Industrial Scale-Up of High-Purity N-(2-ethoxyphenyl)-N'-(4-ethylphenyl)-oxalamide via Solvent-Free Amidation
Introduction to Advanced UV Absorbent Synthesis
The global demand for high-performance ultraviolet absorbents continues to surge across the coatings, plastics, and agrochemical sectors, driven by the critical need to protect materials from photodegradation. Patent CN110343053B introduces a transformative preparation method for N-(2-ethoxyphenyl)-N'-(4-ethylphenyl)-oxalamide, a novel UV stabilizer that addresses longstanding inefficiencies in traditional manufacturing. This technology leverages a streamlined two-step synthetic pathway that bypasses the need for complex purification protocols often associated with oxalamide derivatives. By optimizing reaction stoichiometry and eliminating unnecessary acid-binding agents, the process achieves exceptional yield and purity profiles that are essential for downstream applications requiring rigorous quality standards. The strategic design of this synthesis not only enhances product performance but also aligns with modern green chemistry principles by minimizing waste generation.
For R&D directors and technical leaders, the significance of this patent lies in its ability to produce a highly consistent impurity profile, which is vital for regulatory compliance in sensitive industries. The method utilizes readily available starting materials such as oxalyl chloride ethyl ester and substituted anilines, ensuring that supply chain volatility is minimized. Furthermore, the reaction conditions are carefully tuned to prevent the formation of hydrochloride salts that typically complicate isolation procedures in conventional amide syntheses. This technical advancement represents a paradigm shift from batch-heavy, waste-intensive processes to a more efficient, continuous-flow compatible methodology. As we delve deeper into the mechanistic and commercial implications, it becomes clear that this patent offers a robust foundation for scaling production to meet global market demands.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for oxalamide-based UV absorbents have historically been plagued by low atom economy and cumbersome workup procedures that hinder industrial scalability. Conventional methods often rely on the use of stoichiometric amounts of tertiary amines, such as triethylamine, to scavenge hydrochloric acid generated during the acylation step. This practice results in the formation of large quantities of amine hydrochloride salts, which create significant disposal challenges and increase the volume of wastewater requiring treatment. Additionally, the presence of these salts can trap product within the solid matrix, leading to reduced isolated yields that typically range between 68% and 88%. The necessity for extensive washing and filtration steps to remove these byproducts not only extends production cycles but also introduces opportunities for product loss and contamination.
Furthermore, existing technologies frequently employ solvent-heavy conditions for the final coupling reaction, which complicates the removal of residual solvents to meet stringent purity specifications. The thermal instability of some intermediates under prolonged heating in solution can lead to degradation products that are difficult to separate via standard crystallization techniques. These inefficiencies translate directly into higher operational expenditures and a larger environmental footprint, making conventional routes less attractive for manufacturers aiming to reduce their carbon intensity. The accumulation of organic waste and the energy consumption associated with solvent recovery distillation further exacerbate the economic disadvantages of these legacy processes. Consequently, there is a pressing industry need for a cleaner, more direct synthetic strategy that can overcome these inherent bottlenecks.
The Novel Approach
The innovative methodology described in patent CN110343053B fundamentally reengineers the synthesis of N-(2-ethoxyphenyl)-N'-(4-ethylphenyl)-oxalamide by eliminating the reliance on acid-binding agents in the initial acylation step. By carefully controlling the addition rate of o-ethylaniline to oxalyl chloride ethyl ester at elevated temperatures, the process allows hydrogen chloride gas to evolve continuously rather than precipitating as a salt. This clever manipulation of reaction kinetics ensures that the intermediate oxamide ester is formed with exceptional purity, exceeding 99%, without the generation of solid waste streams. The absence of amine salts simplifies the workup procedure to a straightforward aqueous wash and solvent removal, drastically reducing the time and resources required for isolation. This approach not only improves the overall mass balance of the reaction but also facilitates the recycling of the reaction solvent, contributing to a more sustainable manufacturing cycle.
In the subsequent amidation step, the novel process adopts a solvent-free condition catalyzed by a Lewis base, which represents a significant departure from traditional solution-phase chemistry. Operating at temperatures between 120°C and 170°C, the reaction drives the equilibrium forward by continuously evaporating the ethanol byproduct, thereby pushing the conversion to completion without the need for excess reagents. This solvent-less technique eliminates the energy-intensive distillation steps required to recover bulk solvents, resulting in a drastic reduction in processing time and utility consumption. The final product is obtained through a simple methanol crystallization that effectively removes insoluble impurities, yielding a material with purity levels consistently above 98%. This streamlined workflow demonstrates how process intensification can be achieved through smart reaction engineering rather than brute-force purification.
Mechanistic Insights into Lewis Base-Catalyzed Amidation
The core of this synthetic success lies in the precise orchestration of nucleophilic acyl substitution mechanisms across two distinct stages, each optimized for maximum efficiency and selectivity. In the first stage, the reaction between oxalyl chloride ethyl ester and o-ethylaniline proceeds via a classic addition-elimination pathway where the amine nitrogen attacks the electrophilic carbonyl carbon. The patent specifies a molar ratio of 1.5:1 to 4:1 favoring the acid chloride, which ensures that the mono-substituted intermediate is formed preferentially over the di-substituted byproduct. The evolution of HCl gas is a critical thermodynamic driver; by maintaining the temperature between 30°C and 70°C, the system prevents the protonation of the unreacted amine, which would otherwise deactivate the nucleophile and halt the reaction. This gas-phase removal of the leaving group is a masterful example of leveraging Le Chatelier's principle to drive reaction completion without chemical scavengers.
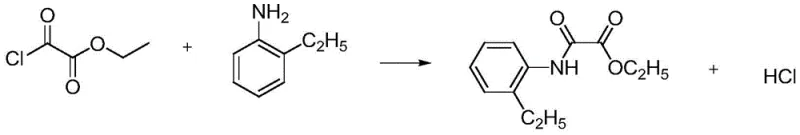
The second stage involves the aminolysis of the ester moiety by o-aminophenethyl ether, a transformation that is typically sluggish due to the lower nucleophilicity of the aniline derivative compared to aliphatic amines. The introduction of a Lewis base catalyst, such as sodium methoxide or potassium tert-butoxide, serves to deprotonate the aniline nitrogen or activate the carbonyl group, significantly lowering the activation energy for the nucleophilic attack. Operating under solvent-free conditions at elevated temperatures enhances the collision frequency between reactant molecules, while the continuous removal of ethanol shifts the equilibrium entirely towards the desired diamide product. This mechanistic understanding highlights the importance of catalyst selection and thermal management in achieving high conversion rates without compromising product integrity. The result is a clean reaction profile that minimizes side reactions and simplifies the downstream purification landscape.
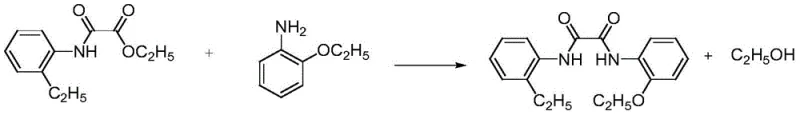
Impurity control is inherently built into this mechanism through the physical properties of the byproducts and the phase behavior of the reaction mixture. Since the primary byproduct in the first step is gaseous HCl, there is no risk of salt entrapment within the crystal lattice of the intermediate. In the second step, the ethanol byproduct is volatile and removed under vacuum or heat, preventing it from participating in reverse reactions or solvating impurities. Any unreacted starting materials or minor side products are effectively separated during the methanol crystallization step, where the target molecule exhibits differential solubility. This multi-layered approach to impurity management ensures that the final API or specialty chemical meets the rigorous specifications required for high-end applications. The robustness of this mechanism against variations in raw material quality further underscores its suitability for large-scale commercial deployment.
How to Synthesize N-(2-ethoxyphenyl)-N'-(4-ethylphenyl)-oxalamide Efficiently
Implementing this patented synthesis requires careful attention to reaction parameters and safety protocols, particularly regarding the handling of acid chlorides and the management of exothermic events. The process begins with the preparation of the oxamide ester intermediate, where the controlled addition of the amine to the acid chloride is paramount to prevent runaway reactions and ensure high selectivity. Following the isolation of this key intermediate, the solvent-free amidation step demands precise temperature control to facilitate ethanol evaporation without degrading the thermally sensitive oxalamide backbone. Operators must utilize equipment capable of withstanding mild vacuum and elevated temperatures to maximize the efficiency of the byproduct removal. Detailed standard operating procedures regarding mixing speeds, addition rates, and crystallization cooling profiles are essential to replicate the high yields reported in the patent literature consistently.
- Synthesize the oxamide ester intermediate by reacting oxalyl chloride ethyl ester with o-ethylaniline in a chlorinated solvent at 30-70°C, utilizing gas evolution to drive the reaction without acid binding agents.
- Perform the final amidation by reacting the purified oxamide ester intermediate with o-aminophenethyl ether using a Lewis base catalyst under solvent-free conditions at 120-170°C.
- Isolate the final high-purity product by evaporating ethanol byproduct, cooling the mixture, and crystallizing from methanol to remove insoluble impurities.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers profound advantages in terms of cost structure and operational reliability. The elimination of expensive acid-binding agents like triethylamine removes a significant line item from the bill of materials, while simultaneously reducing the costs associated with waste disposal and wastewater treatment. The ability to recycle solvents from the first step and operate the second step without any solvent further compresses the variable costs of production, making the final product highly competitive in price-sensitive markets. Moreover, the use of commodity chemicals such as oxalyl chloride derivatives and substituted anilines ensures that raw material sourcing remains stable and unaffected by the supply constraints often seen with exotic reagents. This stability is crucial for long-term supply agreements and helps mitigate the risks associated with market volatility.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by removing the need for stoichiometric acid scavengers and reducing solvent consumption through solvent-free technology. By avoiding the generation of solid salt waste, manufacturers save significantly on hazardous waste disposal fees and the energy costs associated with drying and treating effluent streams. The high yield of the intermediate step, approaching quantitative levels, means that less raw material is wasted, directly improving the overall material efficiency of the plant. Additionally, the simplified workup procedures reduce labor hours and equipment occupancy time, allowing for higher throughput without capital expansion. These cumulative efficiencies translate into a lower cost of goods sold, providing a strategic margin advantage in the competitive UV absorbent market.
- Enhanced Supply Chain Reliability: The reliance on widely available, bulk chemical feedstocks ensures that production schedules are not disrupted by the scarcity of specialized reagents. The robustness of the reaction conditions, which do not require extreme pressures or cryogenic temperatures, reduces the likelihood of unplanned downtime due to equipment failure or utility fluctuations. The high purity of the crude product minimizes the need for reprocessing or second crops, ensuring that delivery timelines are met with greater consistency. This reliability is particularly valuable for customers in the automotive and coatings industries who operate on just-in-time manufacturing models and cannot tolerate supply interruptions. The scalability of the process from pilot to commercial scale further guarantees that supply can be ramped up quickly to meet surging demand.
- Scalability and Environmental Compliance: The process is inherently designed for green manufacturing, aligning with increasingly stringent global environmental regulations regarding volatile organic compounds and hazardous waste. The reduction in solvent usage and the elimination of amine salt waste significantly lower the environmental footprint of the facility, simplifying the permitting process for capacity expansions. The mild reaction conditions and lack of high-pressure requirements make the technology easily transferable to existing multipurpose reactors, facilitating rapid scale-up without major infrastructure investment. This adaptability ensures that manufacturers can respond agilely to market changes while maintaining compliance with ISO 14001 and other sustainability standards. The overall simplicity of the process reduces the training burden on operators and lowers the risk of human error during scale-up.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented UV absorbent synthesis. These insights are derived directly from the experimental data and process descriptions found in patent CN110343053B, providing a factual basis for decision-making. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement teams negotiating supply contracts. The answers highlight the specific advantages of this method over legacy technologies, focusing on purity, yield, and environmental impact. We encourage stakeholders to review these details to fully appreciate the value proposition of this advanced manufacturing approach.
Q: How does this patent improve the purity of the UV absorbent compared to conventional methods?
A: The patented process eliminates the formation of difficult-to-remove salt byproducts in the first step by avoiding acid binding agents, and utilizes a solvent-free high-temperature crystallization in the second step to achieve purity levels exceeding 98%.
Q: What are the environmental benefits of this synthesis route?
A: By replacing traditional acid scavengers like triethylamine with a gas-evolution mechanism and eliminating solvents in the final coupling step, the process significantly reduces hazardous wastewater generation and solvent consumption.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the method utilizes common raw materials, operates at moderate temperatures without high-pressure requirements, and achieves yields up to 90%, making it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ultraviolet Absorbent Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance UV absorbents play in protecting the longevity and aesthetic appeal of advanced materials. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN110343053B are fully realized in practical manufacturing. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our dedication to process excellence means that we can offer a reliable ultraviolet absorbent supplier partnership that guarantees consistency, quality, and technical support throughout the product lifecycle.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals. Let us help you secure a stable, high-quality supply of N-(2-ethoxyphenyl)-N'-(4-ethylphenyl)-oxalamide that empowers your products to withstand the test of time and nature.
