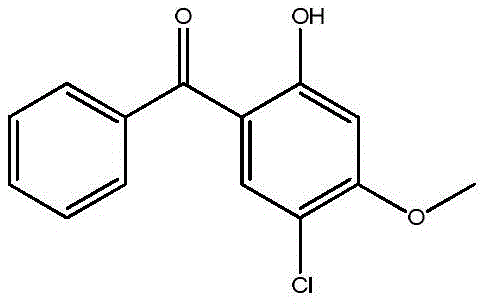
Industrial Recovery and Purification of 5-Chloro-2-Hydroxy-4-Methoxybenzophenone for High-Value Applications
Introduction to Advanced Waste Valorization in Fine Chemicals
The efficient utilization of industrial byproducts represents a critical frontier in modern fine chemical manufacturing, particularly for high-value pharmaceutical intermediates. Patent CN109096075B introduces a groundbreaking separation and purification method specifically designed for 5-chloro-2-hydroxy-4-methoxybenzophenone, a compound traditionally discarded as distillation waste residue. This innovative approach transforms what was once considered hazardous waste into a premium-grade chemical asset, addressing both environmental compliance and raw material scarcity. By leveraging a sophisticated acid reduction and multi-stage crystallization protocol, this technology enables the recovery of target components from the refining mother liquor of 2-hydroxy-4-methoxybenzophenone-5-sulfonic acid production. The structural integrity and chemical identity of this valuable benzophenone derivative are confirmed through rigorous spectral analysis, establishing its viability for downstream applications.
For global procurement teams seeking a reliable pharmaceutical intermediate supplier, this technology offers a unique value proposition by securing supply chains through waste stream valorization. The ability to extract high-purity materials from existing industrial processes significantly reduces the dependency on virgin raw material synthesis, thereby lowering the overall carbon footprint and production costs. This report delves deep into the technical nuances of this recovery method, providing R&D directors and supply chain heads with the actionable intelligence needed to evaluate its integration into their manufacturing portfolios. The transition from treating distillation concentrates as waste to viewing them as a resource marks a paradigm shift in sustainable chemical engineering.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 2-hydroxy-4-methoxybenzophenone-5-sulfonic acid has generated a complex distillation concentrated solution that posed significant disposal challenges for chemical manufacturers. This oily mixture, characterized by a dark color and intricate composition, typically contains residual solvents, unreacted starting materials, and various sulfonated byproducts that are difficult to separate using traditional refining techniques. In conventional workflows, this distillation residue is often classified as hazardous waste, requiring expensive treatment protocols or incineration, which adds substantial operational overhead without generating any revenue. Furthermore, the presence of 5-chloro-2-hydroxy-4-methoxybenzophenone within this matrix went largely unrecognized or unexploited due to the lack of effective separation technologies capable of isolating it in high purity. The inability to fractionate these components meant that valuable chemical potential was literally going down the drain, representing a significant loss of economic value and resource efficiency in the fine chemical sector.
The Novel Approach
The novel methodology outlined in the patent data revolutionizes this landscape by introducing a targeted chemical reduction and fractional crystallization strategy that effectively unlocks the value trapped within the waste stream. Instead of discarding the distillation concentrate, the process subjects it to a controlled reduction reaction using sulfuric acid at elevated temperatures between 80°C and 130°C, which cleaves the sulfonic acid groups and liberates the target benzophenone derivatives. Following this chemical transformation, a precise liquid-liquid extraction separates the organic phase, which is then subjected to a specialized cooling crystallization using a binary solvent system of halogenated hydrocarbons and esters. This dual-solvent approach is critical as it selectively precipitates impurities like 2-hydroxy-4-methoxybenzophenone while keeping the target chloro-derivative in solution or vice versa depending on the specific solubility profiles, ultimately allowing for the isolation of a crude product that is further purified via alcohol recrystallization. This comprehensive workflow ensures that the final product achieves an HPLC purity exceeding 99.5%, making it suitable for sensitive pharmaceutical applications.
Mechanistic Insights into Side-Reaction Recovery and Purification
Understanding the genesis of 5-chloro-2-hydroxy-4-methoxybenzophenone is essential for optimizing its recovery, as it originates from a specific side reaction during the primary sulfonation process. The formation of this chlorinated species is attributed to the presence of residual sulfonyl chloride impurities within the industrial grade chlorosulfonic acid used as a reagent. When 2-hydroxy-4-methoxybenzophenone is exposed to these conditions, an electrophilic aromatic substitution occurs, introducing a chlorine atom at the 5-position of the benzophenone ring. This mechanistic pathway is visually represented in the reaction scheme below, highlighting how process impurities in the reagents can lead to the accumulation of valuable byproducts in the waste stream.
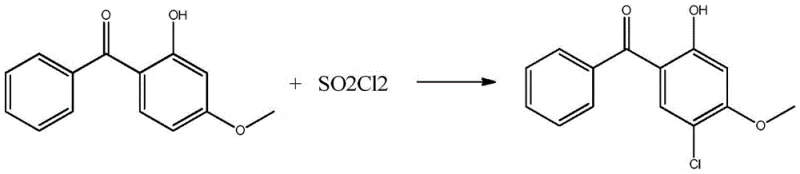
The recovery mechanism relies on the differential stability of sulfonic acid groups under acidic hydrolysis conditions. By heating the distillation concentrate with dilute sulfuric acid, the sulfonic acid moieties attached to the benzophenone core are hydrolyzed, regenerating the parent phenolic structures. This step is crucial because it converts the highly polar, water-soluble sulfonated waste back into lipophilic organic compounds that can be extracted into an organic phase. Subsequent purification exploits the subtle differences in solubility and crystallization kinetics between the chloro-substituted target and the non-chlorinated analog. The use of a mixed solvent system allows for fine-tuning the saturation point, ensuring that the target molecule crystallizes out with minimal inclusion of isomeric impurities or starting materials. This level of control over the solid-state chemistry is what enables the production of high-purity fine chemical intermediates from what was previously considered refuse.
How to Synthesize 5-Chloro-2-Hydroxy-4-Methoxybenzophenone Efficiently
The synthesis and purification protocol described herein offers a robust pathway for converting industrial waste into a high-value commodity, requiring precise control over reaction parameters and solvent compositions. The process begins with the treatment of the refining mother liquor concentrate, where temperature management during the acid reduction phase is critical to ensure complete desulfonation without degrading the sensitive benzophenone backbone. Following phase separation, the choice of extraction and crystallization solvents plays a pivotal role in the yield and purity profile, necessitating the use of specific halogenated hydrocarbons paired with esters to maximize selectivity. For R&D teams looking to implement this technology, the detailed standardized operating procedures regarding solvent ratios, cooling rates, and recrystallization conditions are paramount to success.
- Perform acid reduction on the distillation concentrated solution of 2-hydroxy-4-methoxybenzophenone-5-sulfonic acid using sulfuric acid at 80-130°C.
- Separate the organic phase and utilize a halogenated hydrocarbon-ester mixed solvent system for cooling crystallization to isolate the crude product.
- Recrystallize the crude material in an alcohol solvent such as ethanol or methanol to achieve final purity exceeding 99.5%.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this recovery technology presents a compelling opportunity for cost reduction in pharmaceutical intermediates manufacturing by fundamentally altering the raw material cost structure. By sourcing the target compound from internal waste streams rather than purchasing virgin synthetic materials, manufacturers can drastically reduce their exposure to volatile raw material markets and supply chain disruptions. The elimination of waste disposal costs further enhances the economic model, turning a liability into a profit center. This circular economy approach not only improves the bottom line but also aligns with increasingly stringent environmental regulations regarding industrial effluent and solid waste management, providing a dual advantage of financial savings and regulatory compliance.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the valorization of waste, which effectively lowers the net cost of goods sold by offsetting raw material expenses. Since the feedstock is derived from an existing process stream that would otherwise incur disposal fees, the marginal cost of producing the recovered 5-chloro-2-hydroxy-4-methoxybenzophenone is significantly lower than traditional synthesis routes. Furthermore, the recovery of solvents such as dichloromethane and ethyl acetate through distillation allows for their reuse in subsequent batches, minimizing consumable costs and reducing the overall chemical intensity of the operation. This efficiency translates into substantial long-term savings for large-scale production facilities.
- Enhanced Supply Chain Reliability: Integrating this recovery process diversifies the supply base for critical intermediates, reducing reliance on external vendors who may face capacity constraints or logistical delays. Because the feedstock is generated in-house as a byproduct of sulfonic acid production, the availability of the raw material is intrinsically linked to the plant's own production schedule, offering a high degree of predictability and control. This self-sufficiency mitigates the risk of supply shortages that often plague the global market for specialty chemicals, ensuring continuous operation for downstream synthesis lines. Additionally, the simplified logistics of handling internal waste streams versus importing hazardous reagents streamline the overall supply chain workflow.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard unit operations such as reflux reactors, phase separators, and crystallizers that are easily integrated into existing infrastructure. The ability to scale from pilot batches to multi-ton production without complex catalyst systems or exotic reagents facilitates rapid commercialization. Moreover, by reducing the volume of hazardous waste requiring off-site treatment, the facility significantly lowers its environmental footprint and associated compliance costs. The recovery of acid and solvents further demonstrates a commitment to green chemistry principles, enhancing the corporate sustainability profile which is increasingly important for stakeholders and customers alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this separation technology, based on the specific data points and experimental results provided in the patent literature. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for their own manufacturing lines. The answers reflect the proven capabilities of the method to handle complex mixtures and deliver consistent quality.
Q: What is the primary source of 5-chloro-2-hydroxy-4-methoxybenzophenone in this process?
A: It is generated as a side reaction byproduct during the sulfonation of 2-hydroxy-4-methoxybenzophenone, caused by residual sulfonyl chloride in the chlorosulfonic acid reagent, and is enriched in the refining mother liquor.
Q: How is the purity of the recovered product ensured?
A: The process utilizes a specific mixed solvent crystallization followed by alcohol recrystallization, which effectively removes unreacted starting materials and sulfonic acid derivatives, achieving HPLC purity over 99.5%.
Q: Can the solvents used in this recovery process be recycled?
A: Yes, the process design includes distillation steps to recover both the aqueous acid phase and the organic mixed solvents (halogenated hydrocarbons and esters) for repeated use, enhancing economic efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Chloro-2-Hydroxy-4-Methoxybenzophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of advanced recovery technologies like the one described in CN109096075B to redefine the economics of fine chemical production. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of 5-chloro-2-hydroxy-4-methoxybenzophenone meets the exacting standards required by the global pharmaceutical industry. We are committed to delivering high-purity intermediates that empower our clients to accelerate their drug development pipelines with confidence.
We invite forward-thinking organizations to collaborate with us to leverage these cost-effective and sustainable manufacturing solutions. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in waste valorization and purification can drive value for your supply chain.
