Advanced Pd-Catalyzed Amination Technology for Scalable Production of Complex Agrochemical Intermediates
The pharmaceutical and agrochemical industries constantly face the challenge of synthesizing sterically hindered amines, which serve as critical building blocks for high-value active ingredients. Patent CN101687765B introduces a groundbreaking methodology for the preparation of compounds of formula (I), specifically targeting o-dicyclopropyl-substituted or o-C6-C7 alkyl-substituted primary anilines. These compounds are indispensable intermediates in the manufacturing of potent fungicides, yet their synthesis has historically been plagued by low yields and complex multi-step procedures. The core innovation lies in the utilization of a palladium-catalyzed cross-coupling reaction between a hindered halobenzene and urea, facilitated by a specialized ferrocenyl-diphosphine ligand system. This technological leap allows for the direct conversion of inexpensive chlorobenzene derivatives into valuable amines in a single step, bypassing the limitations of previous copper-catalyzed or two-step palladium methods. By enabling the use of robust chloro-substrates and avoiding costly protecting group strategies, this process represents a significant advancement for any reliable agrochemical intermediate supplier seeking to optimize their production lines.
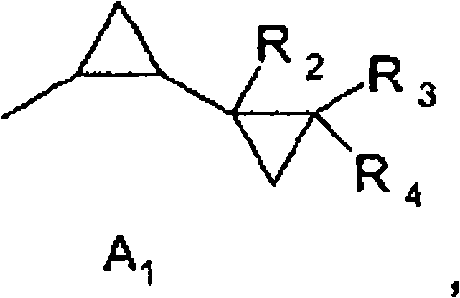
The synthesis of sterically hindered anilines has traditionally been a bottleneck in fine chemical manufacturing due to the severe steric congestion around the reaction site. Conventional methods, such as those described in prior art like WO 03/74491, often relied on a two-step sequence involving the reaction of halobenzenes with benzophenone imine followed by a hydrolysis step. This approach is not only operationally cumbersome but also economically inefficient due to the high cost of benzophenone imine and the additional processing time required for deprotection. Furthermore, alternative attempts using copper catalysts, as seen in WO 06/61226, have struggled with waste management issues and poor reactivity towards less reactive chlorobenzene substrates, often resulting in the formation of unwanted dimer by-products rather than the desired amine. The inability to effectively activate the carbon-chlorine bond in such crowded molecular environments has forced manufacturers to resort to more expensive bromo- or iodo-substituted starting materials, drastically inflating the cost of goods sold.
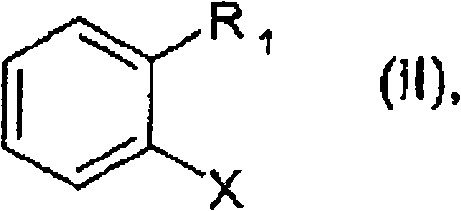
In stark contrast, the novel approach detailed in the patent utilizes a highly active palladium catalyst system comprising a ferrocenyl-diphosphine ligand to overcome these steric and electronic barriers. This method enables the direct reaction of formula (II) compounds, where X can be the economically favorable chlorine atom, with urea in the presence of a base. The use of urea as the nitrogen source is a strategic masterstroke, as it is inexpensive, stable, and avoids the formation of heavy metal waste associated with other amination reagents. The reaction proceeds efficiently at elevated temperatures, typically between 50°C and 180°C, yielding the target primary amine with high selectivity and minimal dimerization. This streamlined one-pot procedure significantly reduces the operational complexity and solvent consumption, making it an ideal candidate for cost reduction in fungicide manufacturing where margin pressure is intense.
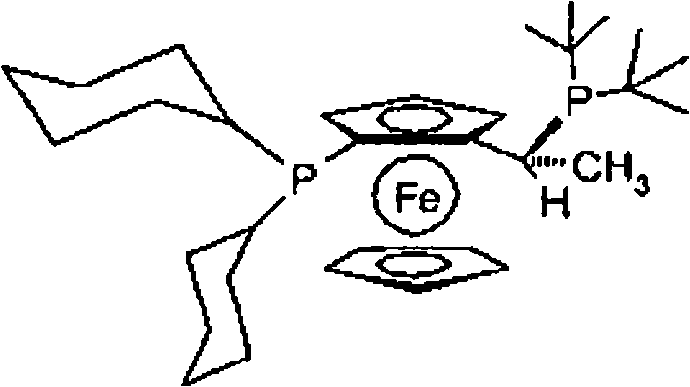
The success of this transformation hinges on the unique mechanistic properties of the ferrocenyl-diphosphine ligand coordinated to the palladium center. Unlike simple phosphine ligands, the ferrocenyl backbone provides a rigid yet tunable geometry that stabilizes the palladium species during the oxidative addition step, which is typically the rate-determining step for unactivated aryl chlorides. The bulky substituents on the phosphorus atoms, such as tert-butyl or cyclohexyl groups found in ligands like (R)-(-)-1-[(S)-2-(dicyclohexylphosphino)ferrocenyl]-ethyl-di-tert-butylphosphine, create a protective pocket that prevents catalyst deactivation while promoting the reductive elimination of the bulky amine product. This precise steric tuning allows the catalyst to insert into the strong C-Cl bond of the ortho-substituted benzene ring, a feat that conventional catalysts cannot achieve without excessive energy input or leading to decomposition. The result is a catalytic cycle that maintains high turnover numbers even in the presence of significant steric hindrance, ensuring consistent batch-to-batch quality.
Furthermore, the choice of base and solvent plays a pivotal role in managing the impurity profile of the final product. The patent specifies the use of strong alkoxides, particularly sodium tert-butoxide, in polar aprotic solvents like N-methylpyrrolidone (NMP) or diethylene glycol diethyl ether. These conditions facilitate the deprotonation of the urea intermediate and stabilize the anionic palladium species, thereby suppressing side reactions such as homocoupling or dehalogenation. By carefully controlling the concentration of the starting material, typically between 0.1M and 2M, the process maximizes space-time yield while minimizing the volume of solvent required for downstream processing. This level of control over the reaction parameters ensures that the impurity spectrum remains narrow, simplifying the purification process and delivering a high-purity agrochemical intermediate that meets stringent regulatory standards for residue limits.
How to Synthesize 2-Dicyclopropylaniline Efficiently
The synthesis of 2-dicyclopropylaniline serves as a prime example of the utility of this patented technology, demonstrating how complex steric environments can be navigated using modern catalytic methods. The process involves charging a reactor with 2-(2-chlorophenyl)dicyclopropyl, urea, and sodium tert-butoxide in a degassed solvent under an inert atmosphere. The addition of the palladium catalyst, generated in situ from palladium acetate and the specific ferrocenyl ligand, initiates the coupling reaction upon heating to approximately 134°C. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the sterically hindered halobenzene substrate, urea, and a strong base such as sodium tert-butoxide in an inert solvent like N-methylpyrrolidone.
- Add a catalytic amount of a palladium complex formed from a palladium precursor and a specialized ferrocenyl-diphosphine ligand to facilitate the cross-coupling reaction.
- Heat the mixture to elevated temperatures between 50°C and 180°C under an inert atmosphere to drive the amination to completion, followed by standard workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process offers tangible strategic benefits that extend beyond mere technical feasibility. The ability to utilize chlorobenzene derivatives instead of their bromo- or iodo-counterparts represents a fundamental shift in raw material sourcing, as chlorinated aromatics are generally more abundant and significantly less expensive on the global market. This substitution directly translates to substantial cost savings in the bill of materials, allowing companies to improve their gross margins without compromising on the quality of the final active ingredient. Moreover, the elimination of the second hydrolysis step required in older imine-based routes reduces the overall cycle time and equipment occupancy, effectively increasing the throughput capacity of existing manufacturing facilities without the need for capital expenditure on new reactors.
- Cost Reduction in Manufacturing: The transition to a one-step urea amination process eliminates the need for expensive benzophenone imine reagents and the associated acid hydrolysis step, which traditionally generated large volumes of salt waste. By streamlining the synthesis into a single catalytic event, the process drastically reduces solvent usage, energy consumption, and labor hours per kilogram of product. The use of catalytic amounts of palladium, which can potentially be recycled, further mitigates the impact of precious metal costs on the overall production budget, ensuring a more predictable and lower cost structure for long-term supply contracts.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like urea and chlorobenzene derivatives enhances supply chain resilience by reducing dependence on niche, high-cost specialty reagents that may be subject to availability fluctuations. The robustness of the catalyst system allows for the processing of technical grade starting materials with minor impurities, reducing the burden on upstream purification units. This flexibility ensures that production schedules can be maintained even when facing minor variations in raw material quality, thereby securing a continuous flow of critical intermediates to downstream formulation plants and preventing costly production stoppages.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, operating at concentrations that minimize solvent volume and maximize reactor efficiency, which is crucial for commercial scale-up of complex agrochemical intermediates. The reduction in waste generation, particularly the avoidance of heavy copper salts and stoichiometric imine by-products, aligns with increasingly stringent environmental regulations and corporate sustainability goals. This cleaner profile simplifies waste treatment protocols and lowers the environmental compliance costs associated with hazardous waste disposal, making the facility more sustainable and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced amination technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: Why is the ferrocenyl-diphosphine ligand critical for this amination process?
A: The ferrocenyl-diphosphine ligand creates a specific steric and electronic environment around the palladium center that enables the activation of sterically hindered and less reactive chlorobenzene substrates, which traditional catalysts fail to couple efficiently.
Q: What are the economic advantages of using urea over benzophenone imine in this synthesis?
A: Using urea eliminates the need for expensive benzophenone imine reagents and avoids a second hydrolysis step required in older methods, significantly simplifying the process flow and reducing raw material costs for large-scale production.
Q: Can this process effectively utilize chlorobenzene derivatives instead of bromobenzene?
A: Yes, the patented method is specifically designed to activate the stronger C-Cl bond in sterically hindered substrates, allowing manufacturers to use cheaper and more readily available chlorobenzene starting materials without sacrificing yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Dicyclopropylaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the competitive agrochemical landscape. Our team of expert chemists has extensively evaluated the technology described in CN101687765B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity intermediates that adhere to stringent purity specifications, utilizing our rigorous QC labs to ensure every batch meets the exacting standards required for global regulatory filings. Our state-of-the-art facilities are equipped to handle the specific solvent and temperature requirements of this palladium-catalyzed process, ensuring a seamless transition from laboratory optimization to full-scale manufacturing.
We invite you to collaborate with us to leverage this innovative technology for your supply chain needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your fungicide production programs.
