Scalable Photocatalytic Synthesis of Alpha-Haloketones for Advanced Pharmaceutical Intermediates
The landscape of organic synthesis is undergoing a transformative shift towards greener, more sustainable methodologies, particularly in the production of critical building blocks for the life sciences sector. Patent CN114539040A introduces a groundbreaking photocatalytic method for synthesizing alpha-haloketone compounds, addressing long-standing challenges associated with traditional halogenation processes. This technology leverages visible light irradiation to drive the oxidative transposition of olefin compounds in the presence of halides, utilizing cost-effective semiconductor photocatalysts rather than expensive noble metals or hazardous reagents. For R&D directors and procurement specialists, this represents a significant opportunity to streamline the supply chain for high-purity pharmaceutical intermediates while adhering to increasingly stringent environmental regulations. The process demonstrates exceptional versatility, accommodating a wide range of substituted styrenes and delivering high conversion rates that are essential for efficient industrial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-halogenated ketones has relied heavily on direct halogenation of ketones using aggressive reagents such as molecular chlorine, liquid bromine, or N-halosuccinimides like NBS and NCS. These traditional pathways are fraught with significant drawbacks, including the generation of toxic by-products, the requirement for harsh reaction conditions involving strong acids or bases, and the inherent safety risks associated with handling volatile halogens. Furthermore, existing methods often suffer from poor selectivity, leading to complex mixtures of mono- and poly-halogenated species that necessitate energy-intensive purification steps. The reliance on stoichiometric amounts of hazardous halogenating agents not only inflates the cost of goods sold but also creates substantial waste disposal burdens, making these processes economically and environmentally unsustainable for large-scale commercial operations in the fine chemical industry.
The Novel Approach
In stark contrast, the novel photocatalytic approach detailed in the patent utilizes a radical-mediated mechanism driven by visible light, enabling the direct conversion of readily available olefins into valuable alpha-haloketones. By employing semiconductor photocatalysts such as nickel-modified graphitic carbon nitride, the reaction proceeds under mild conditions without the need for external oxidants or toxic halogen sources. This methodology effectively bypasses the safety and environmental pitfalls of conventional chemistry, offering a streamlined one-step transformation that minimizes waste generation. The ability to use simple metal halides as the halogen source further reduces raw material costs, while the high selectivity observed across various substrates ensures that downstream purification is simplified, thereby enhancing the overall economic viability of producing these critical intermediates.
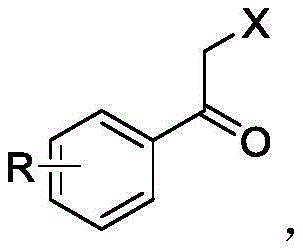
Mechanistic Insights into Photocatalytic Oxidative Halogenation
The core of this innovation lies in the sophisticated interplay between the semiconductor photocatalyst and the substrate under illumination. Upon absorption of photons, the semiconductor catalyst generates electron-hole pairs that facilitate the activation of the halide species and the olefin substrate simultaneously. This photo-induced redox process initiates a radical cascade where the halogen radical adds to the double bond of the olefin, followed by oxidation and rearrangement to form the carbonyl functionality. The use of metal-loaded catalysts, such as nickel-decorated carbon nitride, enhances charge separation efficiency and provides active sites that lower the activation energy for the rate-determining steps. This mechanistic pathway ensures that the reaction proceeds with high atom economy, as the oxygen atom required for the ketone formation is typically derived from ambient air or the solvent system, eliminating the need for stoichiometric oxidants.
From an impurity control perspective, the mild nature of the photocatalytic conditions plays a pivotal role in maintaining product integrity. Unlike thermal halogenation which can promote over-halogenation or skeletal rearrangements due to high energy inputs, the photo-driven process operates at near-ambient temperatures, preserving sensitive functional groups on the aromatic ring. The high selectivity reported in the patent examples, often exceeding 80% for specific isomers, indicates that the catalyst surface exerts a controlling influence on the orientation of the radical addition. This precision is crucial for pharmaceutical applications where regio-isomeric purity is a strict requirement, as it reduces the burden on chromatographic separation and crystallization steps, ultimately leading to a cleaner final product profile that meets rigorous quality standards.
How to Synthesize Alpha-Haloketones Efficiently
The practical implementation of this synthesis route involves a straightforward protocol that is amenable to both laboratory optimization and industrial scale-up. The process begins with the preparation of a homogeneous suspension containing the olefin substrate, a metal halide source, and the photocatalyst in a suitable organic solvent. Reaction parameters such as light intensity and temperature are carefully controlled to maximize the quantum yield of the transformation. While the general procedure is robust, specific optimization of catalyst loading and irradiation time may be required for different substrate classes to achieve optimal conversion. For a detailed breakdown of the standardized synthetic steps and specific reaction conditions validated in the patent examples, please refer to the guide below.
- Prepare a mixed solution by dispersing olefin compounds and halides in a solvent with a semiconductor photocatalyst (e.g., Ni-loaded g-C3N4).
- Subject the mixture to visible light irradiation (0.01-50W/cm²) while stirring at temperatures between -50°C and 200°C for 1 to 24 hours.
- Isolate the final alpha-haloketone product by drying and concentrating the organic phase after the reaction is complete.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this photocatalytic technology offers compelling strategic advantages that extend beyond mere technical feasibility. The shift away from hazardous reagents and complex multi-step sequences translates directly into a more resilient and cost-efficient supply chain. By simplifying the synthetic route and utilizing abundant starting materials, manufacturers can mitigate the risks associated with raw material volatility and regulatory compliance. This section outlines the key commercial benefits that make this technology a superior choice for sourcing high-quality chemical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and toxic halogenating agents results in a substantial decrease in raw material expenditures. Traditional methods often require costly reagents like NBS or specialized organometallic catalysts, whereas this process utilizes inexpensive earth-abundant metals like nickel or iron and simple halide salts. Furthermore, the reduction in purification complexity due to high selectivity lowers the operational costs associated with solvent usage and waste treatment, driving down the overall cost of production for alpha-haloketone derivatives.
- Enhanced Supply Chain Reliability: The reliance on widely available olefin precursors and common inorganic halides ensures a stable and continuous supply of raw materials, reducing the risk of production delays caused by sourcing bottlenecks. Unlike processes dependent on specialized or regulated reagents, the inputs for this photocatalytic method are commodity chemicals with robust global supply networks. This accessibility allows for greater flexibility in procurement planning and ensures that manufacturing schedules can be maintained consistently, even in fluctuating market conditions.
- Scalability and Environmental Compliance: The use of visible light as the energy source and the absence of hazardous by-products align perfectly with modern green chemistry principles, facilitating easier regulatory approval and permitting for large-scale facilities. The process generates minimal waste and avoids the use of volatile organic compounds associated with traditional halogenation, simplifying effluent treatment and reducing the environmental footprint. This sustainability profile not only future-proofs the manufacturing process against tightening environmental regulations but also enhances the brand value of the final pharmaceutical products by supporting greener supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the process capabilities and limitations for potential partners and stakeholders.
Q: What are the advantages of this photocatalytic method over traditional halogenation?
A: Unlike traditional methods requiring toxic halogen sources like chlorine gas or NBS, this photocatalytic approach uses inexpensive metal halides and visible light, significantly reducing environmental hazards and operational costs.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the patent demonstrates high conversion rates (up to 99%) and selectivity using readily available styrene derivatives, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What types of substrates are compatible with this synthesis route?
A: The method exhibits broad substrate scope, successfully converting various substituted styrenes (including fluoro, bromo, chloro, and trifluoromethyl derivatives) into their corresponding alpha-haloketones with high efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Haloketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced photocatalytic technologies in reshaping the production of critical pharmaceutical intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of alpha-haloketone delivered meets the exacting standards required by global drug developers. We are committed to leveraging cutting-edge chemistry to provide our clients with a competitive edge in the marketplace.
We invite you to collaborate with us to explore how this efficient synthesis route can optimize your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the tangible economic benefits of switching to this greener methodology. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a reliable, high-quality supply of these essential building blocks for your next breakthrough therapy.
