Advanced Metal Salt Catalysis for Efficient Alpha-Diketone Manufacturing
Advanced Metal Salt Catalysis for Efficient Alpha-Diketone Manufacturing
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more sustainable, efficient, and cost-effective pathways to high-value intermediates. A significant breakthrough in this domain is documented in patent CN111807941B, which details a novel method for synthesizing alpha-diketone compounds utilizing metal salt catalysis. This technology represents a paradigm shift from traditional oxidative methods, offering a streamlined one-pot procedure that converts readily available propargyl alcohols and acid anhydrides into valuable enedione structures. For R&D directors and procurement specialists alike, this innovation addresses critical pain points regarding yield optimization, impurity control, and raw material accessibility. The ability to generate complex 1,2-dicarbonyl structures under such mild conditions opens new avenues for the production of pharmaceutical intermediates, flavoring agents, and functional materials without the heavy environmental burden typically associated with diketone synthesis.
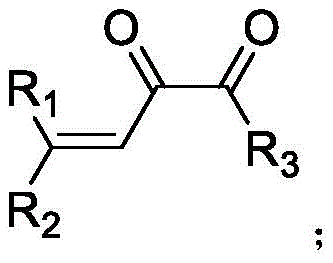
The core of this technological advancement lies in its versatility and operational simplicity. By leveraging specific metal salts, such as silver perchlorate or copper perchlorate, the reaction proceeds efficiently at temperatures ranging from -10°C to 30°C, eliminating the need for extreme thermal inputs. This patent not only defines the chemical scope but also establishes a robust framework for commercial manufacturing, ensuring that the transition from laboratory bench to pilot plant is seamless. As global demand for high-purity alpha-diketones continues to rise across the flavor, fragrance, and pharmaceutical sectors, adopting this catalytic methodology provides a strategic competitive advantage in terms of both supply chain reliability and production economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of alpha-diketones has been fraught with significant technical and economic challenges that hinder large-scale adoption. Traditional nucleophilic addition-elimination reactions, while chemically flexible, often necessitate complex multi-step operations and stringent low-temperature controls that drive up energy costs and equipment requirements. Furthermore, coupling methods, although conceptually simple, have historically suffered from immaturity in process development, frequently yielding complex mixtures of byproducts that are difficult to separate, thereby compromising the overall purity of the final API intermediate. Perhaps most critically, conventional oxidation methods rely heavily on stoichiometric amounts of metal oxidants, which are not only expensive but also pose severe toxicity and disposal issues. These oxidants often lack selectivity, leading to deep oxidation side reactions that degrade the desired product and generate hazardous waste streams, creating a substantial burden on environmental compliance and waste management protocols.
The Novel Approach
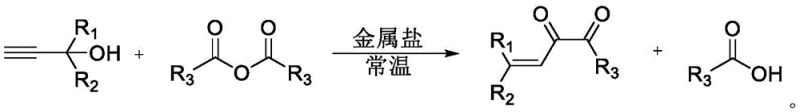
In stark contrast to these legacy techniques, the metal salt-catalyzed synthesis described in the patent offers a transformative solution that simplifies the entire production workflow. This novel approach utilizes a direct one-pot reaction between propargyl alcohols and acid anhydrides, effectively bypassing the need for hazardous oxidizing agents entirely. The reaction conditions are remarkably mild, typically proceeding at room temperature, which significantly reduces the thermal load on the reactor and minimizes the risk of thermal runaway incidents. By employing catalytic amounts of metal salts rather than stoichiometric oxidants, the process drastically reduces the generation of heavy metal waste, aligning perfectly with modern green chemistry principles. Additionally, the broad substrate scope allows for the synthesis of diverse alpha-diketone derivatives simply by varying the starting alcohol or anhydride, providing manufacturers with a flexible platform to produce a wide array of specialty chemicals without retooling the entire production line.
Mechanistic Insights into Metal Salt-Catalyzed Cyclization
From a mechanistic perspective, the efficacy of this transformation relies on the unique ability of the metal salt catalyst to activate the alkyne moiety of the propargyl alcohol towards nucleophilic attack by the acid anhydride. The metal center, typically silver or copper in a high oxidation state, coordinates with the pi-electrons of the triple bond, increasing its electrophilicity and facilitating the initial addition step. This activation lowers the energy barrier for the reaction, allowing it to proceed rapidly even at ambient temperatures. The subsequent rearrangement and elimination steps are carefully orchestrated by the catalytic cycle to ensure the formation of the stable enedione structure rather than polymerization or decomposition products. Understanding this catalytic cycle is crucial for process chemists, as it highlights the importance of maintaining anhydrous conditions and precise catalyst loading to maximize turnover numbers and prevent catalyst deactivation.
Impurity control is another critical aspect where this mechanism offers distinct advantages over oxidative routes. In traditional oxidation, over-oxidation is a persistent issue where the sensitive diketone functionality is further degraded into carboxylic acids or cleaved carbon fragments. However, in this metal salt-catalyzed pathway, the reaction is inherently self-limiting once the enedione structure is formed, as the product does not possess the same reactivity towards the catalyst under the chosen conditions. This intrinsic selectivity results in a much cleaner crude reaction profile, significantly reducing the burden on downstream purification units such as distillation columns or chromatography systems. For quality control teams, this means a more consistent impurity profile and a higher probability of meeting stringent pharmacopeial standards for residual metals and organic impurities without requiring excessive recrystallization steps.
How to Synthesize Alpha-Diketone Compounds Efficiently
Implementing this synthesis route in a practical setting requires careful attention to reagent quality and reaction parameters to ensure optimal performance. The process begins with the preparation of a thoroughly dried reaction vessel to prevent moisture-induced catalyst deactivation, followed by the sequential addition of the metal salt catalyst and the propargyl alcohol substrate. The acid anhydride is then introduced, serving both as a reactant and potentially as a solvent component depending on the specific embodiment chosen. Detailed standard operating procedures regarding mixing rates, temperature ramping, and quenching protocols are essential to replicate the high yields reported in the patent literature consistently. For a comprehensive breakdown of the specific experimental conditions and workup procedures, please refer to the standardized synthesis guide below.
- Prepare the reaction system by adding the metal salt catalyst (such as silver perchlorate) to a dried reaction vessel containing the propargyl alcohol substrate.
- Introduce the acid anhydride reagent to the mixture, maintaining a molar ratio between 1: 1 and 1:5 relative to the alcohol, optionally adding a solvent like toluene or operating solvent-free.
- Stir the reaction mixture at temperatures between -10°C and 30°C for 6 to 72 hours, followed by alkali washing and column chromatography to isolate the pure enedione product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal salt-catalyzed technology translates into tangible strategic benefits that extend far beyond simple chemical yield. The primary driver of value creation here is the drastic simplification of the raw material basket. By utilizing commodity-grade acid anhydrides and propargyl alcohols, manufacturers can decouple their supply chains from the volatile markets associated with specialized oxidizing agents. This shift not only stabilizes input costs but also mitigates the risk of supply disruptions caused by regulatory crackdowns on hazardous chemical transport. Furthermore, the ability to operate under solvent-free conditions or with common industrial solvents like toluene reduces the logistical complexity of managing large volumes of specialized reaction media, streamlining inventory management and warehousing requirements.
- Cost Reduction in Manufacturing: The economic impact of eliminating stoichiometric oxidants cannot be overstated, as these reagents often constitute a significant portion of the variable cost in traditional diketone synthesis. By replacing them with catalytic amounts of metal salts, the direct material cost per kilogram of product is significantly reduced. Additionally, the mild reaction conditions lead to lower energy consumption for heating and cooling, contributing to a smaller utility footprint. The simplified workup procedure, which often requires only basic alkali washing and standard chromatography, reduces the labor hours and consumable costs associated with complex purification trains, resulting in substantial overall cost savings for the manufacturing operation.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials ensures a robust and resilient supply chain capable of withstanding market fluctuations. Propargyl alcohols and acid anhydrides are produced at massive scales for various industries, guaranteeing a steady flow of feedstock even during periods of global chemical shortage. This availability allows for better production planning and the ability to scale up output rapidly in response to increased market demand without the long lead times typically associated with sourcing custom synthetic intermediates. Consequently, manufacturers can offer shorter delivery times to their customers, enhancing their reputation as a reliable partner in the global pharmaceutical and fine chemical marketplace.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a markedly superior profile compared to oxidative methods. The absence of toxic oxidants reduces the hazard classification of the process, lowering insurance premiums and simplifying permitting requirements for new production facilities. The reduction in heavy metal waste and organic byproducts minimizes the cost and complexity of wastewater treatment and solid waste disposal. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening environmental regulations but also appeals to end-customers who are increasingly prioritizing sustainability in their supplier selection criteria, thereby opening up new market opportunities for eco-conscious branding.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metal salt-catalyzed synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production workflows or for assessing the quality of suppliers offering these specific intermediates.
Q: What are the primary advantages of this metal salt catalyzed method over traditional oxidation routes?
A: Unlike traditional oxidation methods that often require toxic oxidants and high temperatures leading to deep oxidation byproducts, this metal salt catalyzed approach operates under mild conditions (room temperature) using inexpensive acid anhydrides, resulting in higher selectivity and simpler purification processes.
Q: Can this synthesis method be adapted for large-scale industrial production?
A: Yes, the method is highly scalable due to its solvent-free capability and the use of stable, commercially available raw materials. The mild thermal requirements reduce energy consumption and safety risks associated with exothermic oxidation reactions, making it suitable for commercial scale-up.
Q: What types of substrates are compatible with this catalytic system?
A: The system demonstrates broad substrate tolerance, successfully converting various propargyl alcohols substituted with methyl, ethyl, cyclohexyl, or phenyl groups, and reacting them with a wide range of acid anhydrides including acetic, propionic, and long-chain anhydrides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Diketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in patent CN111807941B for the production of high-value alpha-diketones. As a leading CDMO and supplier, we possess the technical expertise and infrastructure to translate these innovative laboratory protocols into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch meets the exacting standards required for pharmaceutical and fine chemical applications.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific project requirements. Whether you are looking to optimize an existing supply chain or develop a new product line based on enedione chemistry, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume needs. We encourage you to reach out for specific COA data and route feasibility assessments to determine how our capabilities can best support your strategic goals and drive efficiency in your manufacturing operations.
