Scalable Manufacturing of 3 5 Diethylisoxazole 4 Carboxylic Acid for Dermatological APIs
Scalable Manufacturing of 3 5 Diethylisoxazole 4 Carboxylic Acid for Dermatological APIs
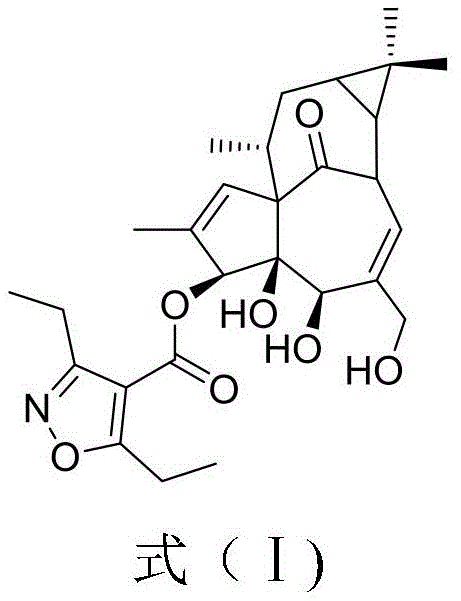
The pharmaceutical landscape for dermatological treatments is undergoing a significant transformation, driven by the need for safer and more effective therapies for conditions such as Actinic Keratosis (AK). Central to this evolution is the development of novel ingenol derivatives, where the structural integrity of the side chain plays a pivotal role in efficacy and safety profiles. Patent CN114716389A introduces a groundbreaking synthetic methodology for 3,5-diethylisoxazole-4-carboxylic acid, a critical building block that replaces the traditional angelic acid moiety in next-generation ingenol esters. This innovation addresses the longstanding challenges associated with impurity profiles and side effects observed in earlier generations of photo-linear keratosis drugs. By leveraging a streamlined condensation strategy, this patent outlines a pathway that not only enhances chemical purity to over 99 percent but also drastically simplifies the operational complexity typically associated with heterocyclic synthesis. For R&D directors and procurement specialists alike, understanding the nuances of this synthetic route is essential for securing a reliable supply chain for high-value dermatological active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isoxazole-based intermediates for complex natural product derivatives has been plagued by inefficient multi-step sequences that rely on hazardous reagents and generate substantial waste. Traditional routes often involve the initial formation of oximes using hydroxylamine hydrochloride, followed by cyclization steps that require rigorous control of pH and temperature to prevent decomposition. As detailed in the comparative examples of the patent data, these legacy methods suffer from poor atom economy and difficult purification protocols, often necessitating extensive column chromatography which is untenable for industrial scale-up. Furthermore, the use of stoichiometric amounts of strong bases and the generation of saline wastewater during workup pose significant environmental compliance hurdles. The inability to consistently achieve high purity without resorting to recrystallization from expensive solvent systems has traditionally driven up the cost of goods sold, creating bottlenecks for procurement teams aiming to reduce the overall manufacturing budget for dermatological APIs.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology disclosed in CN114716389A employs a direct and highly efficient condensation reaction between methyl 3-oxopentanoate and propionyl chloride. This novel approach bypasses the unstable oxime intermediate entirely, utilizing a robust base-catalyzed cyclization that proceeds with remarkable selectivity. The patent highlights three distinct variations of this core transformation, utilizing sodium ethoxide, magnesium-mediated activation, or magnesium chloride/pyridine systems, all of which demonstrate superior operability. By shifting the synthetic logic to a one-pot or near one-pot sequence, the new method significantly reduces the number of unit operations, thereby minimizing solvent consumption and processing time. This strategic pivot not only enhances the overall yield, with Example 1 demonstrating a respectable 46 percent yield alongside exceptional purity, but also ensures that the final crystalline product can be isolated through standard extraction and drying techniques, eliminating the need for resource-intensive purification steps that hinder commercial viability.
Mechanistic Insights into Base-Catalyzed Isoxazole Formation
The core of this technological breakthrough lies in the precise manipulation of enolate chemistry to facilitate the construction of the isoxazole ring system. In the preferred embodiment utilizing metallic sodium and ethanol, the in situ generation of sodium ethoxide creates a strongly nucleophilic environment that deprotonates the alpha-position of methyl 3-oxopentanoate. This enolate species then undergoes a nucleophilic acyl substitution with propionyl chloride, forming a beta-diketone intermediate which is primed for cyclization. The subsequent addition of water and controlled heating promotes the intramolecular condensation with the nitrogen source inherent in the reaction matrix or added subsequently, closing the ring to form the stable isoxazole architecture. This mechanism is particularly elegant because it avoids the harsh acidic conditions often required for traditional isoxazole synthesis, thereby preserving sensitive functional groups that might be present in more complex downstream derivatives. The careful temperature control, maintaining the reaction at 10 +/- 3 degrees Celsius during the addition of the acid chloride, is critical to suppressing side reactions such as polymerization or over-acylation, ensuring a clean reaction profile that translates directly to high crude purity.
From an impurity control perspective, the choice of solvent and base plays a decisive role in defining the final quality of the 3,5-diethylisoxazole-4-carboxylic acid. The patent data elucidates that while magnesium-mediated pathways in toluene or dichloromethane are feasible, the sodium ethoxide route in ethanol offers the most favorable balance between reactivity and selectivity. The polar protic nature of ethanol stabilizes the transition states involved in the cyclization, while the solubility profile of the intermediates allows for easy separation of inorganic salts upon aqueous workup. This mechanistic understanding is vital for process chemists aiming to replicate the >99 percent purity reported in the patent, as deviations in base strength or solvent dryness can lead to the formation of regioisomers or hydrolysis byproducts. By strictly adhering to the specified thermal profile and reagent stoichiometry, manufacturers can effectively minimize the formation of high-molecular-weight impurities, ensuring that the intermediate meets the stringent specifications required for GMP production of the final ingenol derivative drug substance.
How to Synthesize 3,5-Diethylisoxazole-4-carboxylic acid Efficiently
To successfully implement this synthesis in a pilot or production plant, operators must follow a disciplined protocol that prioritizes thermal management and reagent quality. The process begins with the careful preparation of the catalytic base, followed by the sequential addition of the keto-ester and the acid chloride under inert atmosphere conditions to prevent moisture ingress which could quench the reactive intermediates. The reaction mixture requires sustained stirring over a 12-hour period at ambient temperature to ensure complete conversion, after which a controlled quench with water dissolves the inorganic salts and facilitates phase separation. While the patent provides specific gram-scale examples, the underlying principles of heat dissipation and mixing efficiency remain constant regardless of batch size, making this a robust candidate for technology transfer. For a comprehensive breakdown of the exact operational parameters, reagent grades, and safety precautions required for execution, please refer to the standardized synthesis guide below.
- Prepare sodium ethoxide by refluxing metallic sodium in ethanol, then cool to 20-30°C before adding methyl 3-oxopentanoate.
- Cool the mixture to 0°C and slowly add propionyl chloride while maintaining the temperature at 10±3°C to control exothermic reaction.
- Warm to room temperature for 12 hours, quench with water, extract with ethyl acetate, and purify to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route represents a strategic opportunity to optimize the cost structure and reliability of the dermatological API supply chain. The primary economic driver here is the substitution of complex, multi-step sequences with a streamlined process that utilizes commodity-grade starting materials. Methyl 3-oxopentanoate and propionyl chloride are widely available bulk chemicals with stable pricing, insulating the manufacturing process from the volatility often seen with specialized fine chemical reagents. Furthermore, the elimination of transition metal catalysts removes the necessity for expensive and time-consuming heavy metal scavenging steps, which are a significant cost center in modern pharmaceutical manufacturing. This simplification of the downstream processing workflow translates directly into reduced cycle times and lower utility consumption, providing a tangible competitive advantage in a market where speed to market is critical for capturing value in the generic and biosimilar sectors.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived principally from the drastic reduction in solvent usage and the avoidance of chromatographic purification. By achieving high crude purity through crystallization or simple extraction, the facility saves significantly on solvent recovery costs and waste disposal fees associated with silica gel columns. Additionally, the high atom economy of the condensation reaction means that less raw material is wasted as byproduct, maximizing the yield per kilogram of input. This efficiency allows for a substantial reduction in the variable cost of goods, enabling procurement teams to negotiate more favorable terms with downstream API manufacturers or to improve margin profiles for internal production units without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on a short list of robust, non-proprietary raw materials that are sourced from multiple global suppliers. Unlike processes dependent on custom-synthesized chiral auxiliaries or rare earth catalysts, this method mitigates the risk of single-source bottlenecks that can halt production lines. The operational simplicity of the reaction, which tolerates standard stainless steel reactor equipment without requiring exotic lining or specialized agitation, further ensures that the process can be easily replicated across different manufacturing sites. This flexibility is crucial for maintaining business continuity and ensuring that the supply of this critical intermediate remains uninterrupted even in the face of regional logistical disruptions or geopolitical instability affecting specific chemical supply routes.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this synthesis aligns well with green chemistry principles by minimizing the generation of hazardous waste streams. The absence of heavy metals and the use of ethanol, a relatively benign solvent, simplify the effluent treatment process and reduce the regulatory burden associated with volatile organic compound (VOC) emissions. The process is inherently scalable, as the exothermic nature of the acylation step can be managed effectively through standard jacketed cooling systems in large-scale reactors. This scalability ensures that the technology can grow with market demand, supporting the transition from clinical trial material to commercial tonnage production without the need for fundamental process re-engineering, thereby securing long-term supply continuity for partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3,5-diethylisoxazole-4-carboxylic acid. These insights are derived directly from the experimental data and technical disclosures within the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their existing portfolios and assess the potential for process optimization. We encourage technical teams to review these points carefully to align their expectations with the proven capabilities of this synthetic methodology.
Q: What is the achievable purity of 3,5-diethylisoxazole-4-carboxylic acid using this method?
A: According to patent CN114716389A, the optimized synthesis route utilizing sodium ethoxide catalysis can achieve a final product purity exceeding 99%, specifically demonstrated at 99.6% in experimental examples.
Q: How does this new method compare to traditional hydroxylamine-based routes?
A: The novel approach eliminates the need for hydroxylamine hydrochloride and complex multi-step oxime formation, significantly simplifying the workflow and reducing solvent consumption compared to conventional methods.
Q: Is this intermediate suitable for large-scale commercial production?
A: Yes, the process utilizes readily available commodity chemicals like methyl 3-oxopentanoate and propionyl chloride, and avoids expensive transition metal catalysts, making it highly viable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Diethylisoxazole-4-carboxylic acid Supplier
As the demand for advanced dermatological treatments continues to rise, securing a partner with the technical expertise to deliver high-quality intermediates is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to support your drug development lifecycle. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 3,5-diethylisoxazole-4-carboxylic acid meets the exacting standards required for GMP API synthesis. We understand that consistency is key in pharmaceutical manufacturing, and our dedicated process chemistry team is committed to maintaining the highest levels of quality control throughout the production cycle.
We invite you to collaborate with us to unlock the full potential of this innovative synthetic route for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can enhance your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to excellence can drive success in your next project milestone.
