Advanced Synthesis of 2-(4-Chlorophenyl)aniline: Technical Breakthroughs for Commercial Agrochemical Manufacturing
Introduction to Patent CN109970571B and Technical Breakthroughs
The global demand for high-efficiency fungicides continues to drive innovation in the synthesis of critical intermediates, specifically 2-(4-chlorophenyl)aniline, which serves as the foundational building block for the widely used nicotinamide fungicide, Boscalid. Patent CN109970571B, published in late 2021, introduces a transformative synthetic methodology that departs from conventional reliance on costly halogenated anilines or energy-intensive hydrogenation processes. Instead, this novel approach leverages 9-fluorenone as a readily available and economically superior starting material, subjecting it to a sophisticated sequence of ring opening, chlorination, amidation, and rearrangement degradation. This strategic shift in raw material selection not only addresses the volatility of supply chains associated with iodine-based reagents but also fundamentally alters the cost structure of manufacturing this vital agrochemical intermediate. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential for optimizing supply chains and achieving significant cost reduction in agrochemical manufacturing.
The significance of this technology lies in its ability to bypass the traditional bottlenecks of nitro-group reduction or palladium-catalyzed coupling reactions, which often introduce heavy metal impurities and require stringent safety protocols for high-pressure operations. By utilizing a base-mediated ring opening of the fluorenone skeleton followed by a controlled Hofmann rearrangement, the process ensures a cleaner reaction profile with fewer by-products. This results in a final product with exceptional purity specifications, directly impacting the quality of the downstream active pharmaceutical or agrochemical ingredient. As we delve deeper into the mechanistic details and commercial implications, it becomes evident that this patent represents a robust solution for reliable agrochemical intermediate supplier networks seeking to enhance their competitive edge through process intensification and raw material optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-(4-chlorophenyl)aniline has been dominated by two primary routes, both of which suffer from significant economic and technical drawbacks that hinder efficient large-scale production. The first conventional method involves the condensation of o-chloronitrobenzene with p-chlorobenzoic acid to form a nitro-intermediate, which subsequently requires catalytic hydrogenation reduction to yield the target aniline. This hydrogenation step necessitates the use of expensive noble metal catalysts and high-pressure reactors, introducing substantial capital expenditure and safety risks related to hydrogen handling. Furthermore, the removal of trace metal catalysts to meet stringent purity standards adds complex purification steps, increasing both time and cost. The second traditional route utilizes o-iodoaniline and p-chlorobenzoic acid in a coupling reaction; however, the reliance on organoiodine compounds renders this method prohibitively expensive due to the high market price of iodine derivatives and the atom inefficiency of the reaction.
Beyond the direct costs of raw materials, these conventional pathways often struggle with selectivity issues and the formation of difficult-to-remove impurities, such as dehalogenated by-products or over-reduced species, which can compromise the efficacy of the final fungicide. The environmental footprint of these older methods is also considerable, given the generation of heavy metal waste streams and the energy intensity of high-pressure hydrogenation units. For supply chain heads, these factors translate into longer lead times, higher inventory costs for specialized catalysts, and increased vulnerability to fluctuations in the prices of precious metals and iodine. Consequently, there is a pressing industry need for a synthetic route that circumvents these limitations by utilizing more abundant feedstocks and milder reaction conditions, thereby enhancing the overall resilience and sustainability of the manufacturing process.
The Novel Approach
The innovative process disclosed in the patent data offers a compelling alternative by initiating the synthesis with 9-fluorenone, a stable and cost-effective polycyclic aromatic ketone that is abundantly available from coal tar processing or petroleum refining by-products. This new route elegantly constructs the biphenyl backbone through a base-catalyzed ring-opening reaction, effectively transforming the tricyclic fluorenone structure into the desired 2-phenylbenzoic acid scaffold without the need for carbon-carbon bond forming reactions that typically require expensive transition metal catalysts. Following this structural reorganization, the process employs a highly selective chlorination step using chlorine gas and a Lewis acid catalyst, which introduces the crucial chlorine atom at the para-position with high regioselectivity. This is followed by a straightforward conversion to the acid chloride and subsequent amidation, setting the stage for the final key transformation.
The culmination of this synthetic strategy is the Hofmann rearrangement degradation, a classic organic transformation that efficiently converts the benzamide intermediate into the target aniline with the loss of the carbonyl carbon. This step is particularly advantageous as it proceeds under relatively mild conditions using sodium hypochlorite, avoiding the harsh reducing environments of traditional methods. The cumulative effect of these four steps is a streamlined process that drastically simplifies the operational workflow, eliminating the need for high-pressure equipment and precious metal catalysts entirely. For procurement managers, this translates to a dramatic reduction in raw material costs and a decoupling from the volatile markets of iodine and palladium. Moreover, the simplicity of the unit operations—reflux, gas sparging, and distillation—facilitates easier commercial scale-up of complex agrochemical intermediates, ensuring a more stable and predictable supply of this critical fungicide precursor.
Mechanistic Insights into the 9-Fluorenone Ring Opening and Hofmann Rearrangement
To fully appreciate the technical robustness of this synthesis, one must examine the mechanistic underpinnings of the key transformations, starting with the base-mediated ring opening of 9-fluorenone. In the presence of a strong alkali such as potassium hydroxide and a high-boiling solvent like trimethylbenzene, the fluorenone molecule undergoes nucleophilic attack and subsequent skeletal rearrangement. The high temperature achieved during reflux in trimethylbenzene provides the necessary activation energy to break the central five-membered ring of the fluorenone, generating the 2-phenylbenzoic acid intermediate. This step is critical because it establishes the biphenyl connectivity required for the final product without needing cross-coupling chemistry. The choice of trimethylbenzene is strategic, as its high boiling point allows the reaction to proceed at temperatures sufficient to drive the equilibrium towards the open-ring carboxylic acid, while its non-polar nature aids in the subsequent separation of the organic product from the aqueous alkaline phase.
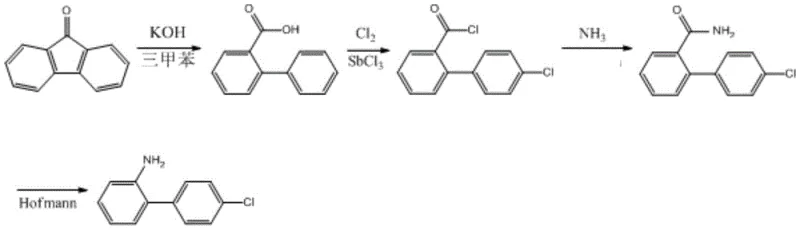
Following the formation of the carboxylic acid, the process moves to the chlorination and amidation sequence, which prepares the molecule for the final Hofmann degradation. The chlorination step utilizes antimony trichloride (SbCl3) as a Lewis acid catalyst to activate chlorine gas, facilitating electrophilic aromatic substitution specifically at the para-position of the pendant phenyl ring. This regioselectivity is vital for ensuring the correct isomer is produced for Boscalid synthesis. Once the chlorinated acid is converted to the corresponding acid chloride and then to the primary amide, the system is primed for the Hofmann rearrangement. In this final mechanistic phase, the treatment of the amide with sodium hypochlorite generates an N-chloroamide intermediate, which undergoes deprotonation and rearrangement to form an isocyanate. Hydrolysis of this isocyanate yields the final primary amine, 2-(4-chlorophenyl)aniline, with the expulsion of carbon dioxide. This mechanism ensures that the nitrogen atom is retained while the carbonyl group is excised, perfectly matching the structural requirements of the target molecule with high atom economy regarding the nitrogen source.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over nitro-reduction routes. Traditional hydrogenation can often lead to incomplete reduction (leaving nitro impurities) or over-reduction (affecting the chloro-substituent), creating a complex impurity profile that is difficult to purge. In contrast, the Hofmann rearrangement is generally clean, and the preceding chlorination step, being electrophilic, avoids the radical mechanisms that often cause poly-chlorination or dehalogenation side reactions when controlled properly. The patent data indicates that by strictly controlling the pH during the acidification step and maintaining specific temperatures during chlorination (60°C), the formation of side products is minimized. This results in a crude product that already possesses high purity, reducing the burden on downstream purification units such as recrystallization or column chromatography, which are often cost-prohibitive at the multi-ton scale. This level of inherent process control is what makes the technology attractive for producing high-purity fungicide intermediates.
How to Synthesize 2-(4-Chlorophenyl)aniline Efficiently
The practical implementation of this synthesis route requires careful attention to reaction parameters to maximize yield and safety, particularly during the gas introduction and rearrangement phases. The process begins with the reflux of 9-fluorenone in trimethylbenzene with potassium hydroxide, a step that demands robust heating capabilities and efficient stirring to ensure homogeneous contact between the solid base and the organic substrate. Following the ring opening, the workup involves a liquid-liquid extraction where pH control is paramount; acidifying the aqueous layer to a pH of 1-2 ensures complete protonation of the carboxylate salt, allowing for efficient extraction into dichloroethane. The subsequent chlorination step requires rigorous exclusion of moisture and air, achieved by nitrogen purging, to prevent the hydrolysis of the acid chloride intermediate and ensure the safety of handling chlorine gas. Finally, the Hofmann rearrangement must be conducted with precise stoichiometry of sodium hypochlorite to avoid over-oxidation while ensuring complete conversion of the amide.
- Perform base-catalyzed ring opening of 9-fluorenone in trimethylbenzene with KOH under reflux to obtain 2-phenylbenzoic acid.
- Conduct electrophilic chlorination using chlorine gas and SbCl3 catalyst at 60°C to form 2-(4-chlorophenyl)benzoyl chloride.
- Execute ammoniation followed by Hofmann rearrangement degradation using sodium hypochlorite to yield the final aniline derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this 9-fluorenone-based synthesis route presents a multitude of strategic advantages that directly impact the bottom line and operational reliability. The most immediate benefit is the drastic reduction in raw material costs, driven by the substitution of expensive o-iodoaniline or specialized nitro-compounds with 9-fluorenone, which is a commodity chemical with a stable and abundant global supply. By eliminating the dependency on iodine derivatives, manufacturers insulate themselves from the extreme price volatility often seen in the halogen market, leading to more predictable budgeting and cost structures. Furthermore, the removal of noble metal catalysts such as palladium or platinum from the process equation not only saves on the direct cost of these precious metals but also eliminates the complex and costly recovery processes associated with them, resulting in substantial cost savings in waste management and catalyst regeneration.
In terms of supply chain reliability, this process enhances continuity by utilizing standard chemical reagents and solvents that are readily available from multiple vendors, reducing the risk of single-source bottlenecks. The operational simplicity of the route, which relies on atmospheric pressure reactions and standard distillation rather than high-pressure hydrogenation, significantly lowers the barrier to entry for contract manufacturing organizations (CMOs) and reduces the lead time for high-purity fungicide intermediates. Facilities do not require specialized high-pressure autoclaves, meaning that existing general-purpose chemical reactors can be utilized, increasing the available capacity for production without the need for massive capital investment in new infrastructure. This flexibility allows for rapid scaling in response to market demand surges for Boscalid, ensuring that downstream customers receive their materials on time and without interruption.
Environmental compliance and scalability are also significantly improved, aligning with modern green chemistry principles and regulatory requirements. The avoidance of heavy metal catalysts simplifies the effluent treatment process, as there is no need for specialized resin beds or precipitation steps to remove trace metals to ppm levels before discharge. The use of sodium hypochlorite in the final step generates benign by-products compared to the metal-laden waste streams of hydrogenation. This cleaner profile facilitates easier permitting and reduces the environmental liability associated with manufacturing. Additionally, the high total yield reported in the patent data implies better atom economy and less waste generation per kilogram of product, further contributing to a sustainable manufacturing model. These factors combined make the process highly attractive for long-term commercial partnerships focused on sustainable and cost-effective agrochemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 2-(4-chlorophenyl)aniline, derived from the detailed analysis of the patent specifications and industry best practices. Understanding these aspects is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing supply chains. The answers provided reflect the specific advantages of the 9-fluorenone route, focusing on purity, yield, and operational safety, which are the primary concerns for technical decision-makers in the fine chemical sector.
Q: What are the primary advantages of the 9-fluorenone route over traditional methods?
A: The 9-fluorenone route eliminates the need for expensive o-iodoaniline or high-pressure catalytic hydrogenation equipment required in traditional nitro-reduction methods, significantly lowering capital and operational expenditures.
Q: What purity levels can be achieved with this synthesis process?
A: According to patent data, this specific four-step process consistently achieves a product content of greater than 98 percent, with a total yield exceeding 75 percent, ensuring high quality for downstream fungicide synthesis.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes standard unit operations such as reflux, gas introduction, and distillation, avoiding exotic conditions, which makes it highly adaptable for commercial scale-up of complex agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-Chlorophenyl)aniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global agrochemical supply chain, and we are uniquely positioned to leverage advanced synthetic technologies like the one described in CN109970571B. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We understand that consistency is key, which is why our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting impurities at trace levels. Whether you require custom synthesis of complex intermediates or reliable bulk supply of established fungicide precursors, our commitment to technical excellence and operational integrity makes us the ideal partner for your long-term growth.
We invite you to collaborate with us to explore how this optimized synthesis route can benefit your specific production needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your volume requirements, helping you identify opportunities to reduce overall manufacturing expenses without compromising on quality. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective, and high-quality supply of 2-(4-chlorophenyl)aniline, empowering your organization to lead in the competitive agrochemical market.
