Advanced Supramolecular Refining Technology for High-Purity Synthetic Capsaicin Production
Advanced Supramolecular Refining Technology for High-Purity Synthetic Capsaicin Production
The pharmaceutical and fine chemical industries constantly face the challenge of purifying complex organic molecules where structural isomers possess nearly identical physical properties. Patent CN110698359A introduces a groundbreaking refining method for high-purity synthetic capsaicin that leverages the precision of supramolecular chemistry. This technology utilizes a specifically engineered calixarene derivative capable of host-guest molecular recognition to selectively identify and sequester impurities such as (trans) 8-methyl-N-vanillyl-6-nonenamide. By shifting the paradigm from brute-force physical separation to intelligent molecular recognition, this process achieves purity levels exceeding 99.8%, addressing a critical bottleneck in the manufacturing of top-tier capsaicin derivatives used in medical and industrial applications.
For R&D directors and procurement specialists, this patent represents a significant leap forward in process chemistry. Traditional methods often rely on energy-intensive rectification of precursors or repetitive recrystallization, which yields diminishing returns when dealing with stubborn isomeric contaminants. The approach detailed in CN110698359A offers a streamlined, green alternative that not only enhances product quality but also introduces a recyclable catalytic-like system. As a reliable synthetic capsaicin supplier, understanding these underlying technological shifts is crucial for maintaining a competitive edge in the global market for fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of synthetic capsaicin has been plagued by the difficulty of separating structurally similar impurities, particularly unsaturated analogs like (trans) 8-methyl-N-vanillyl-6-nonenamide. Conventional wisdom dictated that controlling these impurities required rigorous rectification of the fatty acid precursors, specifically 8-methyl-6-nonenoic acid, before the amidation step. This upstream approach is fraught with inefficiencies, including high energy consumption due to the need for high-vacuum distillation and significant yield losses. Furthermore, even with precise precursor control, trace impurities often persist through the synthesis, and standard recrystallization techniques post-synthesis frequently fail to elevate purity beyond the 99.2% threshold, limiting the material's applicability in high-end pharmaceutical formulations.
The Novel Approach
The novel approach presented in the patent circumvents these thermodynamic limitations by employing a supramolecular strategy. Instead of trying to physically force separation based on boiling points or solubility differences alone, the method introduces a functionalized calixarene (MHZ002) that acts as a molecular sieve. This macrocyclic host is designed with specific binding pockets that preferentially accommodate the impurity molecule over the target N-nonanoic acid vanillimide. Upon mixing, the calixarene forms a stable host-guest complex with the impurity, effectively pulling it out of the solution phase. This allows the desired product to remain in the supernatant or be easily separated, resulting in a refined product with exceptional purity. The visual representation of this complex molecular architecture and its application is detailed below.
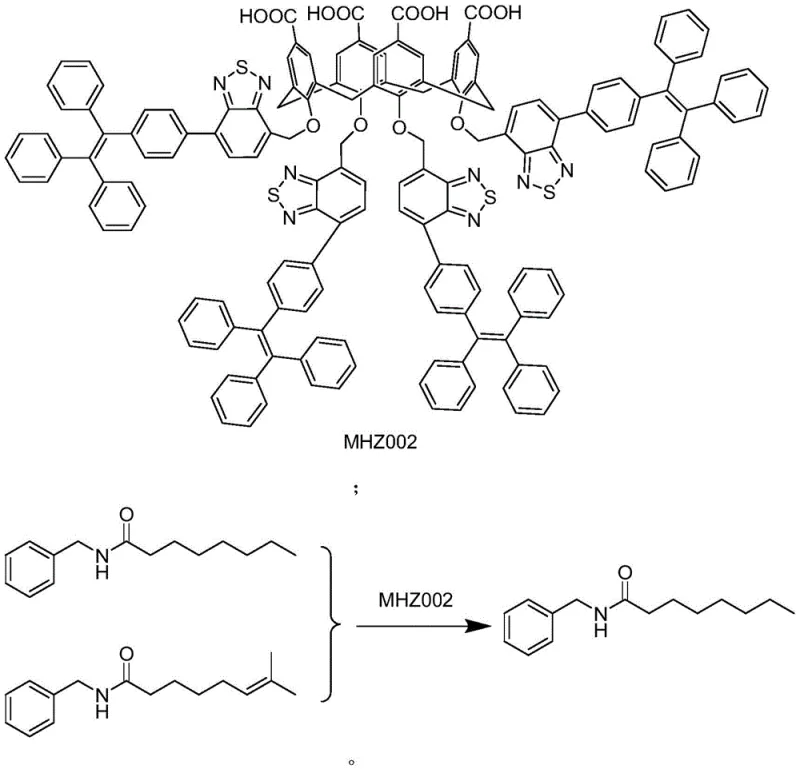
Mechanistic Insights into Supramolecular Host-Guest Recognition
The core of this technology lies in the precise design of the calixarene scaffold. The synthesis begins with the formation of a calixarene backbone capable of specific molecular identification. This involves a multi-step sequence including aldol condensation and Friedel-Crafts reactions to establish the macrocyclic framework, followed by etherification and sulfonation to tune the electronic and steric environment of the cavity. The resulting molecule, MHZ002, possesses a rigid yet adaptable structure that maximizes van der Waals forces and pi-pi interactions with the specific geometry of the (trans) 8-methyl-N-vanillyl-6-nonenamide impurity. This specificity is what drives the high selectivity of the process, ensuring that the target saturated amide is not co-precipitated.
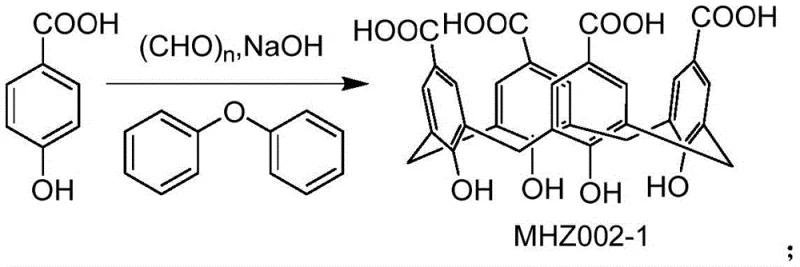
The synthesis of the key calixarene intermediate, MHZ002-1, is achieved through the condensation of p-hydroxybenzoic acid with formaldehyde under basic conditions, followed by thermal rearrangement in diphenyl ether. This creates the foundational cup-shaped structure essential for host-guest chemistry. Subsequent functionalization with benzothiadiazole derivatives via nucleophilic substitution further enhances the binding affinity and solubility profile of the host. The reaction schemes below illustrate the construction of this sophisticated molecular tool, highlighting the chemical transformations required to generate the active refining agent.
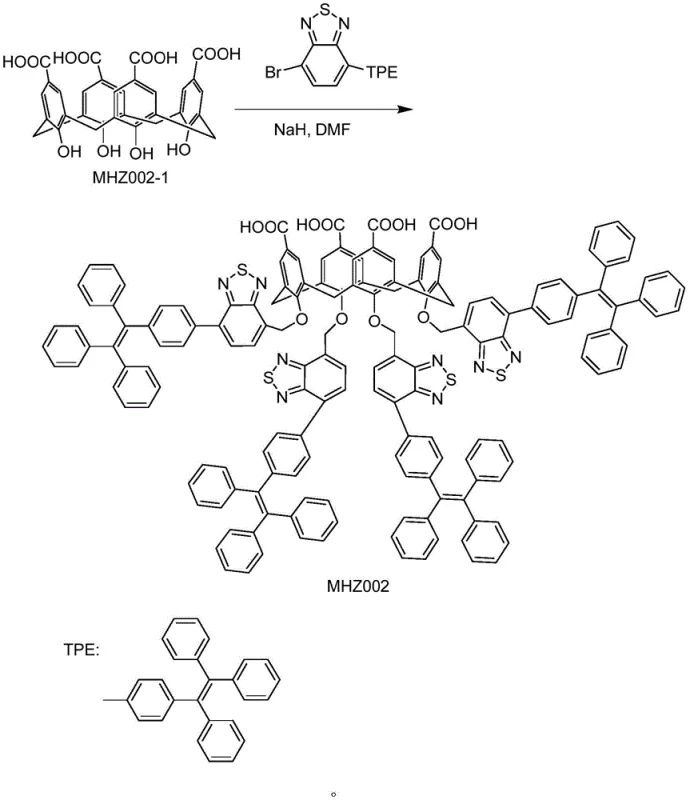
From an impurity control perspective, this mechanism offers a distinct advantage over chromatographic methods which are often too costly for bulk manufacturing. The supramolecular complexation acts as a highly specific scavenger. Once the complex forms, indicated visually by a color change from green to red in the reaction mixture, the separation becomes a simple matter of filtration or phase separation. The robustness of this interaction ensures that even trace amounts of the unsaturated impurity are captured, driving the equilibrium towards the formation of the pure product. This level of mechanistic control allows for consistent batch-to-batch reproducibility, a critical factor for regulatory compliance in pharmaceutical supply chains.
How to Synthesize High-Purity Synthetic Capsaicin Efficiently
The implementation of this refining protocol requires careful attention to stoichiometry and solvent management to maximize the efficiency of the calixarene host. The process is designed to be operationally simple, relying on standard unit operations found in most fine chemical facilities. By adhering to the specific molar ratios and temperature profiles outlined in the patent, manufacturers can achieve the reported purity benchmarks while minimizing waste. The following guide outlines the critical operational parameters for executing this synthesis effectively.
- Synthesize the specific calixarene host molecule (MHZ002) through aldol condensation and etherification reactions to create a cavity capable of recognizing specific impurity structures.
- Mix the crude synthetic capsaicin with the calixarene host in dichloromethane at room temperature, allowing the host-guest complexation to occur selectively with the impurity.
- Separate the formed precipitate or complex via filtration, followed by solvent evaporation and crystallization to recover the refined product with purity exceeding 99.8%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this supramolecular refining technology translates directly into tangible operational benefits. The shift from energy-intensive distillation to a mild, solution-based complexation process significantly reduces the utility load on the manufacturing plant. Furthermore, the ability to recycle the expensive calixarene ligand mitigates the raw material cost impact, making the process economically viable for large-scale production. This method addresses the chronic issue of supply inconsistency caused by difficult purification steps, ensuring a steady flow of high-quality material to downstream customers.
- Cost Reduction in Manufacturing: The elimination of high-energy rectification columns and the reduction in solvent usage during the refining stage lead to substantial cost savings. By operating at near-ambient temperatures and utilizing a recyclable host molecule, the process drastically lowers the variable cost per kilogram of the final product. The removal of the need for extensive chromatographic purification further simplifies the downstream processing train, reducing both capital expenditure on equipment and operational expenditure on consumables.
- Enhanced Supply Chain Reliability: The robustness of the supramolecular separation method ensures high yields and consistent purity, reducing the risk of batch failures that can disrupt supply schedules. The ability to effectively remove difficult isomers means that a wider range of crude feedstocks can be utilized without compromising final quality, thereby diversifying the supply base and reducing dependency on ultra-high purity starting materials which may have long lead times.
- Scalability and Environmental Compliance: This green chemistry approach aligns with increasingly stringent environmental regulations. The process generates less waste compared to traditional methods, and the recycling of the calixarene host minimizes the discharge of organic pollutants. The simplicity of the unit operations—mixing, filtration, and evaporation—facilitates easy scale-up from pilot to commercial production volumes, ensuring that supply can be ramped up quickly to meet market demand without complex engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this refining technology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of the supramolecular approach for potential partners and stakeholders evaluating this production route.
Q: What is the primary advantage of using calixarene for capsaicin refining?
A: The primary advantage is the high selectivity of the supramolecular host-guest interaction. Unlike traditional recrystallization which struggles to separate structurally similar isomers like (trans) 8-methyl-N-vanillyl-6-nonenamide, the functionalized calixarene specifically recognizes and binds the impurity, allowing for the isolation of the target molecule with purity levels exceeding 99.8%.
Q: Is the calixarene ligand reusable in this process?
A: Yes, the process is designed for sustainability and cost-efficiency. The patent explicitly details a recovery mechanism where the calixarene-impurity complex can be treated with methanol to release the impurity and precipitate the calixarene, which is then filtered and recovered for reuse in subsequent batches, significantly reducing material costs.
Q: How does this method compare to traditional rectification?
A: Traditional rectification of fatty acid precursors is energy-intensive and often inefficient for removing specific isomeric impurities post-synthesis. This supramolecular method operates at mild temperatures (room temperature to 30°C) and atmospheric or reduced pressure, offering a greener alternative with lower energy consumption and higher atom economy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Synthetic Capsaicin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires more than just a patent; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that advanced technologies like the supramolecular refining of capsaicin are implemented with precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required by the global pharmaceutical and fine chemical markets.
We invite you to collaborate with us to leverage this cutting-edge refining technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value and reliability in your sourcing of high-purity synthetic capsaicin.
