Advanced Synthesis of Mono-Substituted Zinc Phthalocyanine for High-Purity Pharmaceutical Applications
Introduction to Next-Generation Photosensitizer Intermediates
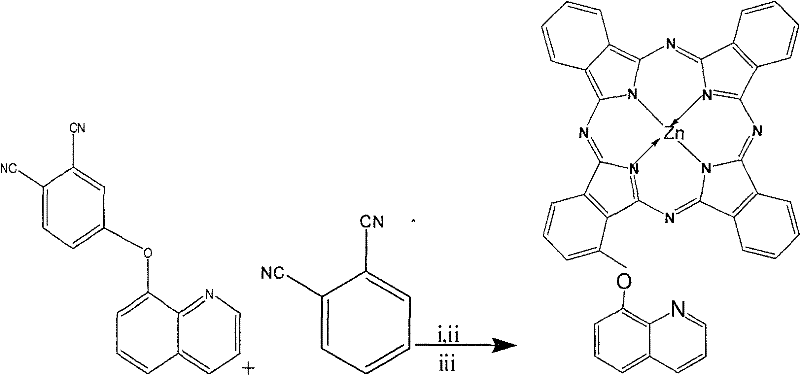
The landscape of photodynamic therapy (PDT) agents is undergoing a significant transformation, driven by the demand for highly defined molecular structures that offer predictable biological activity. Patent CN101260110B introduces a breakthrough methodology for the preparation of α-(8-quinolineoxy) mono-substituted zinc phthalocyanine, a sophisticated metal complex with the molecular formula C41H21N9OZn. Unlike traditional phthalocyanine syntheses that often yield intractable mixtures of isomers, this patented process focuses on generating a single, structurally defined species that exhibits superior photosensitive activity against cancer cells. The strategic incorporation of the 8-quinolineoxy group at the alpha position enhances the molecule's solubility and targeting capabilities, making it an ideal candidate for next-generation anticancer drugs. For R&D directors and procurement specialists in the fine chemical sector, understanding this synthetic route is critical, as it represents a shift from statistical substitution methods to precise, controlled functionalization. This report analyzes the technical merits of this approach, highlighting its potential for cost reduction in pharmaceutical intermediates manufacturing and its alignment with rigorous quality standards required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of substituted phthalocyanines has been plagued by the inherent statistical nature of the cyclotetramerization reaction. When attempting to introduce functional groups onto the phthalocyanine ring using conventional methods, chemists typically encounter a complex distribution of products ranging from unsubstituted to tetra-substituted species, along with a myriad of positional isomers. For instance, polysubstituted zinc phthalocyanines often exist as mixtures where substituents occupy different alpha and beta positions, leading to significant structural heterogeneity. This isomeric complexity creates a nightmare for downstream processing, as the physical and chemical properties of these isomers are often strikingly similar, rendering standard purification techniques like crystallization or simple chromatography ineffective. Consequently, the final product often suffers from batch-to-batch variability, which is unacceptable for pharmaceutical applications where purity and reproducibility are paramount. Furthermore, the presence of multiple isomers can lead to unpredictable pharmacokinetic profiles, complicating toxicology studies and regulatory filings for new drug candidates.
The Novel Approach
The methodology outlined in CN101260110B circumvents these challenges by employing a targeted pre-functionalization strategy combined with controlled stoichiometry. By first synthesizing a specific precursor, 3-(8-quinolineoxy) phthalonitrile, and then co-reacting it with unsubstituted phthalonitrile, the process biases the statistical distribution towards the desired mono-substituted product. Crucially, because the target is a mono-substituted derivative, the issue of positional isomerism is effectively eliminated; a single substituent on the macrocycle does not generate the complex geometric isomers seen in di- or tri-substituted analogs. This structural simplicity translates directly into operational efficiency, as the separation burden is significantly reduced to distinguishing between degrees of substitution (n=0, 1, 2, 3, 4) rather than resolving intricate positional isomers. This approach not only streamlines the purification workflow but also ensures that the final active pharmaceutical ingredient (API) intermediate possesses a defined structure, thereby enhancing the reliability of the supply chain for high-value medical applications.
Mechanistic Insights into DBU-Catalyzed Cyclotetramerization
The core of this synthetic innovation lies in the use of 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) as a catalyst for the cyclotetramerization step. DBU is a potent, non-nucleophilic amidine base that plays a dual role in facilitating the formation of the phthalocyanine macrocycle. Mechanistically, the reaction involves the nucleophilic attack of the nitrile nitrogen on the electrophilic carbon of an adjacent nitrile group, initiated by the coordination of the zinc ion. DBU accelerates this process by deprotonating intermediate species and stabilizing the transition states required for ring closure, allowing the reaction to proceed under relatively mild reflux conditions in pentyl alcohol. This is particularly advantageous for preserving the integrity of the 8-quinolineoxy moiety, which might be susceptible to degradation under the harsher conditions (such as high-temperature fusion with metal salts) often required in traditional phthalocyanine syntheses. The use of a homogeneous organic base also ensures better mixing and heat transfer compared to heterogeneous inorganic bases, leading to more consistent reaction kinetics and higher reproducibility on a commercial scale.
Following the cyclization, the reaction mixture contains a distribution of zinc phthalocyanine species with varying numbers of quinoline substituents (α-QnZnPc, where n=0-4). The separation strategy leverages the differences in hydrophobicity imparted by the quinoline groups. The patent specifies the use of C18 reversed-phase high-performance liquid chromatography (HPLC) for isolation. In this system, the stationary phase is non-polar, while the mobile phase consists of a polar mixture of water, DMF, and acetonitrile. The unsubstituted zinc phthalocyanine (n=0) is the most hydrophobic and retains longest, while the highly substituted species are more polar. The target mono-substituted product (n=1) elutes as a distinct band, allowing for high-purity collection. This chromatographic resolution is far more efficient than attempting to separate positional isomers, reinforcing the strategic advantage of the mono-substitution design. The ability to isolate a single isomer-free compound is a critical quality attribute that supports the development of robust analytical methods for quality control.
How to Synthesize Alpha-(8-quinolineoxy) Zinc Phthalocyanine Efficiently
The practical execution of this synthesis involves a two-stage process that balances yield with purity. The first stage focuses on the nucleophilic aromatic substitution of 3-nitrophthalonitrile with 8-hydroxyquinoline in the presence of potassium carbonate in DMF, yielding the key intermediate 3-(8-quinolineoxy) phthalonitrile. This step requires careful temperature control, typically between 5°C and 40°C, to prevent side reactions while ensuring complete conversion. The second stage involves the statistical cyclotetramerization where the intermediate is mixed with excess unsubstituted phthalonitrile and zinc acetate in pentyl alcohol. The detailed operational parameters, including specific molar ratios and reflux times, are critical for maximizing the proportion of the mono-substituted species in the crude mixture before purification. For process chemists looking to implement this route, adherence to the standardized protocols below is essential to achieve the reported yields and purity levels.
- Synthesize 3-(8-quinoline oxy) phthalonitrile by reacting 3-nitrophthalonitrile with 8-hydroxyquinoline in DMF using potassium carbonate at 5-40°C.
- Perform cyclotetramerization by reacting the intermediate with phthalonitrile and zinc acetate in pentyl alcohol using DBU catalyst under reflux.
- Purify the crude mixture using C18 reversed-phase high-performance liquid chromatography to isolate the mono-substituted target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthetic route offers substantial benefits for procurement managers and supply chain heads focused on cost reduction in specialty chemical manufacturing. The primary economic driver is the simplification of the purification process. By eliminating the need to separate complex positional isomers, manufacturers can reduce the number of chromatographic cycles or recrystallization steps required, leading to lower solvent consumption and reduced waste disposal costs. Furthermore, the use of readily available starting materials such as 3-nitrophthalonitrile and 8-hydroxyquinoline ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or custom-synthesized precursors. The robustness of the DBU-catalyzed reaction also implies a wider operating window, which reduces the likelihood of batch failures and enhances overall production throughput. These factors collectively contribute to a more sustainable and cost-effective manufacturing model, making the final photosensitizer intermediate more accessible for large-scale pharmaceutical production.
- Cost Reduction in Manufacturing: The elimination of complex isomer separation significantly lowers downstream processing costs. Traditional methods often require extensive preparative HPLC or multiple recrystallizations to remove unwanted isomers, which consumes vast amounts of high-grade solvents and stationary phases. By targeting a mono-substituted structure that inherently lacks positional isomers, the process reduces the burden on purification infrastructure. Additionally, the use of DBU allows for milder reaction conditions compared to high-temperature fusion methods, resulting in lower energy consumption and reduced thermal stress on equipment. This operational efficiency translates directly into a lower cost of goods sold (COGS), providing a competitive edge in the pricing of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like 3-nitrophthalonitrile and zinc acetate ensures that raw material availability is not a bottleneck. Unlike processes dependent on specialized catalysts or rare earth metals, this route utilizes standard organic bases and zinc salts that are globally sourced with high consistency. This reduces the risk of supply disruptions caused by geopolitical issues or single-source dependencies. Moreover, the synthetic steps are scalable using standard glass-lined or stainless steel reactors commonly found in fine chemical facilities, facilitating a seamless transition from pilot scale to commercial production. This scalability ensures that suppliers can respond rapidly to fluctuations in demand from pharmaceutical partners without compromising on lead times.
- Scalability and Environmental Compliance: The process generates fewer by-products and waste streams compared to statistical substitution methods that produce large quantities of unusable isomers. The ability to recycle solvents like DMF and pentyl alcohol further enhances the environmental profile of the manufacturing process. From a regulatory standpoint, producing a single, well-defined chemical entity simplifies the documentation required for environmental health and safety (EHS) compliance. The reduced complexity of the waste stream makes treatment and disposal more straightforward, aligning with increasingly stringent global environmental regulations. This sustainability aspect is increasingly important for multinational corporations aiming to reduce their carbon footprint and meet corporate social responsibility (CSR) goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of α-(8-quinolineoxy) mono-substituted zinc phthalocyanine. These insights are derived directly from the technical specifications and experimental data provided in patent CN101260110B, offering clarity on the feasibility and advantages of this synthetic route. Understanding these details is essential for stakeholders evaluating the integration of this intermediate into their drug development pipelines or supply networks.
Q: Why is mono-substituted zinc phthalocyanine preferred over polysubstituted variants for pharmaceutical use?
A: Mono-substituted variants possess a single, defined molecular structure without positional isomers, ensuring consistent pharmacokinetic properties and simplifying regulatory approval compared to complex isomeric mixtures.
Q: What role does DBU play in the cyclization process described in CN101260110B?
A: DBU acts as a strong, non-nucleophilic organic base that facilitates the cyclotetramerization of phthalonitriles at moderate reflux temperatures, minimizing side reactions and degradation of the sensitive quinoline moiety.
Q: How is the final product separated from unsubstituted zinc phthalocyanine?
A: The separation is achieved using C18 reversed-phase high-performance liquid chromatography (HPLC), which effectively resolves components based on their polarity differences arising from the varying number of quinoline substituents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-(8-quinolineoxy) Zinc Phthalocyanine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of structural purity and supply consistency in the development of advanced photodynamic therapy agents. Our technical team has extensively analyzed the route described in CN101260110B and is fully equipped to execute this synthesis with precision. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. Our state-of-the-art facilities are designed to handle complex metal-organic syntheses, featuring dedicated lines for chromatographic purification that meet stringent purity specifications. With our rigorous QC labs, we guarantee that every batch of alpha-(8-quinolineoxy) zinc phthalocyanine meets the exacting standards required for pharmaceutical applications, providing you with a partner who understands the nuances of high-tech chemical manufacturing.
We invite you to collaborate with us to optimize this pathway for your specific needs. Our engineering team can provide a Customized Cost-Saving Analysis to identify further efficiencies in solvent recovery and raw material sourcing tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data from our pilot batches and discuss route feasibility assessments for your upcoming projects. By partnering with NINGBO INNO PHARMCHEM, you secure a supply chain that is not only reliable and compliant but also driven by a commitment to technical excellence and continuous improvement.
