Advanced Manufacturing of High-Purity Cordycepin via Optimized Reductive Debromination for Global Pharma Supply Chains
Advanced Manufacturing of High-Purity Cordycepin via Optimized Reductive Debromination for Global Pharma Supply Chains
The global demand for nucleoside analogs, particularly cordycepin (3'-deoxyadenosine), has surged due to its potent antiviral, antitumor, and immunomodulatory properties. As a critical pharmaceutical intermediate, the consistent supply of high-quality cordycepin is paramount for drug development pipelines worldwide. A pivotal advancement in this domain is detailed in patent CN103709221A, which discloses a novel, semi-synthetic preparation method starting from adenosine. This technology represents a significant leap forward in process chemistry, addressing long-standing challenges related to safety, yield, and environmental impact. By leveraging a streamlined two-step sequence involving regioselective bromination followed by mild reductive debromination, this method offers a robust alternative to hazardous traditional routes. For R&D directors and procurement strategists, understanding the nuances of this patent is essential for securing a reliable cordycepin supplier capable of meeting rigorous GMP standards while optimizing production economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cordycepin has been plagued by significant operational hazards and inefficiencies that hinder scalable manufacturing. Prior art, such as the methods reported in Tetrahedron Letters (2004) and Synlett (2008), frequently relies on aggressive reducing agents like lithium triethylborohydride (Super-Hydride) or lithium aluminum hydride (LiAlH4). These reagents are notoriously pyrophoric, posing severe fire risks and requiring specialized, expensive infrastructure for safe handling and storage. Furthermore, these conventional routes often involve lengthy multi-step sequences with poor atom economy, leading to substantial waste generation and complex purification protocols. The use of such dangerous reagents not only inflates the cost of goods sold (COGS) due to safety compliance measures but also introduces significant supply chain vulnerabilities, as the logistics of transporting hazardous chemicals are heavily regulated and prone to delays.
The Novel Approach
In stark contrast, the methodology outlined in patent CN103709221A introduces a paradigm shift towards safer and more efficient pharmaceutical intermediates manufacturing. The core innovation lies in the conversion of adenosine to a 4-bromo intermediate (Compound I) using acetoxyisobutyryl bromide, followed by a controlled reduction. This approach eliminates the need for pyrophoric hydrides, substituting them with safer alternatives like sodium bis(2-methoxyethoxy)aluminum hydride (Red-Al) or catalytic hydrogenation over Pd/C. The reaction conditions are remarkably mild, often proceeding at room temperature or under simple ice-bath cooling, which drastically simplifies reactor requirements. This transition from hazardous to benign chemistry not only enhances operator safety but also streamlines the downstream processing, making the isolation of the final product far more straightforward and cost-effective.
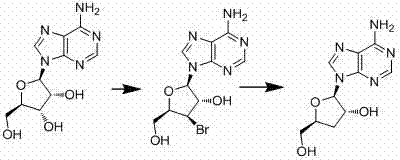
Mechanistic Insights into Reductive Debromination Strategy
The chemical elegance of this process is rooted in its precise control over stereochemistry and regioselectivity. The first step involves the reaction of adenosine with acetoxyisobutyryl bromide in a solvent system comprising acetonitrile and water. This specific reagent system facilitates the selective substitution of the 3'-hydroxyl group's neighboring functionality, ultimately yielding the key intermediate, (2R,3S,4R,5R)-2-(6-amino-9H-purin-9-yl)-4-bromo-5(hydroxymethyl)tetrahydrofuran-3-ol. The presence of water in the initial step is critical, acting as a co-solvent to modulate the reactivity of the brominating agent, ensuring high conversion rates without degrading the sensitive purine base. The formation of the 4-bromo species sets the stage for the subsequent deoxygenation, effectively installing a leaving group that can be cleanly removed in the next phase.
The second mechanistic phase involves the reductive removal of the bromine atom to restore the 3'-deoxy configuration characteristic of cordycepin. When using Red-Al as the reducing agent, the reaction proceeds via a hydride transfer mechanism that selectively targets the carbon-bromine bond while preserving the integrity of the glycosidic bond and the exocyclic amine. Alternatively, the patent describes a catalytic hydrogenation route using Pd/C, which offers an even greener profile by utilizing molecular hydrogen. This step is conducted at low temperatures (3-5°C) initially to control exothermicity, followed by stirring at room temperature to ensure complete conversion. The result is a crude product that requires minimal purification, typically just recrystallization from ethanol, to achieve high-purity cordycepin suitable for further pharmaceutical development.
How to Synthesize Cordycepin Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters defined in the patent to maximize yield and purity. The process is designed to be operationally simple, avoiding the need for cryogenic temperatures or ultra-high vacuum systems that complicate scale-up. The following guide outlines the critical operational phases derived from the patent examples, focusing on the transition from raw adenosine to the final crystalline product. For detailed standard operating procedures (SOPs) and specific batch records, please refer to the technical documentation provided below.
- React adenosine with acetoxyisobutyryl bromide in acetonitrile/water to form the 4-bromo intermediate (Compound I).
- Reduce Compound I using sodium bis(2-methoxyethoxy)aluminum hydride (Red-Al) or catalytic hydrogenation to yield crude cordycepin.
- Purify the crude product via ethanol recrystallization to obtain pharmaceutical-grade cordycepin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates directly into tangible business value and risk mitigation. The shift away from hazardous reagents like LiAlH4 significantly reduces the regulatory burden and insurance premiums associated with chemical manufacturing. By utilizing Red-Al or catalytic hydrogenation, facilities can operate with lower safety overheads, and the waste streams generated are less toxic and easier to treat, aligning with modern environmental, social, and governance (ESG) goals. Furthermore, the high yields reported in the patent examples—reaching up to 97% for the intermediate and over 93% for the final product—imply a drastic reduction in raw material consumption per kilogram of output, driving down the variable costs of production substantially.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous reagents creates a leaner cost structure. Traditional methods often incur hidden costs related to special waste disposal and safety containment; this new route mitigates those expenses entirely. Additionally, the use of common solvents like acetonitrile, ethyl acetate, and ethanol ensures that solvent recovery and recycling are economically viable, further enhancing the margin profile for cost reduction in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: Starting from adenosine, a commodity fermentation product, ensures a stable and abundant supply of raw materials. Unlike complex synthetic building blocks that may have single-source suppliers, adenosine is produced globally at multi-ton scales. This foundational stability allows for better forecasting and inventory management, reducing the risk of production stoppages due to raw material shortages and ensuring consistent commercial scale-up of complex nucleoside analogs.
- Scalability and Environmental Compliance: The process conditions are inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-ton industrial reactors without requiring fundamental changes in equipment. The absence of heavy metal catalysts (in the Red-Al variant) or the use of recoverable Pd/C simplifies the purification process, reducing the environmental footprint. This ease of scale-up supports reducing lead time for high-purity nucleosides, allowing manufacturers to respond rapidly to market demand fluctuations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cordycepin synthesis technology. These insights are derived directly from the experimental data and claims within patent CN103709221A, providing a factual basis for decision-making. Understanding these details is crucial for evaluating the feasibility of integrating this process into existing manufacturing portfolios.
Q: How does this new method improve safety compared to traditional lithium aluminum hydride reductions?
A: Traditional methods often utilize lithium aluminum hydride (LiAlH4) or lithium triethylborohydride, which are pyrophoric and require stringent anhydrous conditions. The patented process utilizes Red-Al or catalytic hydrogenation, which are significantly safer to handle on an industrial scale, reducing operational risks and insurance costs.
Q: What are the expected yields for the commercial scale-up of this cordycepin synthesis?
A: According to the experimental data in patent CN103709221A, the process demonstrates exceptional efficiency. The intermediate formation yields up to 97%, and the final reduction step achieves yields exceeding 93%, resulting in a highly competitive overall process yield suitable for large-scale manufacturing.
Q: Is the starting material adenosine readily available for continuous supply chains?
A: Yes, adenosine is a widely available fermentation product produced at multi-ton scales globally. Using adenosine as the starting material ensures robust supply chain continuity and mitigates the risk of raw material shortages often associated with complex synthetic building blocks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cordycepin Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of cordycepin in the development of next-generation antiviral and oncology therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial reality is seamless. We are committed to delivering stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the identity and potency of every batch. Our facility is equipped to handle the specific solvent systems and reduction chemistries required by this patent, guaranteeing a supply of material that meets the highest international regulatory standards.
We invite you to collaborate with us to leverage this advanced synthesis technology for your pipeline. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
