Industrial Scale-Up of Beta-Nicotinamide Riboside Chloride via Novel Condensation Route
Introduction to Advanced NRC Synthesis Technology
The escalating global demand for NAD+ precursors, driven by extensive research into anti-aging mechanisms and metabolic health, has placed beta-nicotinamide riboside chloride (NRC) at the forefront of pharmaceutical intermediate manufacturing. As detailed in patent CN110642897A, a groundbreaking preparation method has been developed to address the critical bottlenecks of cost, efficiency, and scalability that have historically plagued the production of this vital compound. This technical insight report analyzes the novel synthetic route which transitions from laboratory-scale curiosity to a robust, industrial-grade process capable of supporting the burgeoning nutraceutical and regenerative medicine sectors. By leveraging a strategic sequence of chlorination, condensation, and deacetylation, this methodology offers a reliable beta-nicotinamide riboside chloride supplier pathway that ensures consistent quality and supply continuity for downstream manufacturers.
The significance of this technological advancement cannot be overstated, as NRC serves as a direct precursor to Nicotinamide Mononucleotide (NMN) and ultimately Nicotinamide Adenine Dinucleotide (NAD+), molecules essential for cellular energy metabolism and DNA repair. Traditional synthesis routes often suffer from low yields and complex purification requirements that render them economically unviable for mass production. In contrast, the disclosed invention provides a streamlined approach that not only enhances the overall yield but also simplifies the post-reaction treatment, thereby facilitating the industrialized tonnage production required to meet market demands. This report delves deep into the mechanistic advantages and commercial implications of adopting this superior synthetic strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of beta-nicotinamide riboside chloride was predominantly achieved through methods that, while chemically valid, were operationally inefficient for large-scale application. One prevalent literature method involves the condensation of tetraacetyl ribose with ethyl nicotinate, followed by ammonolysis to generate the final product. 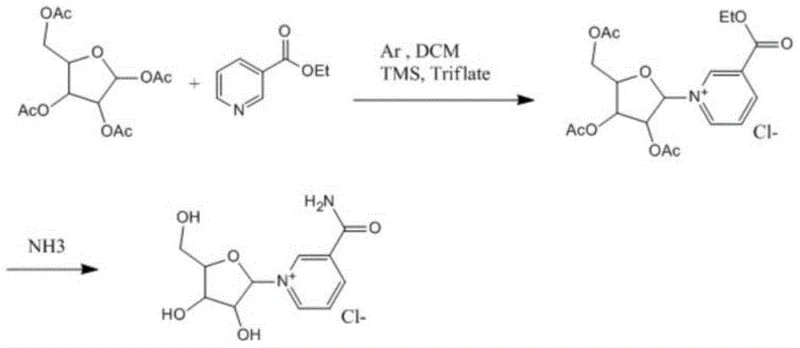 This traditional pathway is fraught with significant drawbacks, including notoriously low reaction yields and arduous post-reaction processing steps that often necessitate the use of chromatographic columns and freeze-drying techniques. These requirements not only inflate the operational expenditure due to the high consumption of solvents and stationary phases but also introduce substantial delays in production cycles, making it difficult to achieve the throughput necessary for a cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the reliance on such complex purification methods often leads to variability in product quality, posing risks to supply chain stability for high-volume buyers.
This traditional pathway is fraught with significant drawbacks, including notoriously low reaction yields and arduous post-reaction processing steps that often necessitate the use of chromatographic columns and freeze-drying techniques. These requirements not only inflate the operational expenditure due to the high consumption of solvents and stationary phases but also introduce substantial delays in production cycles, making it difficult to achieve the throughput necessary for a cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the reliance on such complex purification methods often leads to variability in product quality, posing risks to supply chain stability for high-volume buyers.
The Novel Approach
The patented methodology introduces a paradigm shift by utilizing halogenated acetyl ribose and nicotinamide as the primary starting materials, fundamentally altering the reaction landscape to favor high efficiency and ease of purification. 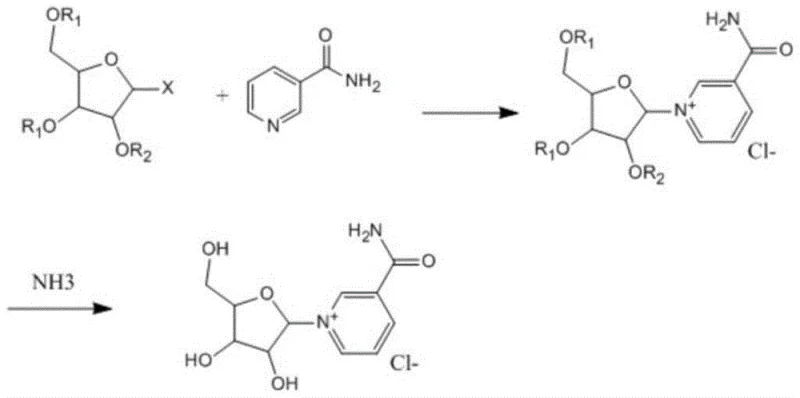 In this optimized route, the condensation step directly couples the activated sugar moiety with the nicotinamide base, bypassing the need for ester intermediates and their subsequent conversion. This direct approach results in a cleaner reaction profile where the desired beta-isomer is favored, and the resulting intermediate can be isolated through simple crystallization rather than chromatography. The elimination of lyophilization and column chromatography represents a monumental leap forward in process chemistry, drastically simplifying the workflow and enabling the commercial scale-up of complex pharmaceutical intermediates. This novel approach ensures that the final product is obtained with high purity and stability, ready for immediate downstream processing into NMN or other NAD+ boosters.
In this optimized route, the condensation step directly couples the activated sugar moiety with the nicotinamide base, bypassing the need for ester intermediates and their subsequent conversion. This direct approach results in a cleaner reaction profile where the desired beta-isomer is favored, and the resulting intermediate can be isolated through simple crystallization rather than chromatography. The elimination of lyophilization and column chromatography represents a monumental leap forward in process chemistry, drastically simplifying the workflow and enabling the commercial scale-up of complex pharmaceutical intermediates. This novel approach ensures that the final product is obtained with high purity and stability, ready for immediate downstream processing into NMN or other NAD+ boosters.
Mechanistic Insights into the Chlorination-Condensation-Deacetylation Sequence
The core of this synthetic breakthrough lies in the precise control of reaction conditions during the initial chlorination and subsequent glycosidic bond formation. The process initiates with the chlorination of tetraacetyl ribose in a solvent system such as dichloromethane or chloroform, where hydrogen chloride gas is introduced at controlled low temperatures ranging from -5 to 20°C. This step generates a reactive chlorotriacetyl ribose intermediate, which is crucial for the subsequent nucleophilic attack by the nicotinamide nitrogen. The careful removal of byproduct acetic acid via reduced pressure distillation ensures that the reaction equilibrium is driven forward, maximizing the conversion of the starting material and minimizing the formation of unwanted side products that could complicate downstream purification. This meticulous attention to intermediate preparation sets the stage for a highly efficient condensation reaction.
Following the formation of the activated sugar, the condensation with nicotinamide is conducted under an inert nitrogen atmosphere at elevated temperatures between 40 and 90°C. This thermal activation facilitates the formation of the N-glycosidic bond, creating the nicotinamide triacetyl ribose intermediate with high stereo-selectivity for the beta-configuration. The final deacetylation step utilizes ammonia gas or saturated ammonia solutions in alcohols like methanol or ethanol at mild temperatures, effectively stripping the acetyl protecting groups without degrading the sensitive glycosidic linkage. The impurity control mechanism is further reinforced by the final refining step, which employs a specific mixed solvent system of ethanol and water to induce crystallization, effectively excluding residual solvents and minor impurities to achieve a final purity specification that exceeds 99.3%.
How to Synthesize Beta-Nicotinamide Riboside Chloride Efficiently
Implementing this synthesis route requires strict adherence to the specified parameters regarding temperature, solvent ratios, and reaction times to ensure optimal yield and purity. The process is designed to be robust, allowing for variations in scale from laboratory benchtop to multi-hundred-kilogram batches without compromising the integrity of the final product. Operators must pay particular attention to the moisture control during the chlorination and condensation steps, as the presence of water can lead to hydrolysis of the activated intermediates. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for technical teams aiming to replicate this high-efficiency production method.
- Chlorination of tetraacetyl ribose using hydrogen chloride gas in solvent A at -5 to 20°C to form chlorotriacetyl ribose.
- Condensation of the chlorinated intermediate with nicotinamide in solvent C at 40-90°C under nitrogen protection.
- Deacetylation reaction using ammonia gas in solvent E at -10 to 20°C to remove acetyl protecting groups.
- Refining the crude product via crystallization in a mixed solvent system of ethanol and water to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method translates into tangible strategic advantages that extend far beyond simple chemical transformation. The most significant impact is observed in the realm of cost reduction in pharmaceutical intermediates manufacturing, driven primarily by the elimination of expensive and time-consuming purification technologies. By replacing chromatographic separation with crystallization, the process drastically reduces the consumption of high-grade solvents and silica gel, leading to a substantial decrease in raw material costs and waste disposal fees. Furthermore, the simplified workflow shortens the overall production cycle time, allowing facilities to increase their throughput and respond more agilely to fluctuating market demands for NAD+ precursors.
- Cost Reduction in Manufacturing: The economic benefits of this process are profound, as the removal of column chromatography and freeze-drying steps eliminates two of the most capital-intensive operations in fine chemical synthesis. This streamlining allows for a significant optimization of operational expenditures, as the facility no longer needs to maintain complex purification infrastructure or manage the hazardous waste streams associated with large-scale chromatography. Additionally, the high yield of the chlorination and condensation steps ensures that the expensive starting material, tetraacetyl ribose, is utilized with maximum efficiency, further driving down the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this synthetic route offers unparalleled reliability for long-term sourcing agreements. The use of readily available and stable raw materials, such as nicotinamide and tetraacetyl ribose, mitigates the risk of supply disruptions that are often associated with exotic or specialized reagents. Moreover, the ability to produce the intermediate nicotinamide triacetyl ribose in a stable, storable form allows manufacturers to build strategic inventory buffers, ensuring reducing lead time for high-purity pharmaceutical intermediates even during periods of peak demand or raw material scarcity.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, as evidenced by the successful execution of 200kg batch sizes in the patent examples, demonstrating its readiness for tonnage-level commercial production. This scalability is matched by a favorable environmental profile, as the reduction in solvent usage and the avoidance of heavy metal catalysts or toxic reagents align with increasingly stringent global environmental regulations. The ability to recover and recycle solvents like ethanol and dichloromethane further enhances the sustainability of the operation, making it an attractive option for companies committed to green chemistry principles and corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and quality of beta-nicotinamide riboside chloride using this advanced methodology. These insights are derived directly from the experimental data and process descriptions found in the source patent, providing a factual basis for decision-making. Understanding these details is crucial for technical buyers evaluating the feasibility of integrating this intermediate into their own supply chains.
Q: Does this synthesis method require column chromatography?
A: No, the patented process eliminates the need for chromatographic purification by utilizing specific crystallization techniques, significantly reducing production costs and time.
Q: What is the achievable purity of the final beta-nicotinamide riboside chloride?
A: The method consistently yields products with purity exceeding 99.3% as confirmed by HPLC analysis, suitable for pharmaceutical and nutraceutical applications.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly demonstrates successful scale-up from kilogram to 200kg batches, proving its viability for tonnage-level commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Nicotinamide Riboside Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of key pharmaceutical intermediates like beta-nicotinamide riboside chloride. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of global nutraceutical and pharmaceutical clients. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of NRC we deliver meets the highest standards for safety and efficacy required for human consumption.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this superior manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance your product quality and strengthen your market position in the rapidly growing NAD+ sector.
