Advanced Glycerol Valorization: High-Purity 1,3-Dihydroxyacetone Manufacturing and Commercial Scale-Up
The global fine chemical landscape is currently witnessing a strategic shift towards valorizing bio-based feedstocks, driven by the massive oversupply of crude glycerol from the biodiesel industry. Patent CN101412706B introduces a robust and highly selective chemical methodology for converting this abundant low-value byproduct into 1,3-dihydroxyacetone (DHA), a high-value multifunctional intermediate. Unlike traditional direct oxidation pathways that struggle with regioselectivity, this invention employs a sophisticated protection-oxidation-deprotection sequence. By temporarily masking the primary hydroxyl groups of glycerol as a benzaldehyde acetal, the process directs oxidation exclusively to the secondary hydroxyl position. This technical breakthrough addresses long-standing challenges in impurity control and yield optimization, offering a viable pathway for industrial manufacturers seeking to diversify their portfolio with high-margin specialty chemicals derived from renewable resources.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 1,3-dihydroxyacetone has relied heavily on enzymatic fermentation or direct catalytic oxidation, both of which present significant scalability and efficiency bottlenecks. Enzymatic processes, while specific, are severely constrained by the toxicity of high glycerol concentrations to the microbial cultures, necessitating dilute reaction conditions that result in low volumetric productivity and energy-intensive downstream concentration steps. Furthermore, direct chemical oxidation using noble metal catalysts like Platinum-Bismuth systems often suffers from poor selectivity, frequently yielding less than 40% of the desired ketone due to over-oxidation and C-C bond cleavage side reactions. These inefficiencies lead to complex purification trains, high solvent consumption, and ultimately, a cost structure that limits the competitiveness of the final API intermediate in price-sensitive markets.
The Novel Approach
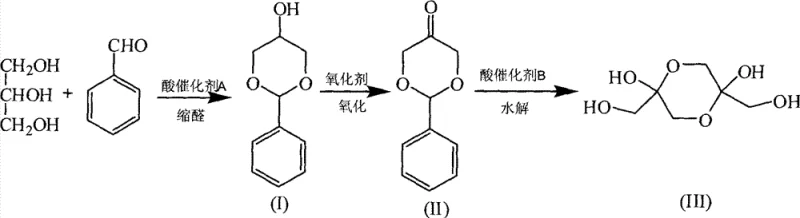
The methodology disclosed in the patent circumvents these selectivity issues through a clever synthetic design that prioritizes functional group differentiation. By first converting glycerol into a cyclic acetal using benzaldehyde, the two primary hydroxyl groups are effectively protected, leaving the secondary hydroxyl group exposed and activated for oxidation. This structural modification ensures that subsequent oxidation steps target only the desired position, drastically reducing the formation of glyceric acid and other degradation byproducts common in direct oxidation routes. The final hydrolysis step efficiently removes the protecting group under mild acidic conditions, regenerating the benzaldehyde for potential recycling and releasing the target 1,3-dihydroxyacetone dimer with exceptional purity levels reaching 99%, thereby streamlining the entire manufacturing workflow.
Mechanistic Insights into Protective Group Strategy and Selective Oxidation
The core of this synthesis lies in the thermodynamic stability of the five-membered cyclic acetal formed during the initial condensation of glycerol and benzaldehyde. Under acid catalysis, typically utilizing p-toluenesulfonic acid or solid acid resins, the reaction proceeds via a carbocation intermediate that selectively cyclizes with the 1,3-diol motif of glycerol. This step is crucial as it locks the molecular conformation, preventing the primary alcohols from participating in the subsequent redox chemistry. The use of a water-carrying agent such as benzene or toluene facilitates the azeotropic removal of water, driving the equilibrium towards the acetal product according to Le Chatelier's principle. This protection strategy is not merely a synthetic convenience but a fundamental requirement for achieving the high regioselectivity that defines the commercial viability of this process.
Following protection, the oxidation of the secondary alcohol is achieved using versatile oxidizing systems ranging from stoichiometric chromium reagents like Collins or PCC to catalytic aerobic systems employing TEMPO and transition metal salts. In the catalytic variant, the TEMPO radical mediates the hydrogen abstraction from the alpha-carbon, while the transition metal co-catalyst facilitates the regeneration of the active oxoammonium species using molecular oxygen or air. This mechanistic pathway allows for mild reaction conditions at room temperature, preserving the integrity of the acid-sensitive acetal linkage. The resulting ketone intermediate is stable and易于 to purify via crystallization, ensuring that impurities carried over from the oxidation step do not contaminate the final hydrolysis product, thus maintaining the stringent quality standards required for pharmaceutical and food-grade applications.
How to Synthesize 1,3-Dihydroxyacetone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the importance of precise temperature control and catalyst selection to maximize yield. The process begins with the acetalization of glycerol, followed by the selective oxidation of the protected intermediate, and concludes with an acid-catalyzed hydrolysis to release the final dimer. Each step has been optimized to balance reaction kinetics with product stability, ensuring that the overall process remains robust even when scaling up. For detailed operational parameters, reagent ratios, and specific workup procedures that guarantee reproducible high-purity results, please refer to the standardized synthesis guide below.
- Perform acetalization of glycerol with benzaldehyde using an acid catalyst and water-carrying agent to form glycerol benzaldehyde acetal ether.
- Oxidize the secondary alcohol of the acetal intermediate using chromium-based reagents or TEMPO catalytic systems to form the ketone.
- Hydrolyze the oxidized intermediate using a solid acid catalyst in aqueous conditions to release the final 1,3-dihydroxyacetone dimer.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers compelling economic advantages primarily driven by the utilization of glycerol, a commodity chemical available at historically low prices due to the biodiesel boom. By transforming a low-cost bulk chemical into a high-value fine chemical, manufacturers can achieve substantial margin improvements without relying on volatile petrochemical feedstocks. Furthermore, the process design inherently supports cost reduction through the implementation of solid acid catalysts in the hydrolysis step. Unlike liquid mineral acids which require neutralization and generate large volumes of saline wastewater, solid acid resins can be filtered, regenerated, and reused multiple times. This capability significantly lowers the operational expenditure related to waste treatment and raw material consumption, directly impacting the bottom line.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts typically required for direct glycerol oxidation represents a major capital and operational cost saving. By utilizing base metal catalysts or organic nitroxyl radicals like TEMPO in conjunction with air or oxygen, the process avoids the financial risks associated with platinum group metal price fluctuations and loss. Additionally, the high selectivity of the protection strategy minimizes the formation of hard-to-separate byproducts, reducing the solvent and energy load required for purification columns and crystallization steps, leading to a leaner and more cost-effective manufacturing process.
- Enhanced Supply Chain Reliability: Sourcing high-purity 1,3-dihydroxyacetone can often be challenging due to the limited number of producers capable of meeting strict pharmacopeial standards. This robust chemical synthesis route enhances supply security by enabling production in standard fine chemical facilities without the need for specialized bioreactors or fermentation suites. The reliance on widely available industrial solvents and reagents ensures that production schedules are not disrupted by niche supply shortages. Moreover, the ability to recycle the benzaldehyde protecting group further insulates the supply chain from raw material volatility, ensuring consistent availability for downstream customers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations such as azeotropic distillation and filtration that are well-understood and easily scaled from pilot to commercial tonnage. The shift towards solid acid catalysts and the potential for aerobic oxidation aligns with modern green chemistry principles, reducing the environmental footprint of the manufacturing site. This compliance with increasingly stringent environmental regulations mitigates the risk of production shutdowns due to regulatory non-compliance and positions the manufacturer as a sustainable partner for global corporations with aggressive ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,3-dihydroxyacetone synthesized via this patented method. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring that stakeholders have accurate information for decision-making. Understanding these nuances is critical for R&D teams evaluating process feasibility and procurement officers assessing supplier capabilities.
Q: Why is this chemical synthesis route preferred over enzymatic methods for 1,3-Dihydroxyacetone?
A: Enzymatic methods often suffer from limitations in glycerol concentration due to fungal inhibition, leading to complex post-treatment and lower overall yields. This chemical route utilizes a protective group strategy that allows for higher substrate concentrations and simplifies purification, achieving product purity up to 99%.
Q: How does the use of solid acid catalysts impact production costs?
A: The patent highlights the use of solid acid cation exchange resins (like CD550 or D072) in the hydrolysis step. These catalysts can be easily separated via filtration and reused multiple times, significantly reducing catalyst consumption costs and minimizing acidic wastewater treatment requirements compared to traditional liquid mineral acids.
Q: What are the primary applications for the 1,3-Dihydroxyacetone produced via this method?
A: The high-purity 1,3-Dihydroxyacetone (DHA) produced is suitable for use as a critical pharmaceutical intermediate, a food additive, and notably as the active ingredient in cosmetic sunless tanning products. Its high purity profile also supports applications in antiviral research and food preservation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Dihydroxyacetone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient glycerol valorization in the modern chemical supply chain. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN101412706B are successfully translated into robust industrial realities. Our state-of-the-art facilities are equipped to handle the specific requirements of acetalization and selective oxidation chemistries, maintaining stringent purity specifications and rigorous QC labs to guarantee that every batch of 1,3-dihydroxyacetone meets the highest international standards for pharmaceutical and cosmetic applications.
We invite global partners to collaborate with us to leverage this advanced technology for their specific product lines. Whether you require custom synthesis services or bulk supply of high-purity intermediates, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive fine chemicals market.
