Industrial Scale-Up of Chiral Warfarin Derivatives via Efficient Asymmetric Organocatalysis
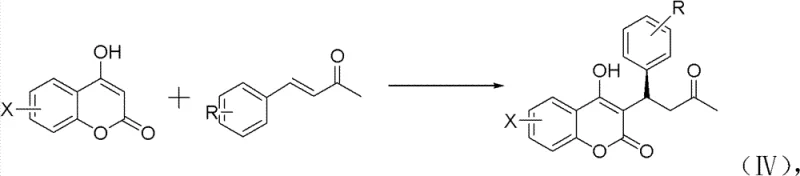
The pharmaceutical industry continuously seeks robust, scalable methodologies for producing single-enantiomer active pharmaceutical ingredients (APIs), particularly for anticoagulants where stereochemistry dictates potency and safety profiles. Patent CN102167689B introduces a groundbreaking preparation method for chiral warfarin and its derivatives, addressing critical limitations in existing synthetic routes. This technology leverages a sophisticated two-step sequence: an initial aldol condensation to generate substituted benzylideneacetone intermediates, followed by a highly stereoselective 1,4-addition reaction. By utilizing chiral primary amine catalysts in conjunction with Lewis acid cocatalysts, this process achieves excellent enantioselectivity under mild conditions, positioning it as a superior alternative for the commercial scale-up of complex pharmaceutical intermediates. The strategic integration of inexpensive organic catalysts not only simplifies the operational workflow but also aligns with modern green chemistry principles, making it an attractive option for a reliable pharmaceutical intermediate supplier aiming to optimize production efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of single-isomer warfarin has been plagued by inefficiencies inherent in classical resolution techniques. Traditional methods often involve synthesizing the racemic mixture first, followed by resolution using chiral acids or enzymatic processes, which theoretically caps the maximum yield at 50% unless dynamic kinetic resolution is employed. Furthermore, earlier asymmetric synthesis attempts frequently relied on expensive transition metal catalysts or harsh reaction conditions that posed significant challenges for industrial hygiene and waste management. These legacy processes often required rigorous purification steps to remove trace metal contaminants, adding substantial time and cost to the manufacturing timeline. Additionally, the sensitivity of some catalytic systems to moisture and oxygen necessitated specialized equipment and inert atmospheres, further complicating the cost reduction in anticoagulant manufacturing efforts for large-scale facilities.
The Novel Approach
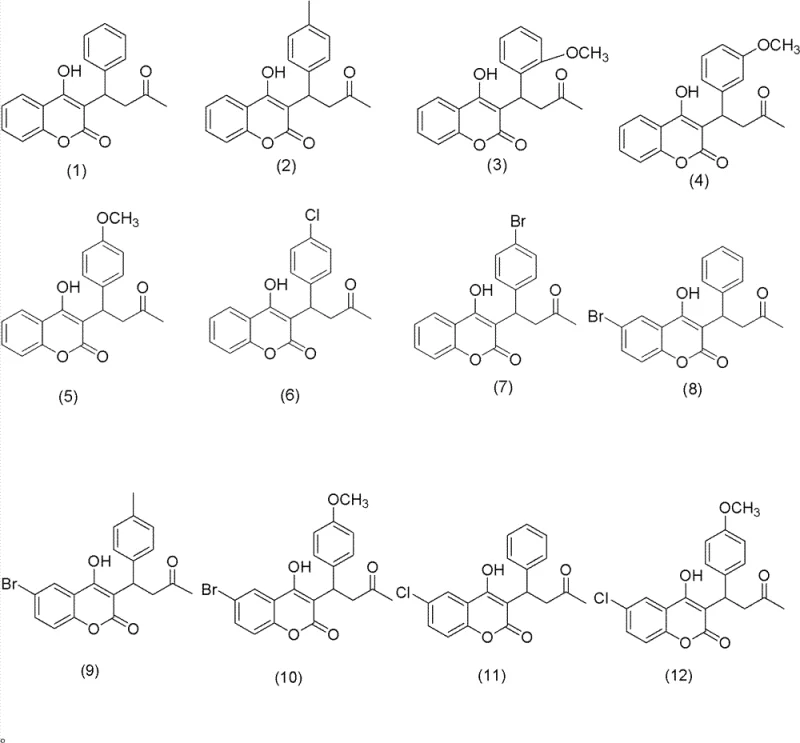
In stark contrast, the methodology outlined in CN102167689B offers a streamlined, direct asymmetric synthesis pathway that bypasses the need for resolution entirely. The process initiates with a base-catalyzed aldol condensation in a benign ethanol-water solvent system, generating the necessary enone precursors with high efficiency. The core innovation lies in the subsequent Michael addition step, where a chiral primary amine catalyst activates the substrate for nucleophilic attack by 4-hydroxycoumarin. This approach operates effectively at ambient temperatures (25-30°C) and utilizes readily available organic solvents like dioxane or THF. By eliminating the resolution step and avoiding precious metals, this route significantly enhances atom economy and reduces the environmental footprint. The versatility of this method is demonstrated by its ability to accommodate various substituents, allowing for the rapid generation of diverse warfarin analogues as shown in the structural overview below.
Mechanistic Insights into Chiral Amine-Catalyzed Michael Addition
The success of this synthetic strategy hinges on the precise interplay between the chiral primary amine catalyst and the Lewis acid cocatalyst during the 1,4-addition step. The chiral amine, such as (R,R)-diphenyl ethylene diamine or cyclohexanediamine, likely forms a transient chiral iminium or enamine intermediate with the benzylideneacetone, thereby lowering the LUMO energy of the electrophile and directing the facial selectivity of the nucleophilic attack. Simultaneously, the presence of additives like acetic acid and metallic salts (e.g., lithium perchlorate or iron trichloride) serves to activate the 4-hydroxycoumarin nucleophile and stabilize the transition state. This dual-activation mode ensures that the reaction proceeds with high stereocontrol, favoring the formation of the desired therapeutic enantiomer. The mechanistic elegance of this system allows for mild reaction conditions that preserve the integrity of sensitive functional groups, ensuring high-purity pharmaceutical intermediates suitable for downstream drug formulation without extensive remediation.
Furthermore, the impurity profile of the final product is meticulously controlled through the specificity of the organocatalyst. Unlike metal-catalyzed reactions that may generate side products via radical pathways or metal-mediated decomposition, this organic catalytic system promotes a clean concerted mechanism. The use of acetic acid as a proton source helps to facilitate the hydrolysis of the intermediate iminium species, releasing the product and regenerating the catalyst efficiently. This minimizes the formation of oligomeric byproducts or over-reaction species that often complicate purification. The result is a crude reaction mixture that requires minimal workup—typically involving simple extraction and pH adjustment—before final purification. This level of control over the reaction trajectory is critical for maintaining consistent quality across batches, a key requirement for regulatory compliance in the pharmaceutical sector.
How to Synthesize Chiral Warfarin Efficiently
The synthesis protocol described in the patent provides a clear roadmap for replicating this high-value transformation in a laboratory or pilot plant setting. The procedure is divided into two distinct stages: the preparation of the benzylideneacetone intermediate and the subsequent asymmetric coupling with 4-hydroxycoumarin. Operators must pay close attention to the molar ratios, particularly the slight excess of the enone intermediate (1:1.5 to 1:1.6 relative to coumarin) to drive the equilibrium towards completion. The detailed standardized synthesis steps for implementing this technology are provided in the guide below.
- Perform aldol condensation of substituted benzaldehyde and acetone in ethanol/water with NaOH to form benzylideneacetone intermediates.
- Conduct asymmetric 1,4-addition of the intermediate with 4-hydroxycoumarin using a chiral primary amine catalyst and Lewis acid cocatalyst.
- Purify the final chiral warfarin product through extraction, drying, and column chromatography or crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this organocatalytic route presents compelling economic and logistical benefits that extend beyond mere technical feasibility. By shifting away from resolution-based strategies, manufacturers can effectively double their theoretical output from the same amount of raw materials, drastically improving material utilization rates. The reliance on commodity chemicals like acetone, substituted benzaldehydes, and simple chiral diamines ensures a stable and resilient supply chain, mitigating risks associated with the scarcity of specialized reagents. Moreover, the elimination of heavy metal catalysts removes the need for costly and time-consuming metal scavenging steps, simplifying the downstream processing workflow and reducing waste disposal costs. These factors collectively contribute to a more agile and cost-effective production model.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the avoidance of racemic resolution, which inherently wastes half of the synthesized material. By directly accessing the chiral product, the process maximizes yield per batch. Additionally, the catalysts employed are organic amines rather than expensive noble metals like palladium or rhodium, leading to substantial savings in reagent costs. The mild reaction temperatures (25-30°C) also imply lower energy consumption for heating or cooling compared to cryogenic or high-temperature alternatives. These cumulative efficiencies translate into a significantly lower cost of goods sold (COGS), allowing for more competitive pricing in the global market.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route is bolstered by the widespread availability of its starting materials. Substituted benzaldehydes and 4-hydroxycoumarins are bulk chemicals produced by numerous suppliers worldwide, reducing dependency on single-source vendors. The tolerance of the reaction to standard laboratory solvents like ethanol, water, and dioxane further simplifies logistics, as these do not require special handling or storage conditions. This accessibility ensures that production schedules can be maintained consistently without interruptions due to raw material shortages, thereby reducing lead time for high-purity pharmaceutical intermediates and enhancing overall supply security.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns well with modern regulatory standards. The absence of toxic heavy metals simplifies the validation process for GMP manufacturing and reduces the burden of environmental, health, and safety (EHS) compliance. The aqueous workup and simple extraction procedures are easily adaptable to large-scale reactors, facilitating a smooth transition from gram-scale optimization to multi-ton commercial production. The reduced generation of hazardous waste and the use of greener solvent systems in the first step demonstrate a commitment to sustainable manufacturing practices, which is increasingly valued by downstream pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral warfarin synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating process transfer and procurement teams assessing vendor capabilities.
Q: What are the key advantages of this organocatalytic route over traditional resolution methods?
A: This method avoids the theoretical 50% yield loss inherent in racemic resolution, utilizes mild reaction conditions (25-30°C), and employs inexpensive, non-toxic amine catalysts instead of precious metals.
Q: Can this process be adapted for various warfarin derivatives?
A: Yes, the patent demonstrates broad substrate scope, successfully synthesizing derivatives with electron-donating (methoxy, methyl) and electron-withdrawing (chloro, bromo) groups on both the benzaldehyde and coumarin rings.
Q: Is the chiral catalyst reusable or cost-effective for large scale production?
A: The process uses commercially available chiral diamines (e.g., diphenyl ethylene diamine) at low loading (5 mol%), significantly reducing catalyst costs compared to transition metal complexes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Warfarin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the one described in CN102167689B can be seamlessly translated into industrial reality. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of chiral warfarin meets the highest international standards. Our infrastructure is designed to handle complex organic syntheses with precision, offering a secure partnership for your long-term supply needs.
We invite you to collaborate with us to leverage this advanced organocatalytic technology for your anticoagulant projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and reduce your overall manufacturing costs.
