Advanced Synthetic Route for Royaljelly Acid: Scaling High-Purity 10-HAD for Global Markets
The pharmaceutical and nutritional industries have long relied on Royaljelly acid, chemically known as trans-10-hydroxy-2-decenoic acid (10-HAD), for its potent bioactive properties ranging from immunomodulation to potential antitumor activity. Historically, sourcing this critical compound involved extraction from royal jelly, a process plagued by low yields, high costs, and inconsistent purity due to the complex biological matrix. A significant technological shift is documented in patent CN102206151B, which introduces a robust chemical synthesis method that bypasses these biological limitations entirely. This novel approach leverages a DMAP-catalyzed condensation strategy combined with TEMPO-mediated oxidation to construct the carbon skeleton with high stereoselectivity. For R&D Directors and Procurement Managers seeking a reliable pharma intermediates supplier, this synthetic pathway represents a paradigm shift towards manufacturing consistency. By eliminating the variability inherent in natural extraction, this method ensures a stable supply of high-purity Royaljelly acid suitable for stringent pharmaceutical applications. The transition from biological sourcing to precise chemical synthesis not only secures the supply chain but also opens new avenues for cost reduction in nutritional ingredients manufacturing without compromising the biological efficacy of the final active molecule.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for obtaining Royaljelly acid have historically been divided between natural extraction and early-generation chemical synthesis, both of which present substantial hurdles for commercial scale-up of complex pharmaceutical intermediates. Natural extraction suffers from inherently low content, typically ranging between 1.4% and 1.8% in royal jelly, necessitating the processing of massive quantities of raw material to obtain negligible amounts of product. This results in exorbitant costs and significant environmental waste, making it unsustainable for large-volume demand. On the chemical synthesis front, classical routes often rely on Wittig-Horner reactions or Knoevenagel condensations to establish the critical unsaturated acid structure. These legacy methods frequently struggle with stereoselectivity, producing mixtures of (E) and (Z) isomers that are notoriously difficult to separate, thereby reducing overall yield and purity. Furthermore, Wittig reagents often require harsh, anhydrous conditions and generate stoichiometric amounts of phosphine oxide waste, creating significant downstream purification burdens and environmental compliance issues. The instability of key intermediates like 8-hydroxy octanal in these traditional routes also complicates storage and handling, introducing risks of polymerization or oxidation that can derail production batches. These cumulative inefficiencies render conventional methods inadequate for meeting the rigorous quality and volume requirements of modern global supply chains.
The Novel Approach
The innovative synthesis method described in the patent data fundamentally re-engineers the production pathway to address the specific bottlenecks of atom economy and stereoselectivity. By utilizing 8-alkanoyloxy octanal as a stabilized intermediate, the process avoids the instability issues associated with free hydroxy aldehydes. The core breakthrough lies in the use of 4-dimethylaminopyridine (DMAP) as a nucleophilic catalyst for the condensation with monoethyl malonate. This catalytic system operates under significantly milder conditions compared to the strong bases required for Wittig reactions, thereby minimizing side reactions and degradation. Crucially, this approach demonstrates high stereoselectivity, predominantly yielding the desired (E)-isomer configuration which is essential for the biological activity of Royaljelly acid. The subsequent saponification step is straightforward and efficient, cleaving the ester protecting groups to reveal the target hydroxy acid without requiring complex chromatographic separations. This streamlined workflow not only simplifies the operational protocol but also drastically reduces the consumption of solvents and reagents. For supply chain heads, this translates to a more predictable manufacturing timeline and reduced lead time for high-purity nutritional ingredients, as the process is less susceptible to the batch-to-batch variability that characterizes older synthetic technologies.
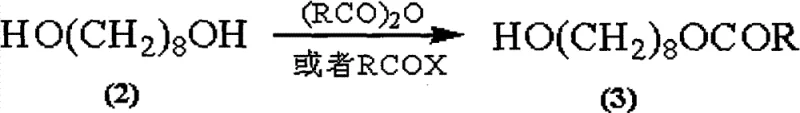
Mechanistic Insights into DMAP-Catalyzed Condensation and TEMPO Oxidation
The chemical elegance of this synthesis lies in its two-stage functionalization strategy, beginning with the precise protection of the diol starting material. The initial step involves the reaction of 1,8-octanediol with acid anhydrides or acyl halides in the presence of a solid carrier like silica gel. This heterogeneous condition enhances chemo-selectivity, ensuring that mono-acylation occurs preferentially over di-acylation, achieving yields exceeding 95%. The resulting 8-alkanoyloxy octanol serves as a robust scaffold for the subsequent oxidation step. Here, the patent employs a TEMPO-catalyzed oxidation system using hypochlorite as the terminal oxidant. TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) acts as a stable nitroxyl radical that mediates the selective oxidation of the primary alcohol to the aldehyde without over-oxidizing to the carboxylic acid at this stage. This selectivity is paramount, as it preserves the oxygenation pattern required for the final molecule while establishing the aldehyde functionality needed for chain extension. The mildness of the TEMPO system, operating effectively at temperatures between 0°C and 20°C, prevents the degradation of the sensitive ester linkage installed in the previous step. This mechanistic control ensures that the intermediate 8-alkanoyloxy octanal is produced with high fidelity, setting the stage for the final carbon-carbon bond formation.
Following the oxidation, the construction of the decenoic acid skeleton is achieved through a DMAP-catalyzed condensation reaction. In this critical transformation, the aldehyde intermediate reacts with monoethyl malonate in the presence of DMAP, which activates the malonate for nucleophilic attack. This mechanism bypasses the need for phosphorous ylides, thereby eliminating the generation of phosphine oxide byproducts and improving the overall atom economy of the process. The reaction conditions favor the formation of the thermodynamically stable (E)-alkene geometry, effectively solving the isomerization issues prevalent in Wittig-type reactions. The resulting (E)-10-alkanoyloxy-2-decenoic acid alkyl ester is then subjected to saponification using aqueous alkaline materials such as sodium hydroxide or potassium hydroxide. This hydrolysis step simultaneously removes the acyl protecting group and the ethyl ester, directly yielding the target Royaljelly acid. The simplicity of this final workup, often requiring only acidification and recrystallization, underscores the process's suitability for industrial enforcement. By integrating these mechanistic advantages, the synthesis achieves a level of purity and structural integrity that is difficult to match with conventional methodologies.
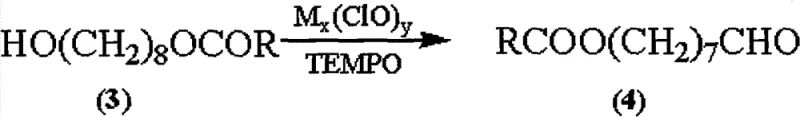
How to Synthesize Royaljelly Acid Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize the benefits of the catalytic systems described. The process begins with the preparation of the protected alcohol intermediate, where the ratio of carrier to substrate and the choice of acylating agent are critical for maintaining selectivity. Following isolation, the TEMPO oxidation must be conducted with precise control over temperature and oxidant addition rates to prevent over-oxidation or chlorination side reactions. The final condensation step relies on the efficient removal of water or alcohol byproducts to drive the equilibrium towards the desired unsaturated ester. While the general chemistry is robust, scaling these reactions requires optimized mixing and heat transfer to maintain the mild conditions that define the process's success. The detailed standardized synthesis steps see the guide below for specific operational parameters and stoichiometry.
- Selective mono-acylation of 1,8-octanediol using silica gel support to achieve high chemo-selectivity.
- TEMPO-catalyzed oxidation of the protected alcohol to the corresponding aldehyde under mild conditions.
- DMAP-catalyzed condensation with monoethyl malonate followed by saponification to yield trans-10-hydroxy-2-decenoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic methodology offers profound advantages that directly address the pain points of procurement managers and supply chain directors. The primary benefit is the drastic simplification of the purification train. By avoiding the formation of difficult-to-separate isomer mixtures and eliminating heavy metal or phosphorous waste streams, the downstream processing costs are significantly reduced. This efficiency translates into a more competitive cost structure for the final active ingredient, allowing for better margin management in the production of finished pharmaceutical or nutraceutical products. Furthermore, the reliance on readily available commodity chemicals such as 1,8-octanediol, acetyl chloride, and malonate esters ensures that the raw material supply chain is resilient and not subject to the volatility of specialized reagent markets. This stability is crucial for maintaining continuous production schedules and meeting long-term contract obligations without interruption. The environmental profile of the process is also markedly improved, with reduced waste generation and the absence of toxic heavy metals, facilitating easier regulatory compliance and lowering the costs associated with waste disposal and environmental remediation.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in solvent usage due to higher reaction concentrations contribute to substantial cost savings. The high selectivity of the DMAP-catalyzed step minimizes the loss of valuable starting materials to side products, thereby improving the overall material yield. Additionally, the simplified workup procedures reduce the labor and energy costs associated with extensive chromatographic purifications or multiple recrystallizations. These cumulative efficiencies result in a lower cost of goods sold (COGS), providing a strategic advantage in price-sensitive markets. The process design inherently supports lean manufacturing principles by reducing the number of unit operations required to reach the final specification.
- Enhanced Supply Chain Reliability: By utilizing a fully synthetic route, the supply of Royaljelly acid is decoupled from the seasonal and biological constraints of beekeeping and royal jelly harvesting. This independence ensures a consistent year-round supply, mitigating the risks of shortages caused by environmental factors or agricultural fluctuations. The use of stable intermediates allows for the stocking of key precursors, enabling rapid response to spikes in demand without the long lead times associated with biological extraction. This reliability is essential for pharmaceutical manufacturers who require guaranteed supply continuity to maintain their own production schedules and regulatory filings. The robustness of the chemical process also means that technology transfer to different manufacturing sites is straightforward, further diversifying supply risk.
- Scalability and Environmental Compliance: The reaction conditions are mild and operate at near-ambient temperatures and pressures, which simplifies the engineering requirements for large-scale reactors. This ease of scale-up allows for seamless transition from pilot plant to commercial production volumes without the need for specialized high-pressure or cryogenic equipment. The absence of hazardous reagents like sodium hydride or toxic phosphines reduces the safety risks for plant operators and lowers the insurance and safety compliance costs. Moreover, the reduced waste profile aligns with green chemistry principles, making the process more attractive to environmentally conscious stakeholders and regulators. This compliance advantage facilitates faster regulatory approvals and enhances the brand reputation of companies adopting this sustainable manufacturing technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Royaljelly acid synthesized via this advanced pathway. These answers are derived from the specific technical advantages and operational data detailed in the patent literature, providing clarity for stakeholders evaluating this technology. Understanding these nuances is critical for making informed decisions about sourcing and process adoption. The responses highlight the superiority of this method over traditional extraction and synthesis techniques in terms of purity, cost, and scalability.
Q: How does this synthetic method improve upon traditional Wittig reactions?
A: Unlike Wittig-Horner reactions which often produce Z/E isomer mixtures and require harsh conditions, this DMAP-catalyzed route ensures high stereoselectivity for the (E)-isomer with simpler purification and better atom economy.
Q: What are the purity specifications achievable with this process?
A: The process utilizes intermediate protection and mild oxidation steps that minimize byproduct formation, allowing for final product purity exceeding 99% HPLC content after recrystallization.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method avoids expensive transition metal catalysts and uses readily available reagents like acetyl chloride and TEMPO, making it highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Royaljelly Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of effective health and pharmaceutical products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative synthesis routes described in patent CN102206151B can be effectively translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Royaljelly acid meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence allows us to navigate the complexities of stereoselective synthesis and deliver products with consistent quality profiles. By leveraging our expertise in catalytic processes and purification technologies, we provide a secure and reliable source for this valuable bioactive compound.
We invite procurement leaders and R&D teams to collaborate with us to optimize their supply chains and reduce manufacturing costs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality specifications. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Partnering with us ensures access to cutting-edge synthesis technologies and a supply chain partner dedicated to your long-term success. Let us help you secure a stable supply of high-purity Royaljelly acid for your next generation of products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
