Scalable Synthesis of Aminomethyl Biphenyl Tetrazole Salts for Global API Production
Scalable Synthesis of Aminomethyl Biphenyl Tetrazole Salts for Global API Production
The pharmaceutical industry's relentless demand for Angiotensin II receptor antagonists, commonly known as Sartans, necessitates robust and efficient supply chains for their critical intermediates. Patent CN101397277A introduces a groundbreaking methodology for synthesizing aminomethyl biphenyl tetrazole and its corresponding salts, specifically targeting the core scaffold found in blockbuster drugs like Irbesartan, Valsartan, and Losartan. This technology addresses long-standing inefficiencies in intermediate production by replacing hazardous reagents and multi-step protection strategies with a streamlined, high-yield catalytic process. For R&D directors and procurement managers alike, understanding this shift is vital, as it promises not only higher purity standards reaching 99.5 percent but also a drastic simplification of the manufacturing workflow. The ability to produce these key building blocks without the burden of complex purification steps represents a significant leap forward in process chemistry, ensuring a more reliable pharmaceutical intermediate supplier landscape for the coming decade.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4'-aminomethyl-2-(1H-tetrazole-5-yl) biphenyl has been plagued by convoluted reaction pathways that hinder cost-effective production. As illustrated in prior art such as US5336677 and WO9308169, traditional routes often rely on a six-step process starting from 2'-cyano-4-methyl diphenyl. A major bottleneck in these legacy methods is the necessity of using a trityl group to protect the tetrazole moiety during subsequent reactions. This protection-deprotection sequence adds substantial material costs and extends the overall cycle time, creating unnecessary bottlenecks in the supply chain. Furthermore, these older methodologies frequently employ hydrazine hydrate, a reagent known for being flammable, highly toxic, and corrosive, which imposes severe safety regulations and expensive waste treatment requirements on the manufacturing facility. The cumulative effect of these factors is a process that is difficult to purify, yields lower overall productivity, and presents significant environmental compliance challenges.
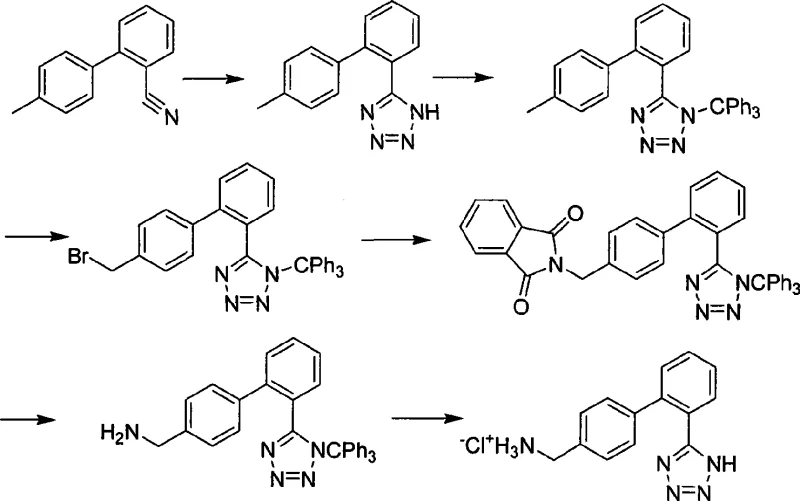
The Novel Approach
In stark contrast, the methodology disclosed in CN101397277A offers a direct and elegant solution by utilizing a specific azido-methyl biphenyl tetrazole intermediate. This novel approach bypasses the need for trityl protection entirely, allowing for a much shorter synthetic sequence that begins with readily available cyano-biphenyl precursors. By employing metal azides in the presence of Lewis acids, the tetrazole ring is constructed efficiently without the side reactions typical of hydrazine-based cyclizations. The subsequent reduction step is equally innovative, utilizing mild catalytic hydrogenation or chemical reduction agents like triphenylphosphine under gentle conditions. This shift eliminates the harsh environments previously required, resulting in a cleaner reaction profile where the product can be easily purified through standard crystallization techniques rather than expensive chromatography. This transition from complex protection chemistry to direct functional group transformation is the key to unlocking substantial cost savings in API manufacturing.
Mechanistic Insights into Lewis Acid-Catalyzed Tetrazole Formation and Reduction
The core innovation of this patent lies in the precise control of the tetrazole formation and the subsequent reduction of the azide group. The synthesis of the critical intermediate, general formula 3, involves reacting a 2-cyano-4'-substituted methyl biphenyl with a metal azide such as sodium azide or lithium azide. Crucially, this reaction is facilitated by a Lewis acid catalyst, which may include zinc chloride, lithium chloride, or tributyltin chloride, in a non-protonic solvent like dimethyl sulfoxide (DMSO) or dimethylformamide (DMF). The Lewis acid activates the nitrile group towards nucleophilic attack by the azide ion, promoting cyclization at temperatures between 80°C and 140°C. This mechanism ensures high conversion rates, often exceeding 95 percent, while minimizing the formation of polymeric byproducts that typically complicate downstream processing.

Following the formation of the tetrazole ring, the process moves to the reduction of the azido-methyl group to the desired aminomethyl functionality. This step is remarkably versatile, supporting both catalytic hydrogenation using palladium on carbon (Pd/C) and chemical reduction using agents like tin dichloride or triphenylphosphine. When using catalytic hydrogenation, the reaction proceeds under mild pressures of 15 to 150 psi and temperatures of 10 to 30°C, conditions that preserve the integrity of the sensitive tetrazole ring. The mechanistic pathway here involves the adsorption of the azide onto the metal catalyst surface, where it is sequentially reduced to the amine without cleaving the biphenyl bond. This selectivity is paramount for maintaining the structural fidelity of the molecule, ensuring that the final salt, whether hydrochloride or hydrobromide, achieves the stringent purity specifications of 99.5 percent required for pharmaceutical applications.
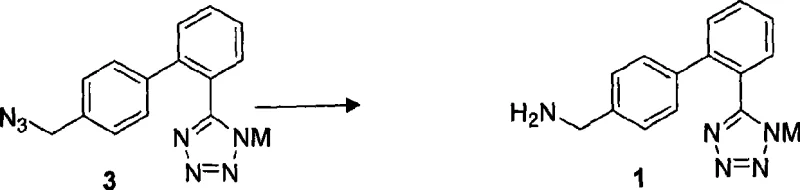
How to Synthesize 4'-Aminomethyl-2-(1H-tetrazole-5-yl) biphenyl Efficiently
Implementing this synthesis requires careful attention to solvent selection and catalyst loading to maximize yield and safety. The process begins with the preparation of the azido intermediate in a polar aprotic solvent, followed by a workup that involves pH adjustment to isolate the crude tetrazole. The subsequent reduction step is the critical control point, where the choice of reducing agent dictates the specific workup procedure; for instance, hydrogenation allows for simple filtration of the catalyst, while chemical reduction may require extraction steps to remove metal salts. Detailed standard operating procedures regarding stoichiometry, temperature ramps, and quenching protocols are essential for reproducibility. For the complete standardized synthesis steps and specific experimental parameters, please refer to the guide below.
- React 2-cyano-4'-substituted methyl biphenyl with metal azide (e.g., NaN3) in a non-protonic solvent like DMSO using a Lewis acid catalyst (e.g., LiCl or ZnCl2) at 80-140°C to form the azido-methyl tetrazole intermediate.
- Perform reduction of the azido intermediate using a metal catalyst (e.g., 10% Pd/C) under hydrogen pressure (15-150 psi) in an organic inert solvent like methanol or ethanol at 10-30°C.
- Acidify the resulting amine with hydrochloric acid or other acids to precipitate the final salt product with purity exceeding 99.5%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates directly into enhanced operational efficiency and risk mitigation. The elimination of the trityl protecting group removes a significant cost driver associated with expensive reagents and the additional reaction steps required for their installation and removal. Furthermore, the avoidance of hydrazine hydrate drastically reduces the regulatory burden and safety costs associated with handling hazardous materials, leading to a more sustainable and compliant manufacturing operation. The simplicity of the purification process, which relies on crystallization rather than chromatography, means that production throughput can be significantly increased without proportional increases in capital expenditure or solvent consumption.
- Cost Reduction in Manufacturing: The streamlined synthetic route eliminates multiple steps associated with protection and deprotection chemistry, directly lowering raw material consumption and labor costs. By utilizing common Lewis acids and avoiding exotic reagents, the bill of materials is optimized, and the removal of chromatographic purification steps significantly reduces solvent waste disposal costs. This logical deduction of process simplification leads to a more competitive pricing structure for the final intermediate without compromising quality.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as 2-cyano-4'-bromomethylbiphenyl and sodium azide ensures a stable supply base that is less susceptible to market volatility. The robustness of the reaction conditions, which tolerate a range of temperatures and pressures, allows for flexible manufacturing scheduling and reduces the risk of batch failures due to minor process deviations. This stability is crucial for maintaining continuous supply lines for downstream API production.
- Scalability and Environmental Compliance: The process is explicitly designed for large-scale industrial manufacturing, utilizing solvents and reagents that are manageable in standard reactor setups. The absence of highly toxic hydrazine and the reduction in heavy metal waste from simplified workups align with modern green chemistry principles. This facilitates easier permitting and environmental compliance, ensuring long-term viability of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide clarity on feasibility and performance. Understanding these details helps stakeholders make informed decisions about integrating this route into their existing manufacturing portfolios.
Q: How does this new method improve purity compared to traditional trityl-protection routes?
A: By eliminating the trityl protecting group and harsh hydrazine hydrate steps found in older patents like US5336677, this method avoids complex byproduct formation, allowing the final product purity to consistently reach 99.5% through simple crystallization.
Q: What are the safety advantages of avoiding hydrazine hydrate in tetrazole synthesis?
A: Traditional routes often utilize flammable and highly toxic hydrazine hydrate. This patented process utilizes metal azides and Lewis acids under controlled conditions, significantly reducing workplace hazards and simplifying waste treatment protocols.
Q: Is this process suitable for large-scale industrial manufacturing of Sartan intermediates?
A: Yes, the patent explicitly states suitability for large-scale industrial manufacturing due to mild reaction conditions (10-30°C for reduction), commercially available raw materials, and high total yield without requiring chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4'-Aminomethyl-2-(1H-tetrazole-5-yl) biphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in the global pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 4'-aminomethyl-2-(1H-tetrazole-5-yl) biphenyl performs consistently in your downstream synthesis. Our capability to adapt complex chemistries like the Lewis acid-catalyzed tetrazole formation allows us to offer a reliable pharmaceutical intermediate supplier partnership that prioritizes both quality and continuity.
We invite you to discuss how this advanced synthesis route can optimize your specific production needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term goals for cost reduction in API manufacturing.
