Scalable Synthesis of Paliperidone Intermediate via Catalytic Cyclization and Hydrogenation
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antipsychotic agents, and the synthesis of paliperidone intermediates remains a focal point for process optimization. Patent CN101440093A discloses a highly efficient method for synthesizing 3-(2-chloroethyl)-6,7,8,9-tetrahydro-9-hydroxy-2-methyl-4H-pyrido[1,2-a]pyrimidin-4-one, a pivotal building block for paliperidone. This technology represents a significant departure from traditional multi-step protections, offering a streamlined three-step sequence that begins with the direct condensation of 2-amino-3-hydroxypyridine and alpha-acetyl-gamma-butyrolactone. By leveraging acid catalysis and transition metal hydrogenation, this route achieves superior selectivity and operational simplicity, addressing the longstanding challenges of incomplete cyclization and difficult purification associated with earlier methodologies. For global supply chains, this innovation translates into a more reliable source of high-purity pharmaceutical intermediates capable of meeting the rigorous demands of modern drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art techniques, such as those described in US5158952, rely heavily on cumbersome protection-deprotection strategies that inherently limit industrial scalability and economic efficiency. These conventional routes typically require the initial protection of the hydroxyl group on 2-amino-3-hydroxypyridine using benzyl chloride, followed by cyclization with alpha-acetyl-gamma-butyrolactone under harsh conditions involving phosphorus oxychloride (POCl3). A major deficiency in this legacy approach is the incomplete cyclization during the second step, which necessitates complex column chromatography for purification, a technique that is notoriously difficult to scale for tonnage production. Furthermore, the subsequent hydrogenation step often suffers from poor selectivity regarding dehalogenation, leading to significant impurity profiles that complicate the isolation of the target molecule. These technical bottlenecks result in lower overall yields and increased waste generation, posing substantial risks for procurement managers focused on cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in CN101440093A introduces a direct catalytic cyclization strategy that bypasses the need for hydroxyl protection entirely, fundamentally reshaping the synthetic landscape for this key intermediate. The process initiates with the reaction of 2-amino-3-hydroxypyridine and alpha-acetyl-gamma-butyrolactone in solvents such as xylene or toluene, catalyzed by organic or mineral acids at temperatures ranging from 120 to 150°C. This direct approach ensures complete cyclization, yielding Compound 3 with high efficiency and eliminating the need for chromatographic purification. The subsequent steps involve a highly selective transition-metal catalyzed hydrogenation of the pyridine ring followed by a specific chlorination of the primary alcohol. This streamlined workflow not only enhances the purity of the final product but also drastically simplifies the operational protocol, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
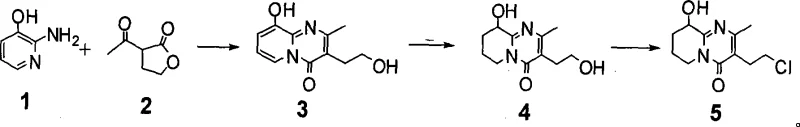
Mechanistic Insights into Acid-Catalyzed Cyclization and Hydrogenation
The core of this technological advancement lies in the precise control of reaction mechanisms, particularly during the initial cyclization and the subsequent ring reduction. In the first step, the acid catalyst facilitates the nucleophilic attack of the amino group on the carbonyl of the lactone, promoting ring closure to form the pyrimidinone structure without interfering with the phenolic hydroxyl group. This chemoselectivity is critical, as it prevents the formation of ether byproducts that typically plague unprotected syntheses. The reaction conditions, specifically the temperature range of 120 to 150°C and the use of water-separating reflux systems, drive the equilibrium towards the product by continuously removing water, thereby pushing the conversion rate to near completion. This mechanistic efficiency ensures that the crude product contains minimal starting material, reducing the burden on downstream purification units.
Furthermore, the hydrogenation step demonstrates exceptional regioselectivity, targeting the pyridine ring while preserving the integrity of the rest of the molecular framework. Utilizing catalysts such as 5% rhodium on carbon or palladium on carbon under hydrogen pressure at moderate temperatures (50 to 60°C), the process effectively reduces the aromatic pyridine ring to a tetrahydropyridine system. The choice of solvent, typically methanol or ethanol, plays a vital role in solubilizing the intermediate while maintaining catalyst activity. Crucially, this step avoids the dehalogenation issues seen in older methods because there are no halogen atoms present to be removed at this stage, a strategic advantage that simplifies the impurity profile. The final chlorination step then selectively converts the primary alcohol to a chloride using reagents like thionyl chloride, completing the synthesis with high fidelity.
How to Synthesize Paliperidone Intermediate Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and purity, particularly regarding the stoichiometry of the acid catalyst and the loading of the hydrogenation catalyst. The process is designed to be operationally robust, allowing for hot filtration and crystallization techniques that are far more scalable than column chromatography. Operators must maintain strict temperature controls during the exothermic cyclization and ensure adequate hydrogen pressure during the reduction phase to prevent stalling. The following guide outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for laboratory and pilot-scale execution.
- Perform acid-catalyzed cyclization of 2-amino-3-hydroxypyridine and alpha-acetyl-gamma-butyrolactone in a solvent like xylene at 120-150°C to obtain Compound 3.
- Conduct catalytic hydrogenation of the pyridine ring in Compound 3 using a transition metal catalyst such as Rh/C or Pd/C in methanol at 50-60°C to yield Compound 4.
- Execute selective chlorination of the primary alcohol in Compound 4 using a chlorinating agent like thionyl chloride to finalize the paliperidone intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the adoption of this novel synthesis route offers tangible benefits that extend beyond mere technical feasibility, directly impacting the bottom line through process intensification. By eliminating the benzyl protection and deprotection steps, the process removes two entire unit operations along with the associated reagents, solvents, and waste disposal costs. This reduction in step count inherently shortens the manufacturing cycle time, allowing for faster turnover and improved responsiveness to market demand fluctuations. Additionally, the ability to purify intermediates via simple crystallization and filtration rather than expensive chromatography significantly lowers the cost of goods sold (COGS), making the final intermediate more price-competitive in the global marketplace.
- Cost Reduction in Manufacturing: The removal of protection groups and the avoidance of column chromatography lead to substantial savings in raw material consumption and solvent usage. Without the need for benzyl chloride and subsequent hydrogenolysis reagents, the bill of materials is streamlined, and the elimination of silica gel and large volumes of eluents reduces waste treatment expenses. This leaner chemical footprint translates directly into a more economical production model, enabling suppliers to offer competitive pricing without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the catalytic hydrogenation step, which achieves yields up to 99.8%, ensures a consistent output of high-quality intermediate, minimizing the risk of batch failures. High selectivity in the chlorination step further guarantees that the final product meets strict specification limits, reducing the likelihood of rejected shipments. This reliability is crucial for maintaining uninterrupted supply lines to downstream API manufacturers, ensuring that production schedules are met without unexpected delays caused by purification bottlenecks.
- Scalability and Environmental Compliance: The process utilizes common industrial solvents like xylene and methanol, which are readily available and easily recycled, supporting sustainable manufacturing practices. The high atom economy of the direct cyclization and the absence of heavy metal contaminants from complex protection schemes simplify wastewater treatment protocols. Consequently, scaling this process from kilogram to multi-ton production is straightforward, aligning with environmental regulations and reducing the regulatory burden associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders evaluating its potential for integration into existing supply chains. The following questions address common inquiries regarding catalyst selection, yield optimization, and purity control, drawing directly from the experimental data provided in the patent documentation. These insights are intended to clarify the operational advantages and assist technical teams in assessing the feasibility of adopting this route for commercial production.
Q: What are the key advantages of this new synthesis route over conventional methods?
A: The novel route eliminates the need for benzyl protection and deprotection steps required in prior art (US5158952), significantly simplifying the process. It achieves complete cyclization with high selectivity, avoiding difficult column purifications and improving overall yield to 70-90% in the first step and up to 99.8% in the hydrogenation step.
Q: Which catalysts are suitable for the hydrogenation step in this process?
A: The process utilizes transition metal catalysts such as Raney nickel, palladium carbon (Pd/C), rhodium carbon (Rh/C), or platinum carbon. Specifically, 5% rhodium carbon has been demonstrated to provide excellent conversion rates under mild conditions (50-60°C) while maintaining high selectivity for the pyridine ring reduction.
Q: How does this method address impurity control during chlorination?
A: The method employs selective chlorination reagents like thionyl chloride or phosphorus trichloride mixtures which specifically target the primary alcohol group. This high selectivity prevents side reactions on the sensitive pyrimidinone ring, ensuring the final product meets stringent purity specifications without complex downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paliperidone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes in maintaining a competitive edge in the pharmaceutical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the catalytic cyclization process described in CN101440093A can be seamlessly transferred to industrial scale. We are committed to delivering high-purity pharmaceutical intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs and state-of-the-art manufacturing facilities. Our capability to handle complex catalytic hydrogenations and selective functionalizations positions us as a strategic partner for companies seeking to optimize their paliperidone supply chain.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your production efficiency and reduce overall manufacturing costs.
