Advanced Enzymatic Synthesis of Destetrazolium Cefotiam for High-Purity API Impurity Standards
The pharmaceutical industry faces increasing regulatory scrutiny regarding the identification and quantification of impurities in active pharmaceutical ingredients (APIs), particularly for critical antibiotics like Cefotiam Hydrochloride. Patent CN107964558B introduces a groundbreaking preparation method for Destetrazolium Cefotiam, a key known impurity that serves as an essential reference standard for quality control. This technology addresses a significant bottleneck in drug development where the lack of stable, high-purity impurity standards hinders effective safety profiling and regulatory approval. By shifting from traditional, often unstable synthesis routes to a refined enzymatic process, this innovation ensures the availability of reference materials with purity exceeding 98 percent, thereby facilitating more accurate adverse reaction tracing and compliance with international pharmacopoeia standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cephalosporin impurities like Destetrazolium Cefotiam has relied on aggressive chemical deacetylation methods that frequently suffer from poor selectivity and harsh reaction conditions. Conventional routes often involve strong acids or bases at elevated temperatures, which can lead to the degradation of the sensitive beta-lactam ring, resulting in complex impurity profiles that are difficult to separate. Furthermore, the traditional handling of reactive intermediates such as aminothiazole acetyl chloride typically requires isolation and drying steps that not only consume significant energy but also expose the thermally unstable acid chloride to decomposition, lowering overall yields and increasing raw material costs. These inefficiencies create a supply chain vulnerability where high-purity reference standards become prohibitively expensive and difficult to source reliably for routine QC testing.
The Novel Approach
The novel approach detailed in the patent data revolutionizes this landscape by integrating a mild enzymatic deacetylation step and a streamlined wet-feeding strategy for reactive intermediates. Instead of isolating the aminothiazole acetyl chloride, the process utilizes the wet product directly from the crystallization mother liquor, preserving its reactivity and eliminating thermal degradation risks associated with drying. The core innovation lies in the application of Cephalosporin C deacetylase, which operates under physiological pH and temperature conditions to selectively remove the acetyl group without compromising the integrity of the cephalosporin nucleus. This biological catalysis not only enhances the stereochemical purity of the final product but also drastically simplifies the downstream purification process, offering a robust solution for the commercial production of high-value impurity standards.
Mechanistic Insights into Enzymatic Deacetylation and Acylation
The synthetic pathway begins with the activation of aminothiazole acetic acid hydrochloride using phosphorus oxychloride (POCl3) and dimethylformamide (DMF) to generate the reactive acid chloride species in situ. This activation step is critical for the subsequent acylation of the 7-aminocephalosporanic acid (7-ACA) nucleus. The reaction is meticulously controlled at low temperatures, typically between -5°C and 0°C, to prevent side reactions and ensure the formation of the desired 7-aminothiazole acetamido cephalosporanic acid intermediate. The use of protective agents such as trimethylchlorosilane or hexamethyldisilazane further stabilizes the reactive sites on the 7-ACA molecule, ensuring that acylation occurs exclusively at the 7-amino position while leaving the other functional groups intact for the subsequent enzymatic transformation.
Following the acylation, the process transitions to an aqueous system where the enzymatic magic occurs. Cephalosporin C deacetylase acts as a highly specific biocatalyst that hydrolyzes the ester bond at the 3'-position of the cephalosporin ring. Unlike chemical hydrolysis which lacks specificity and can attack the beta-lactam amide bond, this enzyme recognizes the specific steric and electronic environment of the acetyl group, cleaving it efficiently at a neutral pH of 7.3 to 7.7. This specificity is paramount for maintaining the structural integrity of the molecule, preventing the formation of ring-opened byproducts that would render the reference standard useless for HPLC calibration. The result is a clean conversion to Destetrazolium Cefotiam with minimal byproduct formation, as illustrated in the reaction scheme below.
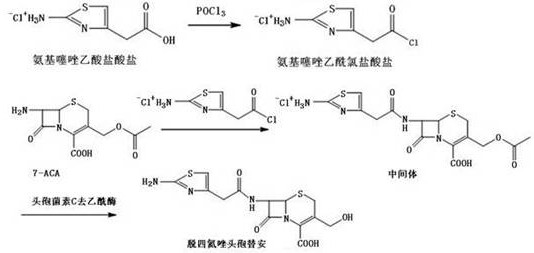
How to Synthesize Destetrazolium Cefotiam Efficiently
Implementing this synthesis route requires precise control over reaction parameters, particularly temperature and pH, to maximize the efficiency of both the chemical acylation and the enzymatic deacetylation steps. The process is designed to be scalable, moving seamlessly from laboratory glassware to industrial reactors by maintaining the specific molar ratios of protective agents and solvents described in the patent examples. Operators must ensure that the wet aminothiazole acetyl chloride is handled under inert atmosphere conditions to prevent hydrolysis before it reacts with the 7-ACA. The following guide outlines the standardized operational procedure derived from the patent data, ensuring reproducibility and high yield for commercial manufacturing teams.
- Prepare aminothiazole acetyl chloride by reacting aminothiazole acetic acid hydrochloride with POCl3 and DMF in an organic solvent at low temperature (-5°C to 0°C), followed by crystallization and wet filtration.
- Synthesize the intermediate 7-aminothiazole acetamido cephalosporanic acid by reacting 7-ACA with a protective agent and the wet aminothiazole acetyl chloride, controlling pH and temperature carefully.
- Perform enzymatic deacetylation using Cephalosporin C deacetylase on the intermediate in an aqueous system at mild temperatures (25-35°C) to yield the final Destetrazolium Cefotiam product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic synthesis route presents a compelling value proposition centered on cost stability and supply reliability. The elimination of the drying step for the acid chloride intermediate translates directly into reduced energy consumption and shorter cycle times, allowing for faster turnover of production batches. Moreover, the high selectivity of the enzymatic step minimizes the generation of hazardous waste and reduces the burden on wastewater treatment facilities, aligning with increasingly strict environmental regulations that often dictate operational continuity in the fine chemical sector. These process intensifications collectively lower the total cost of ownership for producing this critical impurity standard.
- Cost Reduction in Manufacturing: The strategic decision to utilize wet aminothiazole acetyl chloride eliminates the capital and operational expenditures associated with drying equipment and the energy required for solvent removal. By avoiding the thermal stress of drying, the process preserves the potency of the raw material, effectively reducing the stoichiometric requirement of the expensive aminothiazole acetic acid hydrochloride. This material efficiency, combined with the high yield of the enzymatic step, drives down the variable cost per kilogram, making the sourcing of high-purity Destetrazolium Cefotiam significantly more economical compared to legacy chemical methods.
- Enhanced Supply Chain Reliability: The robustness of the enzymatic process ensures consistent batch-to-batch quality, reducing the risk of production failures that can disrupt the supply of critical reference standards. Because the reaction conditions are mild and do not rely on extreme temperatures or pressures, the process is less susceptible to equipment downtime or safety incidents that often plague aggressive chemical syntheses. This operational stability allows suppliers to maintain tighter delivery schedules and provide reliable inventory levels, which is crucial for pharmaceutical companies managing strict regulatory timelines for drug submissions and annual product reviews.
- Scalability and Environmental Compliance: The transition to a biocatalytic step significantly improves the environmental profile of the manufacturing process by reducing the use of harsh chemical reagents and organic solvents in the final purification stages. The aqueous nature of the enzymatic reaction simplifies work-up procedures and facilitates easier waste management, ensuring compliance with green chemistry principles. This scalability is further supported by the commercial availability of the enzyme and the straightforward nature of the upstream acylation, enabling manufacturers to ramp up production volumes from pilot scale to multi-ton annual capacity without encountering significant engineering bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Destetrazolium Cefotiam. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, offering clarity on the feasibility and benefits of this advanced synthesis route for industry stakeholders.
Q: Why is the enzymatic method preferred for synthesizing Destetrazolium Cefotiam?
A: The enzymatic method using Cephalosporin C deacetylase offers mild reaction conditions (25-35°C) and high regioselectivity, significantly reducing product decomposition and impurity formation compared to harsh chemical deacetylation methods.
Q: How does the wet feeding technique impact production costs?
A: Using wet aminothiazole acetyl chloride directly eliminates the energy-intensive drying step, reduces raw material consumption by avoiding degradation during drying, and simplifies the workflow, leading to substantial operational cost savings.
Q: What purity levels can be achieved with this patented process?
A: The process described in patent CN107964558B consistently achieves a final product purity of greater than 98.0%, meeting the stringent requirements for impurity reference substances in pharmaceutical quality control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Destetrazolium Cefotiam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity impurity standards play in ensuring the safety and efficacy of antibiotic therapies. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply of Destetrazolium Cefotiam is both secure and consistent. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for global regulatory submissions and quality control laboratories.
We invite you to collaborate with our technical team to explore how this optimized synthesis route can enhance your supply chain efficiency. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and manufacturing goals, ensuring you have a reliable partner for your complex pharmaceutical intermediate needs.
