Advancing Tenofovir PMPA Production: A Green Solid Acid Catalytic Route for Scalable API Manufacturing
Introduction to Green Tenofovir Manufacturing
The global demand for potent antiviral agents continues to drive innovation in the synthesis of key pharmaceutical intermediates. Patent CN113214322B, published in late 2022, introduces a significant technological breakthrough in the preparation of Tenofovir, also known as (R)-9-[2-(phosphonomethoxy)propyl]adenine or PMPA. This molecule serves as the critical core structure for widely prescribed antiretroviral drugs such as Tenofovir Disoproxil Fumarate (TDF) and Tenofovir Alafenamide (TAF). 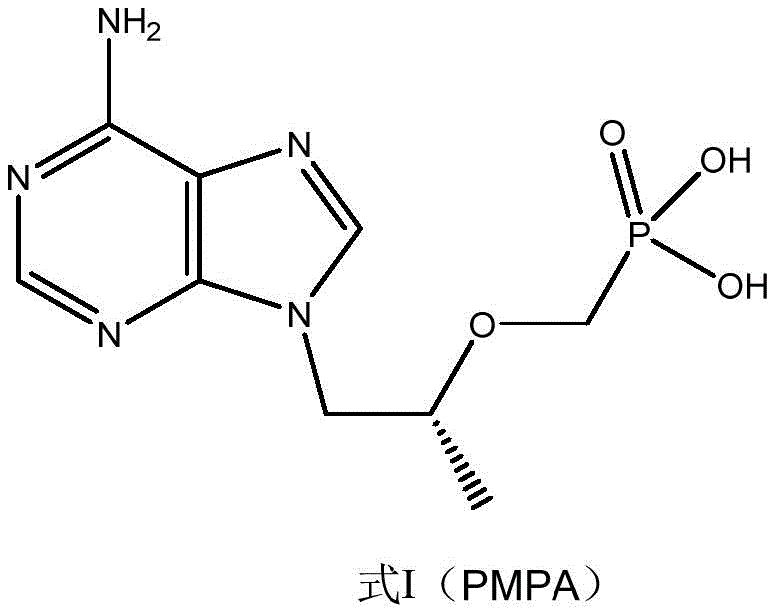 The disclosed method replaces traditional, harsh hydrolysis conditions with a novel solid super acid catalytic system. By utilizing a rare-earth neodymium-doped zirconia solid acid (SO4 2-/ZrO2-Nd2O3), the process achieves high conversion rates while drastically mitigating the environmental hazards associated with conventional strong acid hydrolysis. This development represents a pivotal shift towards sustainable manufacturing practices for high-volume pharmaceutical intermediates, addressing both regulatory pressure for greener chemistry and the economic imperative for cost-efficient production.
The disclosed method replaces traditional, harsh hydrolysis conditions with a novel solid super acid catalytic system. By utilizing a rare-earth neodymium-doped zirconia solid acid (SO4 2-/ZrO2-Nd2O3), the process achieves high conversion rates while drastically mitigating the environmental hazards associated with conventional strong acid hydrolysis. This development represents a pivotal shift towards sustainable manufacturing practices for high-volume pharmaceutical intermediates, addressing both regulatory pressure for greener chemistry and the economic imperative for cost-efficient production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tenofovir has relied heavily on the hydrolysis of phosphonate ester precursors, such as the diethyl or di-tert-butyl esters, using liquid strong acids. As illustrated in prior art routes like those referenced in CN111205326, the standard procedure involves treating the ester intermediate with concentrated hydrochloric acid, hydrobromic acid, or sulfuric acid at elevated temperatures. 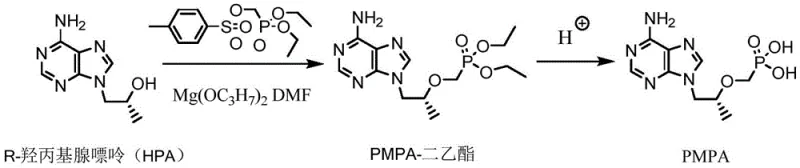 These conventional liquid acid methods present severe engineering and environmental challenges. The use of concentrated mineral acids generates substantial quantities of high-salinity wastewater that requires complex neutralization and desalination treatments before discharge, imposing a heavy burden on effluent processing facilities. Furthermore, the reaction conditions often necessitate high temperatures that promote the evolution of corrosive acidic gases, such as hydrogen chloride or hydrogen bromide. These gases not only pose significant occupational health risks but also accelerate the corrosion of stainless steel reactors and piping, leading to frequent equipment failure, increased maintenance costs, and potential production interruptions. The handling and disposal of these hazardous byproducts create a bottleneck for commercial scale-up of complex nucleotide analogs, limiting the ability of manufacturers to expand capacity efficiently.
These conventional liquid acid methods present severe engineering and environmental challenges. The use of concentrated mineral acids generates substantial quantities of high-salinity wastewater that requires complex neutralization and desalination treatments before discharge, imposing a heavy burden on effluent processing facilities. Furthermore, the reaction conditions often necessitate high temperatures that promote the evolution of corrosive acidic gases, such as hydrogen chloride or hydrogen bromide. These gases not only pose significant occupational health risks but also accelerate the corrosion of stainless steel reactors and piping, leading to frequent equipment failure, increased maintenance costs, and potential production interruptions. The handling and disposal of these hazardous byproducts create a bottleneck for commercial scale-up of complex nucleotide analogs, limiting the ability of manufacturers to expand capacity efficiently.
The Novel Approach
The methodology described in patent CN113214322B offers a transformative solution by substituting corrosive liquid acids with a heterogeneous solid super acid catalyst. In this optimized route, the phosphonate ester intermediate—whether derived from diethyl or di-tert-butyl precursors—is subjected to hydrolysis in the presence of SO4 2-/ZrO2-Nd2O3 and deionized water. This solid acid catalyst functions effectively under controlled thermal conditions, facilitating the cleavage of the phosphonate ester bonds to yield the free phosphonic acid group of Tenofovir. Unlike the chaotic mixture of salts produced by liquid acid neutralization, the solid catalyst remains physically distinct from the reaction medium. This heterogeneity allows for the catalyst to be separated simply by hot filtration, washed, and potentially regenerated for reuse in subsequent batches. The elimination of stoichiometric amounts of mineral acid fundamentally alters the waste profile of the reaction, converting a high-salt waste stream into a manageable aqueous stream with minimal inorganic contamination. This approach not only aligns with green chemistry principles but also simplifies the downstream purification process, enabling the isolation of Tenofovir with superior purity profiles suitable for demanding API intermediate specifications.
Mechanistic Insights into Solid Super Acid Catalyzed Hydrolysis
The efficacy of this novel process hinges on the unique physicochemical properties of the neodymium-doped zirconia solid super acid. The catalyst, formulated as SO4 2-/ZrO2-Nd2O3, is engineered to possess a significantly larger specific surface area compared to undoped zirconia variants. The incorporation of the rare earth element Neodymium (Nd) plays a critical role in stabilizing the sulfate groups on the oxide surface and modifying the electronic environment of the active sites. This doping effect enhances the electron-withdrawing capability of the surface elements, thereby increasing the strength and density of the Brønsted and Lewis acid sites essential for catalyzing the hydrolysis reaction. The robust acid strength allows the catalyst to activate water molecules effectively, promoting nucleophilic attack on the phosphorus center of the ester intermediate even under relatively mild thermal conditions compared to traditional refluxing in strong mineral acids. This mechanistic advantage ensures that the hydrolysis proceeds to completion with high selectivity, minimizing the formation of degradation byproducts that often plague acid-sensitive nucleobase structures.
From an impurity control perspective, the solid acid mechanism offers distinct advantages over liquid acid counterparts. In traditional hydrolysis, the aggressive nature of boiling hydrochloric or sulfuric acid can lead to depurination or other acid-catalyzed decomposition of the adenine moiety, generating difficult-to-remove organic impurities. The tunable acidity of the SO4 2-/ZrO2-Nd2O3 system provides a more controlled reaction environment that preserves the integrity of the sensitive purine ring while efficiently cleaving the phosphonate ester. Furthermore, the physical separation of the catalyst prevents the introduction of metal ions or halide contaminants that could complicate the final crystallization step. The patent data indicates that this controlled catalytic environment consistently yields Tenofovir with an HPLC purity exceeding 99.5%, demonstrating the system's capability to produce high-purity API intermediates that meet rigorous pharmacopeial standards without the need for extensive recrystallization cycles.
How to Synthesize Tenofovir (PMPA) Efficiently
The implementation of this green synthesis route involves a streamlined sequence of unit operations designed for industrial robustness. The process begins with the coupling of R-hydroxypropyl adenine with a phosphonate esterifying agent to form the protected intermediate, followed by the critical solid-acid catalyzed hydrolysis step. The detailed operational parameters, including specific temperature ramps, catalyst loading ratios, and filtration protocols, are essential for maximizing yield and ensuring catalyst recovery. For process engineers and R&D teams looking to adopt this technology, the following guide outlines the standardized synthetic workflow derived from the patent examples.
- React R-hydroxypropyl adenine with dialkyl p-toluenesulfonylmethoxy phosphonate using magnesium isopropoxide as a coupling catalyst to form the phosphonate ester intermediate.
- Hydrolyze the phosphonate ester intermediate using SO4 2-/ZrO2-Nd2O3 solid super acid catalyst in deionized water under heated conditions (100-200°C for diethyl ester).
- Filter to recover the reusable solid acid catalyst, decolorize the filtrate with activated carbon, and adjust pH to precipitate high-purity Tenofovir.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solid acid catalytic route translates into tangible strategic benefits beyond mere technical novelty. The primary value driver lies in the drastic simplification of the waste management infrastructure required for production. By eliminating the generation of high-salinity wastewater and corrosive acidic off-gases, manufacturers can significantly reduce the operational expenditure associated with effluent treatment plants and scrubber systems. This reduction in environmental compliance overhead directly contributes to cost reduction in pharmaceutical intermediates manufacturing, allowing for more competitive pricing structures in a market where margin compression is a constant challenge. Additionally, the mitigation of equipment corrosion extends the lifecycle of capital assets such as reactors, condensers, and piping networks, deferring major capital replacement expenditures and ensuring higher asset utilization rates over time.
- Cost Reduction in Manufacturing: The transition from consumable liquid acids to a reusable solid catalyst fundamentally changes the cost structure of the hydrolysis step. Traditional methods consume vast quantities of mineral acids which are then neutralized into waste salts, representing a double cost of raw material purchase and waste disposal. In contrast, the solid super acid can be recovered via filtration and reused, dramatically lowering the recurring cost of reagents. Furthermore, the simplified workup procedure reduces the consumption of auxiliary materials such as bases for neutralization and solvents for extraction, leading to substantial overall savings in variable production costs without compromising output quality.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by environmental regulatory crackdowns on polluting processes. Facilities utilizing traditional strong acid hydrolysis are at higher risk of production curtailments due to wastewater discharge limits or air emission violations. The green nature of this solid acid process significantly lowers the environmental footprint, making the supply chain more resilient against regulatory shocks. Moreover, the reduced corrosion rate minimizes unplanned maintenance shutdowns caused by equipment failure, ensuring a more consistent and reliable delivery schedule for downstream API manufacturers who depend on steady flows of pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling up exothermic reactions involving corrosive gases presents significant engineering hurdles, often requiring specialized Hastelloy reactors or glass-lined vessels that are expensive and have long lead times. The mild and non-corrosive nature of the solid acid hydrolysis allows for the use of standard stainless steel equipment, facilitating easier and faster commercial scale-up. The process inherently aligns with increasingly stringent global environmental, social, and governance (ESG) criteria, positioning suppliers who adopt this technology as preferred partners for multinational pharmaceutical companies seeking to decarbonize and green their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solid acid catalytic technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs.
Q: How does the solid super acid catalyst improve equipment longevity compared to traditional hydrolysis?
A: Traditional methods utilize corrosive strong acids like hydrochloric or hydrobromic acid, which release corrosive gases and damage reactor linings. The patented solid acid method operates under milder conditions without generating corrosive acidic gases, significantly extending equipment lifespan and reducing maintenance downtime.
Q: What is the expected purity profile of Tenofovir produced via this green route?
A: According to the patent data, this method achieves an HPLC purity of greater than 99.5%, meeting stringent specifications required for downstream antiviral API synthesis such as TDF and TAF.
Q: Is the neodymium-doped zirconia catalyst reusable in this process?
A: Yes, the solid nature of the SO4 2-/ZrO2-Nd2O3 catalyst allows for easy separation via hot filtration. The patent demonstrates that the recovered catalyst can be reused in subsequent batches, enhancing process economics and reducing solid waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tenofovir Supplier
The technological advancements detailed in patent CN113214322B underscore the evolving landscape of antiviral intermediate synthesis, where efficiency and sustainability are paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring such innovative processes to life. Our state-of-the-art facilities are equipped with rigorous QC labs capable of validating the stringent purity specifications required for Tenofovir and its derivatives, ensuring that every batch meets the exacting standards of the global pharmaceutical industry. We are committed to translating laboratory-scale breakthroughs into robust, GMP-compliant manufacturing realities.
We invite procurement leaders and technical decision-makers to engage with us to explore how this green synthesis route can be integrated into your supply strategy. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating the specific economic benefits of switching to this catalytic method. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring a seamless transition to a more sustainable and cost-effective source of high-quality Tenofovir intermediates.
