Scalable Visible Light Synthesis of Alpha-Selenyl Ketones for Commercial Pharmaceutical Manufacturing
Scalable Visible Light Synthesis of Alpha-Selenyl Ketones for Commercial Pharmaceutical Manufacturing
The landscape of organic synthesis is undergoing a paradigm shift towards sustainable, energy-efficient methodologies, and patent CN111333558A represents a significant breakthrough in the field of organoselenium chemistry. This intellectual property discloses a novel visible light-promoted method for the synthesis of α-selenyl ketone compounds, a class of molecules critical for the development of bioactive pharmaceutical ingredients and functional materials. Unlike conventional approaches that rely on harsh conditions or expensive catalysts, this invention leverages the power of ambient visible light to drive the bifunctionalization of alkenes with diselenides. The process is characterized by its operational simplicity, proceeding in an open system at room temperature without the need for exogenous photocatalysts. For R&D directors and process chemists, this technology offers a compelling route to access complex selenium-containing scaffolds with high atom economy and minimal environmental footprint. The ability to utilize simple alkenes and readily available diselenides under such mild conditions suggests a transformative potential for the manufacturing of high-value fine chemicals.
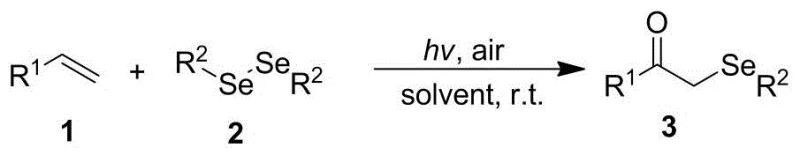
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of α-selenyl ketone motifs has been fraught with significant synthetic challenges that hinder efficient commercial production. Traditional strategies often necessitate the use of highly reactive and hazardous reagents, such as phenylselenenyl bromide, which require stringent handling protocols due to their toxicity and instability. Furthermore, classical nucleophilic substitution pathways typically involve the generation of selenolate anions, demanding the use of strong bases and strictly anhydrous, oxygen-free environments to prevent premature oxidation. Perhaps most critically, many established protocols require cryogenic conditions, often operating at temperatures as low as -78°C, to control regioselectivity and suppress side reactions. These extreme thermal requirements impose a heavy burden on energy consumption and necessitate specialized refrigeration infrastructure, drastically inflating the operational expenditure for large-scale manufacturing. Additionally, the reliance on stoichiometric amounts of expensive selenium reagents and the generation of substantial salt waste during workup further complicate the economic and environmental viability of these legacy processes.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology described in CN111333558A introduces a streamlined, catalyst-free photochemical strategy that fundamentally redefines the synthesis of α-selenyl ketones. By utilizing visible light irradiation from a standard compact fluorescent lamp, the process achieves homolytic cleavage of the selenium-selenium bond in diselenides, generating reactive selenyl radicals under ambient thermal conditions. This radical manifold allows for the direct addition across the carbon-carbon double bond of various alkenes, followed by oxidative trapping with molecular oxygen from the air. The elimination of transition metal photocatalysts is a particularly advantageous feature, as it removes the risk of metal contamination in the final product—a critical parameter for pharmaceutical applications—and avoids the high cost associated with precious metal complexes. The reaction tolerates a wide range of functional groups and proceeds efficiently in common organic solvents like acetonitrile, offering a robust and versatile platform for diversifying chemical libraries without the need for specialized equipment or extreme safety measures.
Mechanistic Insights into Visible Light-Promoted Radical Bifunctionalization
The mechanistic underpinning of this transformation relies on the unique photophysical properties of diselenides, which possess a relatively weak Se-Se bond that can be cleaved upon absorption of visible light photons. Upon irradiation, the diselenide undergoes homolysis to generate selenyl radicals, which act as the primary chain carriers in this radical cascade. These electrophilic radicals add regioselectively to the terminal position of the alkene substrate, generating a stabilized benzylic or alkyl radical intermediate. In the presence of air, this carbon-centered radical is intercepted by molecular oxygen, leading to the formation of a peroxyl radical species that eventually collapses to yield the carbonyl functionality observed in the final α-selenyl ketone product. This elegant sequence effectively merges selenylation and oxidation into a single operational step, bypassing the need for separate oxidation reagents or multi-step sequences. The absence of an external photocatalyst suggests that the diselenide itself acts as the photo-initiator, simplifying the reaction mixture and reducing the potential for catalyst-induced side reactions or decomposition pathways that often plague photoredox chemistry.
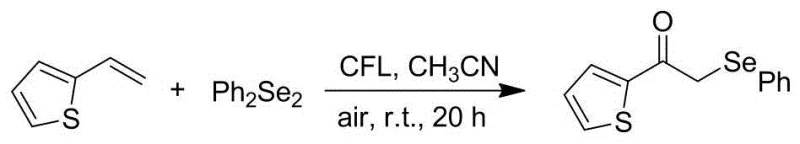
From an impurity control perspective, this radical mechanism offers distinct advantages over ionic pathways. Since the reaction does not involve strong bases or nucleophiles, issues related to base-sensitive functional groups or elimination side products are virtually eliminated. The use of air as the oxidant ensures that the only byproduct is water or reduced oxygen species, avoiding the accumulation of heavy metal waste or toxic stoichiometric oxidants like chromium or manganese salts. However, careful control of light intensity and oxygen flow is essential to prevent over-oxidation of the selenium moiety to selenoxides or selenones, which can occur if the reaction is pushed too far. The patent data indicates that yields ranging from 68% to 90% are achievable across a broad substrate scope, including electron-rich and electron-deficient styrenes as well as heteroaromatic vinyl derivatives. This consistency suggests a highly reliable kinetic profile where the rate of radical generation is well-matched to the rate of substrate consumption, minimizing the formation of oligomeric byproducts or homocoupled diselenide residues that could complicate downstream purification.
How to Synthesize Alpha-Selenyl Ketones Efficiently
The practical execution of this synthesis is designed for maximum accessibility, allowing chemists to produce high-purity intermediates using standard laboratory glassware and lighting equipment. The protocol involves simply mixing the alkene and diselenide in a solvent such as acetonitrile within a round-bottom flask, which is then placed under a 23-watt compact fluorescent lamp. The detailed standardized synthesis steps, including precise molar ratios, stirring rates, and purification parameters, are outlined in the guide below to ensure reproducibility and safety during scale-up operations.
- Charge a reactor with the alkene substrate and diaryl or dialkyl diselenide in a suitable solvent such as acetonitrile.
- Irradiate the open reaction mixture with a 23-watt compact fluorescent lamp at room temperature for approximately 20 hours.
- Remove the solvent under reduced pressure and purify the crude residue via silica gel column chromatography to isolate the target alpha-selenyl ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this visible light technology presents a compelling value proposition centered on cost efficiency and operational resilience. The primary driver of cost reduction lies in the drastic simplification of the reaction infrastructure; by eliminating the need for cryogenic cooling systems and inert gas manifolds, facilities can significantly lower their capital expenditure and utility costs. Furthermore, the exclusion of expensive transition metal photocatalysts removes a major variable from the raw material budget, while also simplifying the regulatory compliance landscape regarding heavy metal residuals in active pharmaceutical ingredients. The use of commodity chemicals like styrenes and diphenyl diselenide ensures a stable and competitive supply base, mitigating the risks associated with sourcing specialized or proprietary reagents. This robustness translates directly into enhanced supply chain reliability, as the process is less susceptible to disruptions caused by the scarcity of exotic catalysts or the logistical challenges of transporting hazardous cryogenic reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the elimination of energy-intensive cooling steps and costly catalytic systems. Traditional methods often require maintaining temperatures at -78°C for extended periods, consuming vast amounts of energy and requiring specialized chillers, whereas this method operates at room temperature, drastically cutting utility expenses. Additionally, the catalyst-free nature of the reaction means there is no need for expensive scavenging resins or complex filtration steps to remove trace metals, which streamlines the downstream processing workflow. The high atom economy of the bifunctionalization strategy ensures that a larger proportion of the starting mass is incorporated into the final product, reducing waste disposal costs and improving overall material efficiency. These factors combine to create a manufacturing process that is not only cheaper per kilogram but also more predictable in terms of total cost of ownership.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of widely available, stable starting materials that do not require special storage conditions or hazardous shipping classifications. Diselenides and styrenes are commercially abundant fine chemicals with established global supply networks, reducing the risk of single-source dependency. The operational simplicity of the reaction, which tolerates open-air conditions, means that production can be maintained even if there are temporary interruptions in inert gas supplies or if specialized glovebox equipment is undergoing maintenance. This flexibility allows for more agile production scheduling and reduces the lead time associated with setting up complex reaction vessels. Consequently, manufacturers can respond more rapidly to market demand fluctuations without being bottlenecked by intricate process requirements or fragile reagent stability issues.
- Scalability and Environmental Compliance: Scaling this technology from bench to plant is facilitated by the benign nature of the reaction conditions, which do not pose the same thermal runaway risks associated with exothermic cryogenic reactions. The use of visible light can be adapted to flow chemistry setups, where thin tubing ensures uniform irradiation, offering a clear path to continuous manufacturing and increased throughput. From an environmental standpoint, the process aligns with green chemistry principles by using air as a renewable oxidant and generating minimal hazardous waste, thereby simplifying effluent treatment and reducing the environmental compliance burden. The absence of heavy metals and toxic oxidants makes the waste stream easier to manage and dispose of, supporting corporate sustainability goals and reducing the liability associated with hazardous chemical handling. This combination of scalability and environmental stewardship makes the technology highly attractive for long-term commercial deployment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process evaluation. Understanding these details is crucial for assessing the feasibility of integrating this technology into existing production lines or R&D pipelines.
Q: Does this visible light method require expensive transition metal photocatalysts?
A: No, the patented process operates without external photocatalysts. The visible light directly activates the diselenide bond, significantly reducing raw material costs and eliminating the need for complex metal removal steps typically required in transition metal catalysis.
Q: What are the typical reaction conditions for this synthesis?
A: The reaction proceeds under remarkably mild conditions: room temperature, open to air, and using standard compact fluorescent lighting. This eliminates the need for cryogenic cooling (-78°C) or inert atmosphere techniques common in traditional organoselenium chemistry.
Q: Is this method suitable for large-scale production of pharmaceutical intermediates?
A: Yes, the simplicity of the setup (open vessel, ambient temperature) and the use of commodity chemicals like styrenes and diselenides make it highly amenable to commercial scale-up, offering a robust alternative to hazardous traditional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Selenyl Ketone Supplier
As the demand for specialized organoselenium intermediates grows in the pharmaceutical and agrochemical sectors, partnering with an experienced CDMO is essential for navigating the complexities of commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped to handle photochemical processes safely and effectively, adhering to stringent purity specifications and rigorous QC labs to guarantee that every batch meets the highest international standards. We understand the critical nature of supply chain consistency and are committed to delivering high-quality intermediates that support your drug development timelines without compromise.
We invite you to collaborate with us to leverage this innovative visible light technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your synthesis of complex alpha-selenyl ketones while maximizing value and minimizing risk.
