Advanced Synthesis of Trans-(N-4-Boc-aminocyclohexane)acetic Acid for Commercial Scale-up
Introduction to Patent CN114426504A
The pharmaceutical industry constantly seeks robust, scalable, and environmentally benign pathways for constructing complex amino acid derivatives, and patent CN114426504A presents a groundbreaking solution for the synthesis of trans-(N-4-Boc-aminocyclohexane)acetic acid (CAS: 189153-10-4). This intellectual property outlines a sophisticated three-step sequence that fundamentally reimagines the construction of the cyclohexane-acetic acid scaffold, moving away from hazardous traditional chemistries toward a safer, more efficient protocol. By leveraging a strategic combination of chemoselective reduction, Appel bromination, and Grignard carboxylation, the inventors have established a route that delivers high-purity products suitable for demanding API applications. The significance of this technology lies not just in its chemical elegance, but in its practical ability to circumvent the use of genotoxic reagents and expensive transition metal catalysts that have historically plagued this chemical space. For global supply chains, this represents a pivotal shift toward more sustainable and reliable manufacturing capabilities for critical pharmaceutical intermediates.
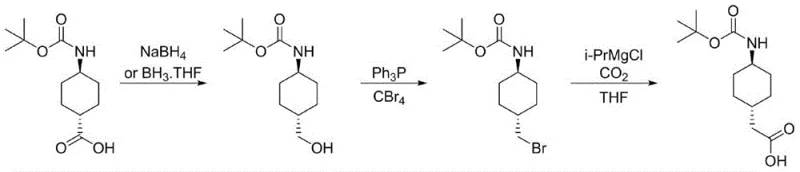
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of trans-(N-4-Boc-aminocyclohexane)acetic acid has been fraught with significant technical and economic hurdles that limit its viability for large-scale commercial operations. Conventional methodologies often depend on the catalytic hydrogenation of nitro precursors, such as 4-nitrophenylacetonitrile, which necessitates the use of costly precious metal catalysts like palladium or platinum on carbon. Furthermore, these hydrogenation processes frequently yield mixtures of cis and trans isomers, forcing manufacturers to engage in multiple, yield-depleting recrystallization steps to isolate the desired trans-configuration. Even more concerning are the alternative routes that rely on potassium cyanide for carbon chain extension, introducing severe safety hazards and complex waste disposal challenges due to the extreme toxicity of cyanide salts. These legacy processes result in poor atom economy, high operational costs, and significant environmental liabilities that are increasingly unacceptable in modern green chemistry frameworks.
The Novel Approach
In stark contrast to these outdated techniques, the novel approach detailed in the patent utilizes a streamlined, three-step sequence that prioritizes safety, cost-efficiency, and stereochemical integrity from the outset. The process begins with the direct reduction of trans-4-Boc-aminocyclohexanecarboxylic acid, utilizing readily available reducing agents like sodium borohydride or borane-THF complexes to generate the corresponding alcohol intermediate with high fidelity. This is followed by a mild Appel reaction using carbon tetrabromide and triphenylphosphine to convert the hydroxyl group into a reactive bromide, effectively activating the molecule for the subsequent carbon-carbon bond formation. The final step employs a Grignard exchange with isopropylmagnesium chloride followed by carbon dioxide trapping, a clever maneuver that extends the carbon chain by one unit without introducing any toxic cyanide species. This methodology not only simplifies the purification workflow but also ensures that the final product meets stringent purity specifications required for downstream pharmaceutical synthesis.
Mechanistic Insights into Grignard Carboxylation and Reduction
The core of this synthetic strategy relies on a highly chemoselective reduction mechanism that preserves the sensitive Boc protecting group while efficiently converting the carboxylic acid moiety into a primary alcohol. Whether utilizing sodium borohydride in a mixed solvent system of THF and methanol or employing borane-THF complexes, the reaction conditions are meticulously tuned to prevent over-reduction or deprotection of the amine functionality. The subsequent activation of the alcohol via the Appel reaction proceeds through the formation of an alkoxyphosphonium salt, which serves as an excellent leaving group for nucleophilic attack by the bromide ion generated in situ. This transformation is critical as it converts a relatively inert hydroxyl group into a highly reactive alkyl bromide, setting the stage for the pivotal carbon chain extension step that defines the utility of this entire pathway.
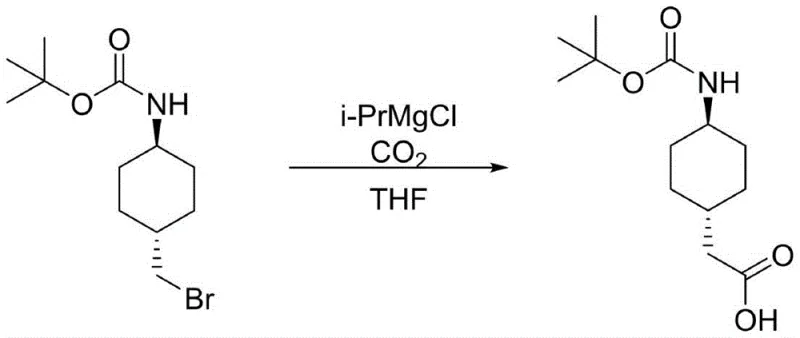
The final mechanistic highlight is the halogen-magnesium exchange followed by electrophilic trapping with carbon dioxide, which elegantly constructs the acetic acid side chain. Upon treatment with isopropylmagnesium chloride at controlled low temperatures ranging from -10°C to 0°C, the alkyl bromide undergoes a rapid exchange to form the corresponding organomagnesium species. This nucleophilic carbon center then attacks the electrophilic carbon of carbon dioxide, forming a carboxylate salt that is subsequently protonated during the acidic workup to yield the final trans-(N-4-Boc-aminocyclohexane)acetic acid. This sequence avoids the formation of genotoxic impurities often associated with cyanide-based homologation reactions, ensuring a cleaner impurity profile that is easier to control during scale-up.
How to Synthesize Trans-(N-4-Boc-aminocyclohexane)acetic Acid Efficiently
Implementing this synthesis requires precise control over reaction parameters, particularly temperature and stoichiometry, to maximize yield and minimize byproduct formation during the sensitive Grignard step. The patent provides explicit guidance on molar ratios, suggesting a slight excess of reducing agent (1:1.10-1.20) and careful management of the Grignard reagent (1:1.15-1.20) to ensure complete conversion without excessive waste. Operators must maintain strict anhydrous conditions during the bromination and Grignard phases to prevent hydrolysis of the reactive intermediates, which could otherwise lead to significant yield losses. The following guide outlines the standardized operational procedure derived directly from the patent examples to ensure reproducible results in a GMP-compliant environment.
- Reduce trans-4-Boc-aminocyclohexanecarboxylic acid using sodium borohydride or borane-THF to obtain the corresponding alcohol intermediate.
- Convert the alcohol to the alkyl bromide using carbon tetrabromide and triphenylphosphine in dichloromethane via an Appel reaction.
- Perform a halogen-magnesium exchange with isopropylmagnesium chloride followed by carbon dioxide trapping to extend the carbon chain and form the final acetic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers profound strategic advantages that directly impact the bottom line and operational resilience of pharmaceutical manufacturing. By eliminating the dependency on volatile and heavily regulated toxic cyanide reagents, companies can drastically simplify their logistics, reduce insurance premiums, and avoid the costly infrastructure investments required for handling extremely hazardous materials. Furthermore, the removal of expensive precious metal catalysts from the process flow significantly lowers the raw material cost base, allowing for more competitive pricing structures in the final API market. The simplified purification workflow, which avoids multiple recrystallizations, translates directly into reduced processing time and lower energy consumption, enhancing the overall sustainability profile of the supply chain.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the avoidance of complex isomer separation steps lead to substantial cost savings in raw materials and processing time. By utilizing commodity chemicals like sodium borohydride and carbon tetrabromide instead of specialized hydrogenation catalysts, the direct material costs are significantly lowered while maintaining high reaction efficiency. Additionally, the higher overall yield achieved through this streamlined route means less starting material is wasted, further driving down the cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: Sourcing non-toxic, widely available reagents ensures a more stable and resilient supply chain that is less susceptible to regulatory disruptions or vendor shortages. Unlike cyanide-based processes which face strict transportation and storage regulations, the reagents used in this novel method are standard industrial chemicals that can be procured from multiple global suppliers with ease. This diversification of the supply base reduces the risk of production stoppages and ensures consistent availability of the critical intermediate for downstream drug manufacturing.
- Scalability and Environmental Compliance: The process is inherently designed for large-scale production, utilizing simple workup procedures like extraction and distillation that are easily transferable from pilot plant to commercial manufacturing scales. The absence of heavy metal residues and genotoxic cyanide byproducts simplifies waste treatment protocols, ensuring full compliance with increasingly stringent environmental regulations across major pharmaceutical markets. This 'green' profile not only mitigates regulatory risk but also aligns with the corporate sustainability goals of major multinational pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, providing clarity on its operational feasibility and quality advantages. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, offering a transparent view of the technology's capabilities. Understanding these details is crucial for technical teams evaluating the integration of this intermediate into their existing development pipelines.
Q: Why is this new synthesis route preferred over traditional catalytic hydrogenation methods?
A: Traditional methods often rely on expensive heavy metal catalysts for hydrogenation and require multiple recrystallizations to separate cis/trans isomers, leading to low overall yields. This novel route avoids heavy metals entirely and utilizes a stereospecific starting material strategy that bypasses the need for difficult isomer separation.
Q: Does this process involve the use of toxic cyanide reagents?
A: No, a significant advantage of this patented method is the complete elimination of toxic cyanide sources like potassium cyanide, which are traditionally used for chain extension. This makes the process significantly safer for operators and easier to manage from an environmental compliance perspective.
Q: What represents the key chain-extension step in this methodology?
A: The critical step involves converting the intermediate alcohol to a bromide, followed by a Grignard exchange using isopropylmagnesium chloride. The resulting organomagnesium species is then trapped with carbon dioxide to effectively add one carbon atom, forming the acetic acid side chain with high efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-(N-4-Boc-aminocyclohexane)acetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to ensure the consistent supply of high-quality pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel patent can be fully realized at an industrial level. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of trans-(N-4-Boc-aminocyclohexane)acetic acid meets the exacting standards required for global drug registration. Our commitment to technical excellence allows us to navigate the complexities of organic synthesis while delivering products that support your critical R&D and manufacturing timelines.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project, ensuring a competitive edge through superior process chemistry. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and product quality.
