that the final product contains less than 500ppm of chlorinity, a specification that is often difficult to meet with traditional base-mediated methods. The reaction pathway minimizes the generation of hydrochloric acid byproducts that could otherwise degrade the phosphazene ring or require extensive neutralization steps. By maintaining a closed-loop system where the catalyst remains active and the solvent remains consistent, the process achieves a level of atomic economy that translates directly into reduced waste generation. For technical teams evaluating process safety and impurity profiles, this mechanism offers a predictable and controllable route that mitigates the risks associated with exothermic runaway reactions common in highly exothermic nucleophilic substitutions. The ability to tune the molecular weight of the PEG catalyst (between 500 and 1000) provides an additional handle for optimizing viscosity and mass transfer characteristics during scale-up.
How to Synthesize Hexaphenoxycyclotriphosphazene Efficiently
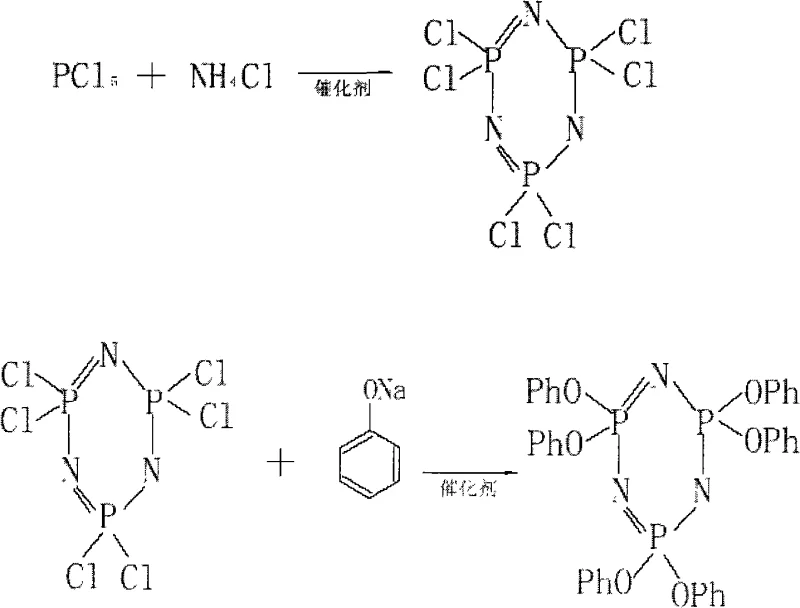
The synthesis protocol described in the patent is divided into three distinct but integrated phases: the preparation of high-purity hexachlorocyclotriphosphazene, the generation of sodium phenolate, and the final coupling reaction. The initial step involves the condensation of phosphorus pentachloride and ammonium chloride in chlorobenzene, catalyzed by metal salts such as magnesium chloride or zinc chloride, to form the critical HCTP intermediate with minimal tetramer contamination. Following the isolation or direct use of the HCTP solution, sodium phenolate is generated in situ by reacting phenol with a mixed hydroxide solution under nitrogen protection, ensuring anhydrous conditions that prevent hydrolysis of the sensitive phosphazene ring. The final coupling utilizes the PEG catalyst to drive the substitution to completion, followed by a streamlined workup involving alkaline washing, acid neutralization, and vacuum concentration to induce crystallization of the ultra-pure product.
- Preparation of Hexachlorocyclotriphosphazene (HCTP) via controlled reaction of phosphorus pentachloride and ammonium chloride in chlorobenzene with metal salt catalysts.
- In-situ generation of sodium phenolate using phenol and mixed hydroxides in chlorobenzene under nitrogen protection.
- Coupling reaction of HCTP and sodium phenolate using PEG-500~1000 as a phase transfer catalyst at 60-140°C, followed by concentration crystallization purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers transformative benefits that extend far beyond simple yield improvements. The elimination of volatile, low-boiling solvents like THF and acetonitrile in favor of chlorobenzene creates a much safer and more manageable solvent recovery infrastructure, significantly lowering the capital expenditure required for distillation and waste treatment facilities. The shift from stoichiometric amounts of expensive amine bases to catalytic quantities of inexpensive, recyclable PEG results in a drastic reduction in variable raw material costs per kilogram of finished product. Moreover, the simplified purification process, which relies on concentration crystallization rather than multiple recrystallization steps, reduces processing time and energy consumption, thereby increasing the overall throughput of the manufacturing plant. These efficiencies collectively contribute to a more resilient supply chain capable of meeting large-volume orders with consistent quality and shorter lead times.
- Cost Reduction in Manufacturing: The replacement of high-cost catalysts like DMAP and triethylamine with recoverable PEG eliminates a major cost driver in the bill of materials, while the single-solvent system reduces the complexity and cost of solvent procurement and recycling. By avoiding the need for multiple solvent types, the facility can optimize its storage and handling infrastructure, leading to substantial long-term operational savings. The high selectivity of the reaction minimizes the loss of valuable starting materials to byproduct formation, ensuring that a greater proportion of input costs are converted into saleable high-purity product. Additionally, the reduced need for extensive purification steps lowers utility costs associated with heating, cooling, and filtration equipment.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals such as chlorobenzene, phenol, and PEG ensures that the supply chain is not vulnerable to the shortages or price volatility often associated with specialized reagents. The robustness of the process against minor variations in reaction conditions means that production schedules are less likely to be disrupted by batch failures or off-spec material. The ability to recycle the catalyst and solvent internally creates a closed-loop system that reduces dependence on external waste disposal services and mitigates regulatory risks related to hazardous waste generation. This self-sufficiency enhances the stability of supply, making it easier to guarantee delivery commitments to downstream customers in the flame retardant and polymer industries.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, with reaction conditions that are easily manageable in large reactors without requiring exotic high-pressure or cryogenic equipment. The reduction in solvent diversity simplifies compliance with environmental regulations regarding VOC emissions and wastewater treatment, as the effluent stream is more uniform and easier to treat. The high purity of the final product reduces the burden on customers to perform additional purification, adding value to the supply chain and strengthening customer relationships. Furthermore, the lower energy intensity of the process aligns with global sustainability goals, positioning the manufacturer as a responsible partner in the green chemistry initiative.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this technology. Understanding these details is crucial for technical teams assessing the feasibility of integrating this process into existing production lines or for procurement specialists evaluating the total cost of ownership. The answers provided reflect the specific advantages of the PEG-catalyzed chlorobenzene system over legacy methods.
Q: What is the primary advantage of using PEG catalyst over traditional amine bases?
A: The use of PEG-500~1000 eliminates the need for expensive and difficult-to-recycle acid binding agents like triethylamine or DMAP, significantly reducing raw material costs and simplifying downstream purification.
Q: How does this process achieve purity levels exceeding 99%?
A: By utilizing a single solvent system (chlorobenzene) throughout both synthesis stages and employing a highly selective catalytic mechanism that minimizes tetramer byproduct formation, the process avoids the impurities common in multi-solvent methods.
Q: Is the chlorobenzene solvent recoverable in this manufacturing route?
A: Yes, the process is designed for industrial scalability where chlorobenzene serves as the sole reaction medium, allowing for efficient distillation and recycling, thereby minimizing waste and environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hexaphenoxycyclotriphosphazene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN101648978A are fully realized in every batch we deliver. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every shipment of hexaphenoxycyclotriphosphazene meets the exacting standards required for high-performance flame retardant applications. Our commitment to quality assurance means that you can rely on us not just as a vendor, but as a strategic extension of your own R&D and supply chain operations.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to our high-purity intermediates. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate the tangible value of partnering with a leader in fine chemical manufacturing. Let us help you secure a competitive advantage through superior material quality and supply chain efficiency.
