Advanced Synthesis of Chiral Doranidazole Intermediates for Commercial Scale-up
Advanced Synthesis of Chiral Doranidazole Intermediates for Commercial Scale-up
The development of efficient synthetic routes for oncology drug intermediates remains a pivotal challenge in modern pharmaceutical manufacturing, particularly for radiosensitizers like Doranidazole. Patent CN101805323B introduces a robust and scalable methodology for synthesizing specific diastereoisomers of the key intermediate, (4R,5R)-4,5-di(benzoyl oxido-methyl)-1,3-dioxolane and its enantiomer. This technology addresses critical bottlenecks in prior art by utilizing readily available chiral pool starting materials, specifically L-diethyl tartrate or D-diethyl tartrate, to construct the core dioxolane scaffold. The process involves a strategic sequence of cyclization, reduction, and acylation that ensures high stereochemical fidelity and operational safety. By shifting away from hazardous reagents and difficult-to-source chiral precursors, this patent provides a foundation for reliable high-purity pharmaceutical intermediate production that aligns with stringent global regulatory standards.
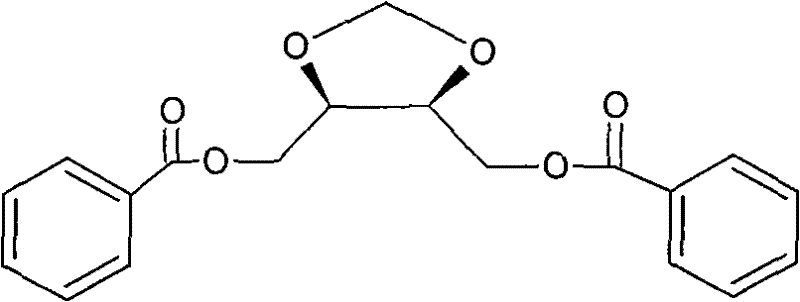
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dioxolane-based intermediates for Doranidazole has been plagued by significant safety hazards and supply chain fragility. Prior art, such as that disclosed in EP0632028, relied heavily on the use of methylal (dimethoxymethane) and vanadium pentoxide for the cyclization step. Methylal is characterized by a low boiling point and inherent instability, presenting a substantial explosion risk during large-scale operations, while vanadium pentoxide reacts exothermically with water, complicating waste treatment and process control. Furthermore, alternative pathways described in patents like JP2001-158782 necessitate the use of specific optically active starting materials, such as 1,4-dibenzoyloxybutanetetrol, which are not only difficult to procure commercially but also result in undesirable yields and purity profiles. These legacy methods create unacceptable barriers to entry for generic manufacturers seeking cost-effective and safe production capabilities.
The Novel Approach
The methodology outlined in CN101805323B represents a paradigm shift by leveraging the abundance and stability of diethyl tartrate isomers. Instead of grappling with volatile acetals or exotic chiral alcohols, this route initiates with a Lewis acid-catalyzed condensation between diethyl tartrate and paraformaldehyde. This approach eliminates the need for explosive methylal and toxic heavy metal catalysts, replacing them with safer, standard laboratory reagents like boron trifluoride etherate and anhydrous calcium chloride. The subsequent transformation involves a controlled reduction followed by a mild acylation, ensuring that the sensitive ester functionalities remain intact throughout the synthesis. This streamlined three-step process not only simplifies the operational workflow but also drastically improves the feasibility of industrial amplification, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks.
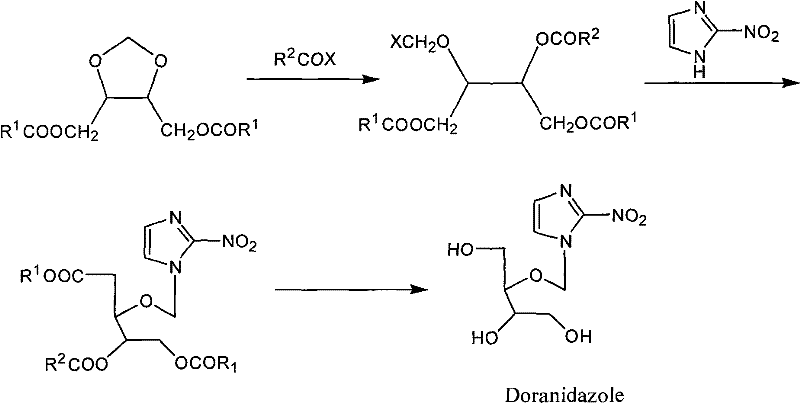
Mechanistic Insights into BF3-Catalyzed Cyclization and Stereoselective Acylation
The core of this synthetic strategy lies in the precise control of the cyclization mechanism, where boron trifluoride acts as a potent Lewis acid to activate the formaldehyde source derived from paraformaldehyde. In the presence of anhydrous calcium chloride, which serves as an effective water scavenger, the reaction drives the equilibrium towards the formation of the 1,3-dioxolane ring with high efficiency. The chirality of the starting tartrate is perfectly preserved during this ring-closing event, dictating the stereochemistry of the resulting (4R,5R) or (4S,5S) configuration. A critical mechanistic nuance identified in this patent is the workup procedure; the use of organic bases like triethylamine for neutralization is essential to quench the acidic catalyst without inducing hydrolysis of the newly formed ester groups, a side reaction that frequently plagues aqueous workups in similar acidic environments. This attention to chemical compatibility ensures that the intermediate proceeds to the next step with minimal degradation.
Following the cyclization, the reduction step utilizes lithium aluminum hydride to convert the diester into the corresponding diol, a transformation that requires careful temperature management to prevent over-reduction or ring opening. The final acylation step employs benzoyl chloride in the presence of catalytic DMAP and stoichiometric triethylamine, facilitating the formation of the stable benzoyl protecting groups. This specific protection pattern is crucial for the downstream synthesis of Doranidazole, as it stabilizes the molecule against unwanted side reactions while maintaining the necessary solubility profile for further functionalization. The entire sequence is designed to minimize impurity generation, with chromatographic purification strategies integrated to ensure the final product meets the rigorous purity specifications required for oncology applications.
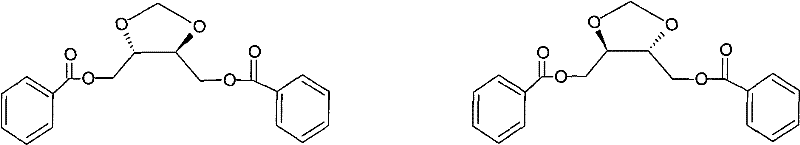
How to Synthesize Doranidazole Intermediate Efficiently
The synthesis protocol detailed in the patent offers a clear pathway for producing high-quality intermediates, beginning with the cyclization of diethyl tartrate in toluene at elevated temperatures. The process emphasizes the importance of strict temperature control during the addition of reagents and the subsequent neutralization phase to maintain product integrity. Following the isolation of the cyclic diester, the reduction is performed in tetrahydrofuran under reflux, followed by a careful quench to isolate the diol intermediate. The final step involves low-temperature acylation to install the benzoyl groups, yielding the target compound with excellent optical purity. For a comprehensive breakdown of the specific molar ratios, reaction times, and purification techniques required to replicate this success, please refer to the standardized guide below.
- Cyclization of L- or D-diethyl tartrate with paraformaldehyde using BF3 etherate and anhydrous CaCl2 in toluene.
- Reduction of the resulting cyclic diester using Lithium Aluminum Hydride (LiAlH4) in THF to form the diol.
- Acylation of the diol with benzoyl chloride in the presence of triethylamine and DMAP to yield the final protected intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the transition to this tartrate-based methodology offers profound advantages in terms of raw material security and cost structure. By eliminating the dependency on specialized, hard-to-source chiral building blocks, manufacturers can leverage the global commodity market for diethyl tartrate, which is produced in massive volumes for the food and fragrance industries. This shift significantly reduces the risk of supply chain disruptions caused by single-source dependencies on exotic fine chemicals. Furthermore, the removal of hazardous reagents like methylal and vanadium pentoxide simplifies the regulatory compliance landscape, reducing the costs associated with hazardous waste disposal and specialized storage infrastructure. These factors combine to create a more resilient and economically viable supply chain for high-value oncology intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents directly translates to lower operational expenditures. By avoiding the need for specialized safety equipment required to handle explosive methylal and toxic vanadium compounds, facilities can reduce capital investment and maintenance costs. Additionally, the use of common solvents like toluene and dichloromethane, along with standard bases like triethylamine, ensures that consumable costs remain predictable and low. The simplified purification process, which avoids complex resolution steps, further enhances the overall economic efficiency of the production line, allowing for substantial cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Sourcing L- or D-diethyl tartrate is significantly more reliable than procuring custom-synthesized chiral alcohols, as these materials are available from multiple global suppliers with consistent quality. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. The stability of the reagents also means that inventory can be held for longer periods without degradation, providing greater flexibility in production planning. Consequently, this leads to reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API synthesis schedules are met without delay.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, utilizing reaction conditions that are easily managed in standard stainless steel reactors. The absence of heavy metal catalysts simplifies the environmental footprint, making it easier to meet increasingly strict discharge regulations. The straightforward workup procedures minimize solvent usage and waste generation, aligning with green chemistry principles. This scalability ensures that production can be ramped up from pilot plant quantities to multi-ton annual capacities without the need for fundamental process redesigns, securing long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on process robustness and product quality. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their own manufacturing portfolios.
Q: Why is the use of diethyl tartrate preferred over methylal in this synthesis?
A: Diethyl tartrate is a stable, commercially available chiral pool material, whereas methylal (dimethoxymethane) used in prior art is volatile, unstable, and potentially explosive, posing significant safety risks during industrial amplification.
Q: How does this method ensure high optical purity?
A: The method utilizes optically active starting materials (L- or D-diethyl tartrate) which transfer their chirality directly to the dioxolane ring during the cyclization step, ensuring the formation of specific (4R,5R) or (4S,5S) diastereoisomers without the need for complex resolution steps.
Q: What is the critical control point during the cyclization step?
A: The neutralization step is critical; using organic bases like triethylamine effectively neutralizes the corrosive boron trifluoride catalyst without causing hydrolysis of the sensitive ester groups, which is a common failure point in acidic workups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Doranidazole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of secure and high-quality supply chains for oncology therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our capability to implement the safe and efficient tartrate-based synthesis described in CN101805323B positions us as a strategic partner for companies seeking to optimize their Doranidazole supply chain.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic potential of switching to this safer methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your development timelines are supported by a robust and reliable manufacturing partner dedicated to excellence in fine chemical production.
