Scalable Synthesis of Functionalized Tetramethyl Isoindoline Oxides for Advanced Covalent Organic Frameworks
The landscape of advanced material science is rapidly evolving, driven by the demand for functionalized precursors capable of constructing sophisticated organic porous covalent framework (COF) materials. A pivotal development in this sector is detailed in patent CN108101831B, which discloses a robust methodology for synthesizing aromatic amine compounds containing tetramethyl isoindoline or its oxide structure. Specifically, the patent highlights the preparation of 4,7-bis(4-aminophenyl)-1,1,3,3-tetramethylisoindoline oxide (Compound III) and its reduced counterpart, 4,7-bis(4-aminophenyl)-1,1,3,3-tetramethylisoindoline (Compound IV). These molecules are not merely academic curiosities; they serve as critical building blocks for embedding stable nitroxide radicals into imine-linked COFs, thereby imparting unique catalytic, magnetic, and energy storage properties to the final material. For R&D directors and procurement specialists in the fine chemical and advanced materials sectors, understanding the nuances of this synthesis is essential for securing a reliable supply chain of high-purity intermediates.
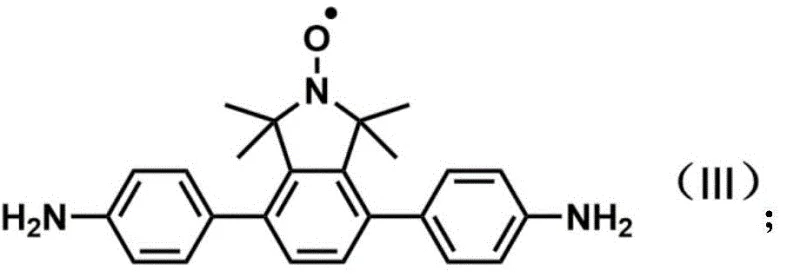
The strategic value of these compounds lies in their ability to functionalize porous frameworks without compromising structural integrity. The introduction of the tetramethyl isoindoline oxide moiety allows for the precise tuning of nitroxide radical concentration within the COF matrix, a feature highly desirable for applications ranging from alcohol catalytic oxidation to organic radical batteries. By mastering the synthesis of these complex architectures, manufacturers can offer clients tailored solutions that bridge the gap between theoretical material design and practical industrial application, ensuring that the next generation of electronic and energy storage materials can be realized with consistent quality and performance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
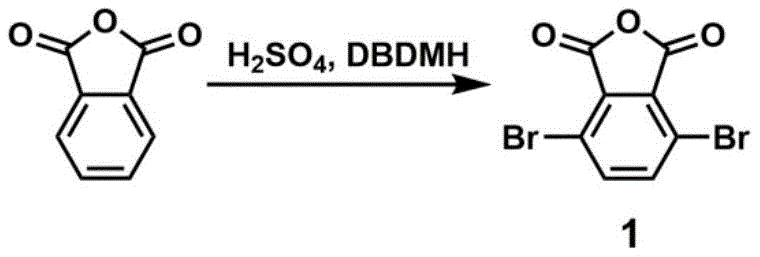
Historically, the synthesis of key intermediates like 3,6-dibromophthalic anhydride, which serves as the foundational scaffold for these tetramethyl isoindoline derivatives, has been fraught with significant safety and operational hazards. Traditional protocols typically rely on the direct bromination of phthalic anhydride using elemental liquid bromine in the presence of fuming sulfuric acid. This conventional approach necessitates harsh reaction conditions, often requiring heating under reflux for extended periods, such as 24 hours, to achieve acceptable conversion rates. The use of liquid bromine introduces severe handling risks due to its high volatility, corrosivity, and toxicity, demanding specialized containment infrastructure and rigorous safety protocols that drive up capital expenditure. Furthermore, the reliance on fuming sulfuric acid exacerbates these dangers, creating a reactive environment that is difficult to control on a large scale and generates substantial acidic waste streams that complicate downstream processing and environmental compliance.
The Novel Approach
In stark contrast, the methodology outlined in patent CN108101831B introduces a paradigm shift by utilizing 1,3-dibromo-5,5-dimethylhydantoin (DBDMH) as a solid brominating agent in conjunction with concentrated sulfuric acid. This innovative modification allows the bromination of phthalic anhydride to proceed under remarkably mild conditions, specifically at temperatures ranging from 20°C to 60°C, effectively eliminating the need for energy-intensive heating reflux. The transition from a liquid halogen source to a solid reagent fundamentally enhances process safety, mitigating the risks associated with vapor exposure and uncontrolled exotherms. Moreover, this method achieves the desired dibromination with high selectivity, yielding 3,6-dibromophthalic anhydride (Compound I) in a state suitable for subsequent transformation without the extensive purification burdens often associated with traditional halogenation. This streamlined approach not only safeguards personnel and equipment but also simplifies the workflow, making it an ideal candidate for cost reduction in advanced material manufacturing.
Mechanistic Insights into the Multi-Step Functionalization Strategy
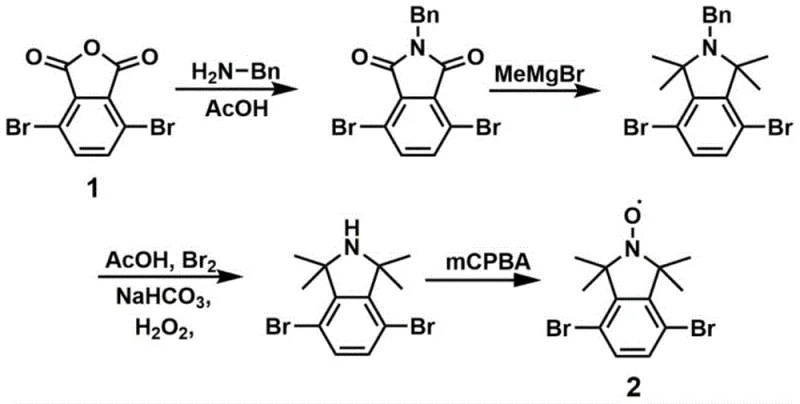
The construction of the target tetramethyl isoindoline oxide architecture involves a sophisticated cascade of organic transformations that demonstrate high chemoselectivity and functional group tolerance. The process initiates with the benzylation of the dibrominated anhydride using benzylamine in acetic acid, forming a protected isoindoline-1,3-dione intermediate. This is followed by a critical methylation step employing methyl magnesium bromide (MeMgBr), a Grignard reagent, which installs the four methyl groups at the 1,1,3,3-positions. This steric bulk is mechanistically crucial as it protects the sensitive nitrogen center and stabilizes the eventual radical species. Following methylation, a clever debenzylation-oxidation sequence is executed. The benzyl protecting group is removed using bromine in an acetic acid solution, and the resulting secondary amine is immediately oxidized, often using agents like m-chloroperoxybenzoic acid (mCPBA) or hydrogen peroxide systems, to generate the stable nitroxide radical core. This sequence ensures that the radical functionality is established prior to the final coupling step, preventing potential side reactions that could degrade the radical during harsh cross-coupling conditions.
The final assembly of the molecule is achieved through a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction, which links the functionalized isoindoline core with p-aminobenzeneboronic acid pinacol ester. This step is pivotal for extending the conjugation and introducing the terminal amine groups required for COF polymerization. The use of tetrakis(triphenylphosphine)palladium as the catalyst in a dioxane-water solvent system facilitates the efficient formation of carbon-carbon bonds between the aryl bromide and the boronic ester. Mechanistically, this cycle involves oxidative addition of the palladium into the carbon-bromine bond, transmetallation with the boronate species, and reductive elimination to forge the biaryl linkage. The robustness of this catalytic cycle allows for the preservation of the sensitive nitroxide radical and the primary amine functionalities, resulting in the high-purity 4,7-bis(4-aminophenyl)-1,1,3,3-tetramethylisoindoline oxide (Compound III) needed for downstream material synthesis.
How to Synthesize 4,7-bis(4-aminophenyl)-1,1,3,3-tetramethylisoindoline Oxide Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to maximize yield and minimize impurity profiles. The process begins with the careful preparation of the dibrominated starting material, followed by a sequential build-up of the isoindoline core. Each step, from the initial condensation to the final cross-coupling, demands specific stoichiometric ratios and temperature controls to ensure the integrity of the nitroxide radical. For instance, the methylation step must be conducted under an inert atmosphere to prevent the quenching of the Grignard reagent, while the oxidation step requires controlled addition of oxidants to avoid over-oxidation. Detailed standard operating procedures for executing this multi-step synthesis, including specific workup and purification protocols like column chromatography and recrystallization, are essential for reproducibility.
- Synthesize 3,6-dibromophthalic anhydride using phthalic anhydride and DBDMH in concentrated sulfuric acid under mild conditions.
- Perform benzylation with benzylamine followed by methylation using methyl magnesium bromide to form the tetramethyl isoindoline core.
- Execute debenzylation and oxidation to generate the nitroxide radical, followed by Suzuki coupling with p-aminobenzeneboronic acid pinacol ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the raw material sourcing and handling infrastructure. By replacing hazardous liquid bromine with solid DBDMH, facilities can significantly reduce the costs associated with hazardous material storage, ventilation systems, and emergency response preparedness. This shift translates directly into lower operational expenditures and a reduced regulatory burden, allowing for more flexible manufacturing scheduling. Furthermore, the mild reaction conditions eliminate the need for high-energy heating reflux systems, contributing to a smaller carbon footprint and lower utility costs, which aligns with modern sustainability goals in chemical manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous reagents like fuming sulfuric acid and liquid bromine leads to substantial cost savings in raw material procurement and waste disposal. The use of commodity chemicals such as phthalic anhydride and benzylamine ensures a stable and affordable supply base. Additionally, the high selectivity of the new bromination method reduces the formation of by-products, thereby minimizing the loss of valuable intermediates during purification and increasing the overall atom economy of the process. This efficiency gain allows for a more competitive pricing structure for the final high-purity intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent batch-to-batch quality, which is critical for customers producing sensitive electronic or energy storage materials. The avoidance of harsh conditions reduces the risk of reactor corrosion and equipment failure, leading to fewer unplanned downtime events and more reliable delivery schedules. Moreover, the scalability of the Suzuki coupling and Grignard steps means that production volumes can be ramped up quickly to meet surging demand for advanced COF precursors, providing a secure supply channel for long-term projects.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations like filtration, extraction, and distillation that are easily transferable from pilot plant to commercial production. The milder conditions generate less hazardous waste, simplifying effluent treatment and ensuring compliance with stringent environmental regulations. This environmental compatibility not only mitigates legal risks but also enhances the brand reputation of the supplier as a responsible partner in the green chemistry initiative, appealing to eco-conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized aromatic amine compounds. Understanding these details helps stakeholders evaluate the feasibility of integrating these materials into their own R&D pipelines. The answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industrial applications.
Q: What are the safety advantages of the new bromination method for 3,6-dibromophthalic anhydride?
A: The patented method replaces hazardous liquid bromine and fuming sulfuric acid with solid DBDMH and concentrated sulfuric acid, eliminating the need for heating reflux and significantly reducing operational risks.
Q: How is the nitroxide radical stability maintained during the synthesis?
A: The tetramethyl substitution at the 1,1,3,3-positions provides steric hindrance that stabilizes the nitroxide radical, allowing it to survive the subsequent Suzuki coupling conditions.
Q: Can this process be scaled for industrial production of COF precursors?
A: Yes, the use of mild reaction temperatures (20-60°C for bromination) and standard reagents like Grignard and Palladium catalysts makes the process highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,7-bis(4-aminophenyl)-1,1,3,3-tetramethylisoindoline Oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance intermediates play in the development of next-generation materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4,7-bis(4-aminophenyl)-1,1,3,3-tetramethylisoindoline oxide meets the exacting standards required for covalent organic framework synthesis. Our commitment to quality assurance means that you can trust our materials to perform consistently in your most demanding applications.
We invite you to collaborate with us to optimize your supply chain and accelerate your time-to-market. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your innovation goals. Let us be your partner in turning complex chemical challenges into commercial successes.
