Advanced Copper-Catalyzed Ester Aminolysis for High-Purity L-Menthane Carboxamide Production
The global demand for high-performance cooling agents in the flavor, fragrance, and pharmaceutical sectors has driven intense research into next-generation menthol derivatives. Patent CN111393293B, published in September 2022, introduces a groundbreaking ester aminolysis reaction catalyst composition that fundamentally transforms the synthesis of L-menthane carboxamide, widely known in the industry as WS-3. Unlike traditional L-menthol, which suffers from volatility and a pungent odor, L-menthane carboxamide offers a cooling sensation 3-5 times stronger with no bitterness and prolonged action time. 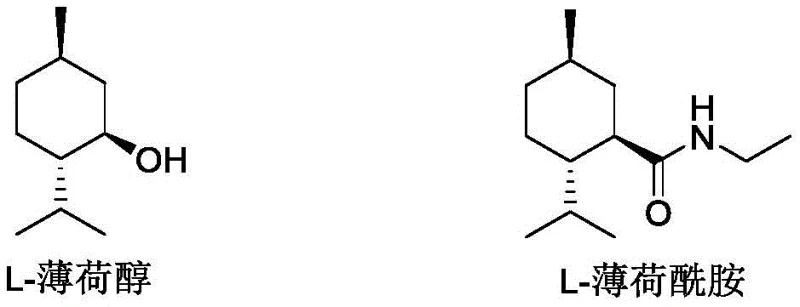 This patent discloses a sophisticated catalytic system utilizing monovalent copper salts complexed with chiral (R)-N,N,N,N-tetraalkyl binaphthylamine ligands. This innovation addresses the critical bottlenecks of previous manufacturing methods, specifically the issues of low yield, racemization, and environmental toxicity. For procurement and R&D leaders seeking a reliable cooling agent intermediate supplier, this technology represents a paradigm shift towards greener, more efficient, and higher-purity manufacturing protocols that align with modern sustainability goals.
This patent discloses a sophisticated catalytic system utilizing monovalent copper salts complexed with chiral (R)-N,N,N,N-tetraalkyl binaphthylamine ligands. This innovation addresses the critical bottlenecks of previous manufacturing methods, specifically the issues of low yield, racemization, and environmental toxicity. For procurement and R&D leaders seeking a reliable cooling agent intermediate supplier, this technology represents a paradigm shift towards greener, more efficient, and higher-purity manufacturing protocols that align with modern sustainability goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of L-menthane carboxamide has relied on routes that are chemically inefficient and operationally hazardous. The earliest methods, such as those reported by Wilkinson Sword, utilized a three-step sequence involving Grignard reaction, carboxylation with carbon dioxide, and subsequent acylation. As illustrated in the prior art reaction schemes, the carboxylation step is particularly problematic; the reaction of menthyl magnesium chloride with CO2 often results in significant racemization at the chiral center due to the harsh conditions required, limiting the overall yield to approximately 40%. 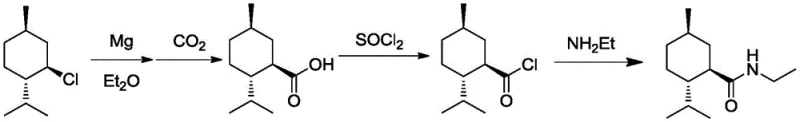 Furthermore, alternative pathways involving nitrile intermediates require the use of highly toxic sodium cyanide (NaCN), posing severe safety risks and complicating waste disposal. Another common approach involves ammonolysis under strong alkaline conditions, which inevitably leads to the epimerization of the alpha-carbonyl chiral center, degrading the optical purity of the final product and necessitating costly purification steps to remove unwanted isomers.
Furthermore, alternative pathways involving nitrile intermediates require the use of highly toxic sodium cyanide (NaCN), posing severe safety risks and complicating waste disposal. Another common approach involves ammonolysis under strong alkaline conditions, which inevitably leads to the epimerization of the alpha-carbonyl chiral center, degrading the optical purity of the final product and necessitating costly purification steps to remove unwanted isomers.
The Novel Approach
The methodology described in CN111393293B circumvents these historical deficiencies by employing a streamlined two-step strategy centered on a mild ester aminolysis. Instead of struggling with CO2 insertion or toxic nitriles, the process converts menthyl magnesium chloride directly into a menthyl formate ester using chloroformates. 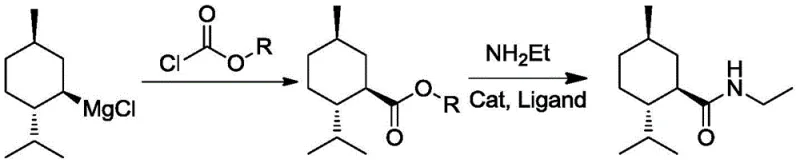 The true breakthrough lies in the subsequent conversion of this ester to the amide. By utilizing a specialized copper catalyst system, the reaction proceeds efficiently at ambient temperatures (20-30°C) without the need for strong bases. This not only preserves the stereochemical integrity of the molecule but also drastically simplifies the workup procedure. The elimination of aqueous acid-base washes means there is virtually no wastewater discharge, offering a substantial advantage in cost reduction in flavor & fragrance manufacturing by lowering effluent treatment expenses.
The true breakthrough lies in the subsequent conversion of this ester to the amide. By utilizing a specialized copper catalyst system, the reaction proceeds efficiently at ambient temperatures (20-30°C) without the need for strong bases. This not only preserves the stereochemical integrity of the molecule but also drastically simplifies the workup procedure. The elimination of aqueous acid-base washes means there is virtually no wastewater discharge, offering a substantial advantage in cost reduction in flavor & fragrance manufacturing by lowering effluent treatment expenses.
Mechanistic Insights into Copper-Catalyzed Ester Aminolysis
The core of this technological advancement is the unique catalyst composition, which comprises a monovalent copper salt (CuX, where X is Cl, Br, I, or AcO) and a bulky, chiral ligand. The ligand, specifically an (R)-N,N,N,N-tetraalkyl binaphthylamine derivative, plays a dual role that is critical for both activity and selectivity. Mechanistically, the copper center coordinates with the carbonyl oxygen of the ester and the nitrogen of the ethylamine, effectively enhancing the nucleophilicity of the amine. This activation allows the aminolysis to occur rapidly under mild thermal conditions, avoiding the high energy barriers that typically necessitate harsh reagents.
Crucially, the steric bulk provided by the tetraalkyl groups on the binaphthyl backbone creates a protective environment around the reaction center. 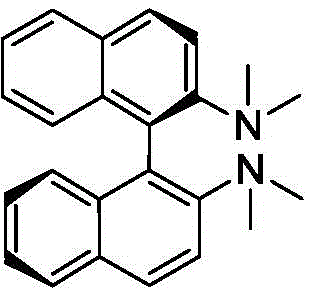 This steric hindrance prevents the formation of the planar enolate intermediate that leads to racemization in base-catalyzed systems. By maintaining a rigid transition state, the catalyst ensures that the configuration of the alpha-carbon is preserved throughout the transformation. Experimental data from the patent confirms that this mechanism yields products with chemical purity greater than 99.0% and optical purity exceeding 99.8% ee, a level of quality that is difficult to achieve with conventional non-catalytic or base-catalyzed methods.
This steric hindrance prevents the formation of the planar enolate intermediate that leads to racemization in base-catalyzed systems. By maintaining a rigid transition state, the catalyst ensures that the configuration of the alpha-carbon is preserved throughout the transformation. Experimental data from the patent confirms that this mechanism yields products with chemical purity greater than 99.0% and optical purity exceeding 99.8% ee, a level of quality that is difficult to achieve with conventional non-catalytic or base-catalyzed methods.
How to Synthesize L-Menthane Carboxamide Efficiently
The synthesis protocol outlined in the patent is designed for operational simplicity and robustness, making it highly attractive for commercial scale-up of complex flavor intermediates. The process begins with the formation of the Grignard reagent, followed by a controlled condensation with chloroformate to generate the ester intermediate. The critical step involves the addition of the copper-ligand complex to the ester solution, followed by the introduction of ethylamine. The reaction is exothermic but manageable, requiring only standard cooling to maintain the optimal 20-30°C range. Detailed standardized synthesis steps see the guide below.
- Prepare menthyl magnesium chloride from menthyl chloride and magnesium, then react with chloroformate at 20-30°C to form menthyl formate.
- Add a catalyst system comprising CuX and a chiral (R)-N,N,N,N-tetraalkyl binaphthylamine ligand to the ester solution.
- Introduce ethylamine at 20-30°C to effect aminolysis, followed by concentration and crystallization to isolate the product with >99.8% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain directors and procurement managers, the adoption of this catalytic technology translates into tangible strategic benefits beyond mere chemical efficiency. The shift from a three-step, low-yield process to a high-yield, two-step sequence fundamentally alters the cost structure of production. By eliminating the need for expensive cryogenic equipment for CO2 handling and removing the requirement for toxic cyanide reagents, the capital expenditure and operational risk profiles are significantly improved.
- Cost Reduction in Manufacturing: The most immediate impact is the dramatic increase in overall yield, rising from roughly 40% in traditional routes to over 90% with this new method. This more than doubling of efficiency means that less raw material is required to produce the same amount of finished goods, directly lowering the cost of goods sold (COGS). Additionally, the catalyst loading is extremely low (0.05-0.2 mol%), minimizing the cost contribution of the precious metal and ligand system while maximizing turnover.
- Enhanced Supply Chain Reliability: The reliance on hazardous materials like sodium cyanide in older nitrile-based routes creates significant supply chain vulnerabilities due to strict regulatory controls and transportation restrictions. By switching to a chloroformate-based esterification followed by safe aminolysis, manufacturers can secure a more stable supply of raw materials. The mild reaction conditions also reduce the likelihood of batch failures due to thermal runaways, ensuring consistent delivery schedules for downstream customers.
- Scalability and Environmental Compliance: Modern chemical manufacturing is increasingly constrained by environmental regulations regarding wastewater and hazardous waste. This process generates no wastewater during the reaction phase, as the byproduct (MgCl2) is removed by filtration prior to the aminolysis step. The absence of aqueous waste streams simplifies the environmental compliance burden and reduces the overhead costs associated with effluent treatment plants, facilitating easier scale-up from pilot to commercial production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed ester aminolysis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on performance metrics and operational parameters.
Q: How does the new copper-catalyzed method prevent racemization compared to traditional base catalysis?
A: Traditional strong base catalysis promotes enolization at the alpha-carbon, leading to racemization. The novel Cu-ligand system operates under mild, neutral conditions (20-30°C) without strong bases, preserving the chiral center and ensuring >99.8% optical purity.
Q: What are the yield improvements over the conventional Grignard-carboxylation route?
A: The conventional route involving CO2 carboxylation typically suffers from low conversion and racemization, yielding around 40%. The patented ester aminolysis route achieves total yields exceeding 90%, significantly reducing raw material consumption.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process eliminates the need for hazardous cyanide reagents and avoids high-temperature reflux or extreme low-temperature CO2 handling. The mild reaction conditions (20-30°C) and lack of wastewater generation make it highly scalable and environmentally compliant.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Menthane Carboxamide Supplier
The technological breakthroughs detailed in CN111393293B highlight the immense potential for producing high-purity cooling agents with superior economic and environmental profiles. At NINGBO INNO PHARMCHEM, we specialize in translating such advanced academic and patent innovations into robust industrial realities. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical optical purity metrics required for high-end flavor applications.
We invite you to leverage our technical expertise to optimize your supply chain for L-menthane carboxamide and related intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can drive value for your organization.
