Advanced DMP Oxidation Strategy for High-Purity Ocotillol-Type Saponin Intermediates Manufacturing
The pharmaceutical industry's relentless pursuit of potent anti-tumor agents has placed Ocotillol-type saponins at the forefront of oncology research, particularly for their ability to reverse multidrug resistance via P-glycoprotein inhibition. A pivotal advancement in this domain is documented in Chinese Patent CN111704645A, which discloses a highly efficient application of Dess-Martin Periodinane (DMP) for synthesizing key intermediates of these bioactive molecules. This technological breakthrough addresses critical bottlenecks in the functionalization of the dammarane skeleton, specifically targeting the selective oxidation of hydroxyl groups at the C-3 and C-12 positions. By replacing traditional chromium-based oxidants with hypervalent iodine reagents, this methodology not only enhances chemical selectivity but also drastically streamlines the downstream purification processes required for high-purity pharmaceutical intermediates. For R&D teams and supply chain strategists, understanding this shift is essential for optimizing the production of next-generation anticancer therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ocotillol-type saponin derivatives relied heavily on Pyridinium Chlorochromate (PCC) for the oxidation of the C-3 hydroxyl group. However, this legacy approach is fraught with significant operational and chemical deficiencies that hinder scalable API manufacturing. The primary drawback lies in the physical nature of the reaction byproducts; PCC reactions frequently generate viscous, tar-like residues that adhere tenaciously to the product, making separation exceptionally difficult. To isolate the desired ketone, manufacturers are forced to employ labor-intensive filtration techniques using diatomaceous earth, followed by multiple solvent extractions, which inevitably leads to substantial product loss. Furthermore, the chemical selectivity of PCC is often inadequate, frequently resulting in the over-oxidation of the C-12 position or incomplete conversion, capping the maximum yield at approximately 85% and generating complex impurity profiles that complicate regulatory compliance.
The Novel Approach
In stark contrast, the methodology outlined in Patent CN111704645A utilizes Dess-Martin Periodinane (DMP) to achieve a level of precision and operational ease previously unattainable with chromium reagents. This novel approach leverages the unique solubility properties of DMP byproducts, which remain dissolved in organic solvents rather than forming insoluble sludges. This fundamental difference eliminates the need for cumbersome filtration through filter aids, allowing for a simple aqueous workup involving sodium bicarbonate and sodium sulfite washes. The result is a dramatic improvement in isolation efficiency and overall yield. Moreover, the reaction conditions are remarkably mild, typically proceeding at temperatures between -5°C and room temperature, which preserves the integrity of other sensitive functional groups within the complex saponin structure. This transition represents a paradigm shift towards greener, more efficient cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into DMP-Catalyzed Selective Oxidation
The superior performance of the DMP oxidation system is rooted in its distinct mechanistic pathway, which involves a ligand exchange process that is both kinetically favorable and thermodynamically driven. The reaction initiates with the nucleophilic attack of the substrate's C-3 hydroxyl group on the hypervalent iodine center of the DMP molecule, displacing one of the acetate ligands to form an alkoxy-periodinane intermediate. Subsequently, a base-mediated deprotonation facilitates the elimination of the reduced iodine species, resulting in the formation of the carbonyl group at the C-3 position. Crucially, this mechanism avoids the radical pathways often associated with metal-based oxidants, thereby minimizing side reactions and preserving the stereochemical integrity of the chiral centers at C-20 and C-24. The absence of transition metals also means there is no risk of heavy metal contamination, a critical quality attribute for any reliable pharmaceutical intermediate supplier to guarantee.
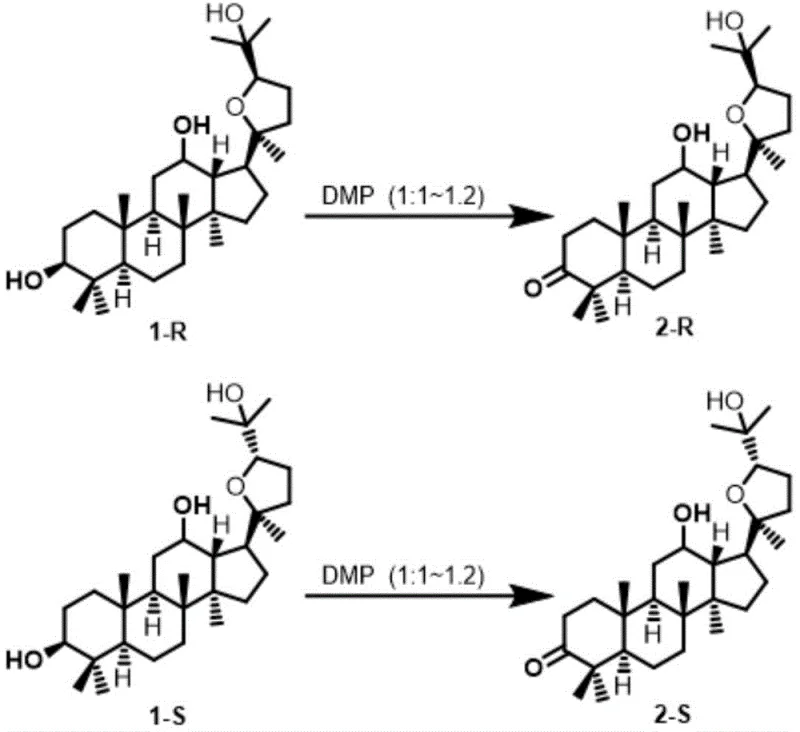
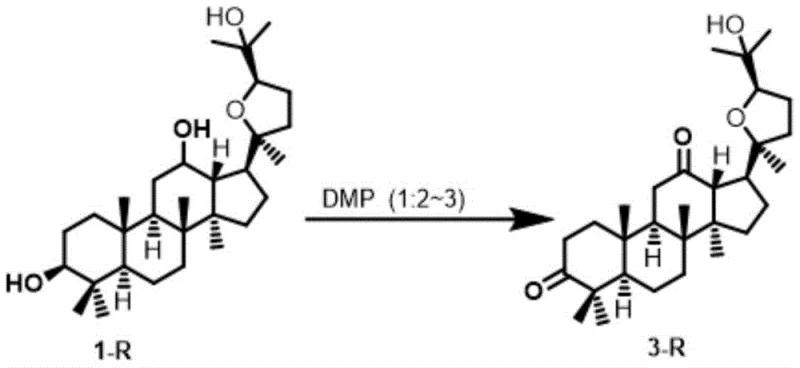
Furthermore, the patent elucidates a sophisticated strategy for controlling the degree of oxidation through precise stoichiometric modulation. By adjusting the molar ratio of DMP to the substrate, chemists can dictate whether the reaction stops at the mono-ketone stage or proceeds to the di-ketone. When the molar ratio is maintained between 1:1 and 1:1.2, the reaction exhibits high chemoselectivity for the C-3 position, yielding the mono-oxidized product with efficiencies exceeding 94%. Conversely, increasing the DMP loading to a ratio of 1:2 to 1:3 drives the oxidation of the secondary hydroxyl at C-12, producing the 3,12-dione derivative with yields surpassing 90%. This tunability allows for the flexible production of diverse building blocks from a single starting material, enhancing the versatility of the synthetic route for complex pharmaceutical intermediates.
How to Synthesize Ocotillol Saponin Ketones Efficiently
Implementing this advanced oxidation protocol requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and maximum yield. The process begins with the dissolution of the dammarane triol substrate and a mild base, such as sodium bicarbonate, in an anhydrous dichloromethane solvent system. The addition of tert-butanol serves as a co-solvent to enhance the solubility of the DMP reagent, ensuring a homogeneous reaction mixture. Temperature control is paramount; the reaction is initiated at sub-zero temperatures (-5°C to 0°C) to manage the exotherm and prevent localized overheating, before being allowed to warm to ambient conditions for completion. Detailed standard operating procedures for scaling this reaction from gram to kilogram quantities are provided below to assist process engineers in technology transfer.
- Dissolve the dammarane triol substrate and sodium bicarbonate in dichloromethane, then cool the mixture to between -5°C and 0°C using an ice bath.
- Add Dess-Martin Periodinane (DMP) and tert-butanol to the reaction mixture, maintaining the molar ratio at 1: 1.2 for mono-oxidation or 1:2.5 for di-oxidation.
- Allow the reaction to warm to room temperature over 4 to 5 hours, then quench with saturated sodium bicarbonate and sodium sulfite solution before extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this DMP-based oxidation route offers compelling strategic advantages that extend beyond mere chemical yield. The elimination of chromium-based reagents removes the significant environmental and disposal costs associated with heavy metal waste treatment, aligning production with increasingly stringent global environmental regulations. Additionally, the simplified workup procedure, which replaces complex filtration and adsorption steps with straightforward liquid-liquid extraction, significantly reduces the cycle time per batch. This operational efficiency translates directly into increased throughput capacity without the need for additional capital investment in specialized filtration equipment. The robustness of the reaction also ensures consistent supply continuity, mitigating the risks of batch failures that often plague older, less selective synthetic methods.
- Cost Reduction in Manufacturing: The transition to DMP oxidation eliminates the need for expensive and hazardous chromium reagents, as well as the consumables required for their removal, such as large quantities of diatomaceous earth. By avoiding the formation of viscous byproducts, the process minimizes product entrapment and loss during purification, leading to a substantial increase in the effective mass balance of the final product. Although the unit cost of DMP may be higher than PCC, the overall process economics are improved through higher yields and reduced labor hours for post-processing, resulting in significant overall cost optimization for the production of high-value saponin derivatives.
- Enhanced Supply Chain Reliability: The reagents utilized in this protocol, including DMP, sodium bicarbonate, and dichloromethane, are commodity chemicals with stable and widespread global availability. This reduces the dependency on niche suppliers and mitigates the risk of raw material shortages that can disrupt production schedules. Furthermore, the mild reaction conditions reduce the stress on reactor vessels and utility systems, lowering maintenance requirements and extending the lifespan of manufacturing assets. This reliability ensures that commercial scale-up of complex pharmaceutical intermediates can proceed without unexpected interruptions, securing the supply chain for downstream drug development.
- Scalability and Environmental Compliance: The homogeneous nature of the DMP reaction mixture makes it inherently easier to scale from laboratory to industrial production compared to heterogeneous slurries formed by PCC. The absence of solid chromium waste simplifies effluent treatment and reduces the facility's environmental footprint, facilitating easier permitting and compliance with green chemistry initiatives. The high selectivity of the reaction also reduces the burden on downstream chromatography or crystallization steps, allowing for larger batch sizes and more efficient use of purification columns. This scalability is crucial for meeting the growing demand for Ocotillol-type derivatives in clinical and commercial markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this DMP oxidation technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on the practical benefits of this method over traditional approaches. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this route for their specific production needs.
Q: How does the DMP oxidation method improve upon traditional PCC oxidation for saponin derivatives?
A: Unlike PCC, which forms viscous byproducts requiring difficult diatomaceous earth filtration, DMP oxidation produces soluble byproducts that are easily removed via aqueous washing, significantly simplifying post-processing and improving yield.
Q: Can the selectivity between mono-oxidation and di-oxidation be controlled in this process?
A: Yes, selectivity is precisely controlled by the stoichiometric ratio of the oxidant. A molar ratio of 1:1 to 1:1.2 favors the formation of the C-3 mono-ketone, while a ratio of 1:2 to 1:3 drives the reaction toward the C-3,12-di-ketone.
Q: What are the typical yields achieved with this novel oxidation protocol?
A: The patent data indicates that mono-oxidation products can be achieved with yields as high as 96%, while di-oxidation products consistently exceed 90% yield under optimized conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ocotillol Saponin Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the development of life-saving oncology therapies. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Ocotillol saponin intermediates meets the highest international standards. We are committed to leveraging advanced technologies, such as the DMP oxidation method described in CN111704645A, to deliver superior quality and consistency for our global partners.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis strategy can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this high-efficiency oxidation protocol. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and operational reliability.
